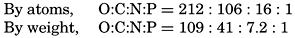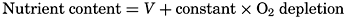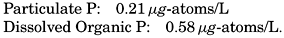VII. Organisms and the Composition of Sea Water [PDF]
Chemical Composition of Marine Organisms
Alterations of the concentration of the dissolved constituents of sea water are brought about by the development and subsequent death and disintegration of organisms. Virtually all of the substances extracted from the water are returned to solution by metabolic processes or by disintegration of the organisms, but the elements removed are returned to solution at some later time and often in some other part of the water column. Hence, the modifications may be in opposite directions at different times and localities. A small fraction of the organic remains accumulates on the sea bottom and is lost to the cycle.
Sea water probably contains in solution all of the chemical elements, although only some fifty have yet been detected. There is a large amount of data on the occurrence of various elements in marine plants and animals, but unfortunately the material is far from complete for any one biological group. Either a few elements only have been determined—such as iodine, for example, which has been thoroughly investigated—or only a portion of the organisms—for example, the skeletal structures—has been analyzed.
Vinogradov (1935, 1937) has compiled the chemical analyses of the lower plants and animals, both aquatic and terrestrial. He reports some sixty of the elements that have been found in one or more species. Webb and Fearon (1937) have tabulated thirty-nine elements that are commonly found and have divided these into two groups according to their apparent importance to living things: (1) eighteen invariable elements, and (2) twenty-one variable elements. These classes are further sub-divided on the basis of the concentration in which the elements are present. Seven elements are listed as contaminants (table 46).
The primary invariable elements are the essential constituents of carbohydrates, lipides (fats), and proteins. Some of the invariable elements classed as secondary or as microconstituents are always present in the lipides and proteins. This list is for plants and animals in general and not for marine forms alone. Comparison of tables 46 and 36 shows that nine elements (starred) detected in organisms have not yet been
| Invariable (18) | Variable (21) | Contaminants | |||
|---|---|---|---|---|---|
| Primary 1–60% | Secondary 0.05–1% | Microconstituents <0.05% | Secondary | Microconstituents | |
| Hydrogen | Sodium | Boron | Titanium[*] | Lithium | Helium |
| Carbon | Magnesium | Fluorine | Vanadium | Beryllium[*] | Argon |
| Nitrogen | Sulphur | Silicon | Zinc | Aluminum | Selenium |
| Oxygen | Chlorine | Manganese | Bromine | Chromium[*] | Gold |
| Phosphorus | Potassium | Copper | Cobalt[*] | Mercury | |
| Calcium | Iodine | Nickel | Bismuth[*] | ||
| Iron | Germanium[*] | Thallium[*] | |||
| Arsenic | |||||
| Rubidium | |||||
| Strontium | |||||
| Molybdenum | |||||
| Silver | |||||
| Cadmium[*] | |||||
| Tin[*] | |||||
| Cesium | |||||
| Barium | |||||
| Lead | |||||
The lack of comparable data for the different types of organisms makes it necessary to consider their composition under three headings—namely, organic material (largely carbohydrates, lipides, and proteins), inorganic skeletal structures, and inorganic solutes in the body fluids. Although the proportions of carbohydrates, lipides, and proteins may vary considerably, the composition of any one type is rather constant, so that the average values in table 47 can be used with some confidence. Furthermore, there are numerous determinations of lipides (ether extract) and protein (based on nitrogen determinations), and from these measurements and the loss on ignition the carbohydrate may be computed. Skeletal structures differ so much in composition and in their mass, compared to that of the organic material, that they must be considered separately. Inorganic solutes in the body fluids are considered as a separate class, because they apparently do not differ very much in composition
| Percentage composition | Relative proportions by weight, C = 100 | |||||||
|---|---|---|---|---|---|---|---|---|
| Element | Carbohydrates | Lipides | Proteins | Element | Sea water | Lipides | Proteins | |
| O | 49.38 | 17.90 | 22.4 | C | 100 | 100 | 100 | |
| C | 44.14 | 69.05 | 51.3 | P | 0.05 | 3.1 | 1.4 | |
| H | 6.18 | 10.00 | 6.9 | N | 0.5 | 0.88 | 34.7 | |
| P | 2.13 | 0.7 | S | 3150 | 0.45 | 1.6 | ||
| N | 0.61 | 17.8 | Fe | 0.07 | 0.2 | |||
| S | 0.31 | 0.8 | ||||||
| Fe | 0.1 | |||||||
In table 47 are given the average compositions of the three great classes of organic material (Rogers, 1938) and the relative proportions in which their component elements occur in sea water. The oxygen and hydrogen are not considered, and the values are adjusted to C = 100. The values for C, S, and Fe are from table 36; those for N and P are the average winter values in the English Channel (pp. 252, 258). In the lipides, phosphorus is concentrated, and in the proteins the nitrogen and phosphorus show a great increase with respect to carbon. The fact that sulphur, which is one of the relatively abundant elements in sea water, is a minor constituent of the lipides and proteins in organic material indicates that carbon, here used as the reference element, is itself markedly concentrated. The values given in table 47 are general averages, and those for marine organisms may differ slightly. It should be noted that changes in the proportions of carbohydrates, lipides, and proteins will modify the ratios in which the above-mentioned elements will be removed from the water. Many of the other elements that are concentrated by organisms—for example, iodine, iron, and copper—probably form a part of the organic material or they occur in the skeletal structures, as it is difficult to see how the free ions could be retained in the body fluids
In table 48 are given analyses of certain types of skeletal material. In each case there is some organic matter, which is highest in the lobster carapace, and even in the phosphatic brachiopod shell it forms a large fraction. Of course, there are wide ranges in the proportion of inorganic skeletal structures in the whole organism, and in some cases such structures may be entirely lacking. The first three examples are for calcareous types with CaCO3 predominating, but in some groups MgCO3 forms an important part of the shell. The lobster may be considered as representative of the arthropods in general, although the proportion of organic matter is probably even greater in the small forms. The amount of phosphate is notable in the lobster and even more so in the phosphatic brachiopod shell, which is predominantly calcium phosphate. The sponge spicules are virtually pure hydrated silica and may be taken as representative of the diatom and radiolarian skeletons. The silica, iron, and aluminum in the other analyses probably represent impurities introduced by the presence of clay and sand grains. These analyses cannot be regarded as complete, and further examination will undoubtedly reveal many other elements present in small amounts. It should be noted that chlorine and sodium, the two most abundant elements in sea water, are not shown in any of these analyses. These elements form soluble compounds and hence would not be suitable for skeletal structures. From table 48 it can be seen that the development or re-solution of skeletal structures of marine organisms may be expected to affect the concentrations
| Substance | Foraminifera (Orbitolites marginatis) | Coral (Oculina diffusa) | Calcareous alga (Lithophyllum antillarum) | Lobster (Homarus sp.) | Phosphatic brachiopod (Discinisca lamellosa) | Siliceous sponge (Euplectella speciosa) |
|---|---|---|---|---|---|---|
| Ca | 34.90 | 38.50 | 31.00 | 16.80 | 26.18 | 0.16 |
| Mg | 2.97 | 0.11 | 4.36 | 1.08 | 1.45 | 0.00 |
| CO3 | 59.70 | 58.00 | 62.50 | 22.40 | 7.31 | 0.24 |
| SO4 | 0.68 | 0.52 | 4.43 | 0.00 | ||
| PO4 | tr | tr | tr | 5.45 | 34.55 | 0.00 |
| SiO2 | 0.03 | 0.07 | 0.04 | 0.30 | 0.64 | 88.56 |
| (Al,Fe)2O3 | 0.13 | 0.05 | 0.10 | 0.44 | 0.32 | |
| Organic matter, etc | 2.27 | 3.27 | 1.32 | 53.45 | 25.00 | 10.72 |
The relative concentrations of the elements that are abundant in the body fluids do not differ very much from those in sea water (table 49). Although little is known concerning the less abundant elements, it appears that the inorganic portion of the body fluids can be considered as slightly altered sea water. Therefore, this part of the organism cannot play any appreciable part in modifying the composition of the water. Although the composition and concentration of the inorganic solutes is of no particular importance in the present problem, these features have been studied intensively in problems of osmotic pressure relations (chapter VIII) and in connection with the mechanism of solute and water exchange between aquatic organisms and their environment. These fields have been reviewed by Rogers (1938).
| Element | Sea water | Echinus esculentus (Sea urchin) | Homarus vulgaris (Lobster) | Cancer pagurus (Crab) |
|---|---|---|---|---|
| Cl | 180 | 182 | 156 | 156 |
| Na | 100 | 100 | 100 | 100 |
| Mg | 12.1 | 12.0 | 1.5 | 5.7 |
| S in SO4 | 8.4 | 8.5 | 2.2 | 6.7 |
| Ca | 3.8 | 3.9 | 5.0 | 4.8 |
| K | 3.6 | 3.7 | 4.7 | 4.0 |
Thus far only the various fractions of the organisms have been discussed, and it is of interest to consider the composition of the entire plant or animal. As the plants are the primary “consumers” of inorganic material, it would be desirable to know the composition of such important groups as the diatoms and peridinians, but no complete analyses of these forms have been made. What information we have will be discussed below. The data for animals is also far from complete, but in table 50 are given three examples. The relative compositions have been adjusted to Na = 100, and, for comparison, the constituents of sea water are given in the same way. The relatively high proportions of the elements abundant in sea water which occur in the copepod analysis indicate the presence of considerable sea water in the original sample. As Archidoris possesses internal calcareous structures, the calcium content is high. Consequently the proportions of the elements constituting the organic material are rather low in these two cases. It is immediately obvious, however, that the essential constituents of the organic material, such as carbon, nitrogen, and phosphorus, are very high when compared to their relative concentrations in sea water.
| Element | Calanus (Copepod) Vinogradov, 1938 | Fish[a] (Average) | Archidoris britannica (Nudibranch) McCance & Masters, 1937–38 | Sea water | Concentration factors | ||
|---|---|---|---|---|---|---|---|
| Copepod | Fish | Nudibranch | |||||
| Cl | 194 | 180 | 180 | 1.1 | 1.0 | ||
| Na | 100 | 100 | 100 | 100 | 1.0 | 1.0 | 1.0 |
| Mg | 5.6 | 36 | 156 | 12.1 | 0.46 | 3.0 | 12.9 |
| S | 25.9 | 259 | 7.1 | 8.4 | 3.1 | 31 | 0.85 |
| Ca | 7.4 | 52 | 262 | 3.8 | 1.9 | 13.7 | 69 |
| K | 53.7 | 383 | 20 | 3.6 | 15 | 109 | 5.5 |
| Br | 1.7 | 0.6 | 3 | ||||
| C | 1113 | ca 4100 | ca 480 | 0.26 | 4,300 | 15,800 | 1,850 |
| Sr | 11 | 0.12 | 92 | ||||
| Si | 1.3 | 0.001[b] | 13,000 | ||||
| F | 69 | 0.01 | 6,900 | ||||
| N | 280 | 1276 | 107 | 0.001[b] | 280,000 | 1,276,000 | 107,000 |
| P | 24.1 | 256 | 6 | 0.0001[b] | 241,000 | 2,560,000 | 60,000 |
| I | 0.04 | 0.0005 | 80 | ||||
| Fe | 1.3 | 1.3 | 0.23 | 0.0002[c] | 6,000 | 6,000 | 1,000 |
| Mn | 0.0008 | 0.0001[c] | 8 | ||||
| Cu | 0.008 | 0.43 | 0.0001[c] | 80 | 4,300 | ||
If the relative amounts of the various elements in these animals are divided by their relative concentrations in sea water, a series of concentration factors, referred to sodium, are obtained. It will be seen that chlorine would give virtually identical results. The concentration factors range from about unity up to over two million for phosphorus in the fish, and in all three cases they are greatest for nitrogen and phosphorus. If it is assumed that the rates of diffusion of all substances and their rates of absorption by the organisms depend only upon the amounts of the ions in the water, then the concentration factors should be a measure of the time required to accumulate them. Those elements having the highest concentration factors would then be the ones that might limit the rate of growth. The data in table 50 indicate that nitrogen and phosphorus may very well be limiting elements in the sea, although it should be remembered that the examples are for animals that must obtain their supply of these elements either directly or indirectly from the plants. If the total carbon in sea water had been used as the reference element, only nitrogen and phosphorus would have significantly larger factors. But it is obvious that carbon is itself concentrated more than one thousandfold with reference to the major elements in sea water. According to table 50 the relative concentration
| Element | Sea water | Diatoms | Peridinians | Copepods | Concentration factors (referred to carbon) | ||
|---|---|---|---|---|---|---|---|
| Diatoms | Peridinians | Copepods | |||||
| C | 100 | 100 | 100 | 100 | 1 | 1 | 1 |
| N | 0.5[a] | 18.2 | 13.8 | 25.0 | 36 | 28 | 50 |
| P | 0.05[a] | 2.7 | 1.7 | 2.2 | 54 | 34 | 44 |
| Fe | 0.07[b] | 9.6 | 3.4 | 0.13 | 137 | 49 | 2 |
| Ca | 1420 | 12.5 | 2.7 | 0.66 | 0.01 | 0.002 | 0.0005 |
| Si | 0.4[a] | 93.0 | 6.6 | 0.13 | 232 | 16 | 0.3 |
In table 51 are given the relative concentrations of certain elements in diatoms, peridinians, and copepods, adjusted to C = 100. The data for the photosynthetic forms are recomputed from Vinogradov (1935), and for copepods are the same as those given in table 50. The concentration factors for nitrogen and phosphorus are about the same in all three forms. In the diatoms, iron is higher, while silicon has the highest factor, which may indicate that these elements also limit the rate of growth. For the peridinians the factors for nitrogen, phosphorus, and iron are nearly the same.
Interrelations Between Elements Whose Distribution Is Affected by Biological Activity
Because the relative composition of organisms living in the sea differs from that of sea water, their growth will tend to modify the composition of the water. The ultimate regeneration of the inorganic substances by biological processes will return the elements to solution, but the net effects will usually be in opposite directions at different times and in different parts of the water column. Tables 50 and 51 show that certain elements present in the water in low concentrations, such as nitrogen, phosphorus, iron, and silicon, are those removed in the largest relative amounts. The distribution of these elements, known as the plant nutrients, is profoundly affected by biological activity, their concentrations are virtually independent of salinity, and they are commonly referred to as nonconservative, in contrast to those elements that bear a constant ratio to the total dissolved solids.
Plants are the most important “consumers” of the inorganic substances. Their activity is restricted to the upper layers of the sea (the euphotic zone), where there is adequate light for them to carry on photosynthesis. In nearshore areas the thickness of the euphotic layer may be only a few meters, and even in the open sea, where the transparency is great, the growth of plants is restricted to the upper few hundred meters (chapter XVI). Animals living below the euphotic layer may remove elements from solution which are necessary for the secretion of skeletal structures, but most of the materials must come directly or indirectly from plants that develop near the surface. The metabolic activities of the plants, animals, and bacteria return the elements to inorganic form. Part of the regeneration must occur in the euphotic layer, but there is a general downward movement of the particulate matter, either living or dead, and, consequently, a continuous transport of the elements away from the surface layer. As described thus far, it
Redfield (1934), following an earlier suggestion by Harvey, showed that regardless of the absolute concentrations a constant ratio exists between the nitrate-nitrogen and phosphate-phosphorus content of sea water, that these elements are apparently removed from the water by organisms in the same proportions in which they occur, and that on the death and decomposition of the organisms they are returned to solution simultaneously. Cooper (1938a) proposed a modified ratio, pointing out that the phosphorus data for sea water used by Redfield had not been corrected for salt error. Fleming (1940) obtained from examination of additional data a slightly different relationship for the N:P in plankton. All these figures are given in table 52, which also shows the relation of carbon to the other two elements in plankton.
| Source | By weight | By atoms | ||||
|---|---|---|---|---|---|---|
| C | N | P | C | N | P | |
| Redfield (1934) Plankton | 53.2 | 8.2 | 1 | |||
| Redfield (1934) Seawater | 9.0 | 1 | 20 | 1 | ||
| Cooper (1938a) Seawater | 6.8 | 1 | 15 | 1 | ||
| Fleming (1940) Phytoplankton | 42 | 7 | 1 | |||
| Fleming (1940) Zooplankton | 40 | 7.4 | 1 | |||
| Fleming—Average: Plankton. | 41 | 7.2 | 1 | 106 | 16 | 1 |
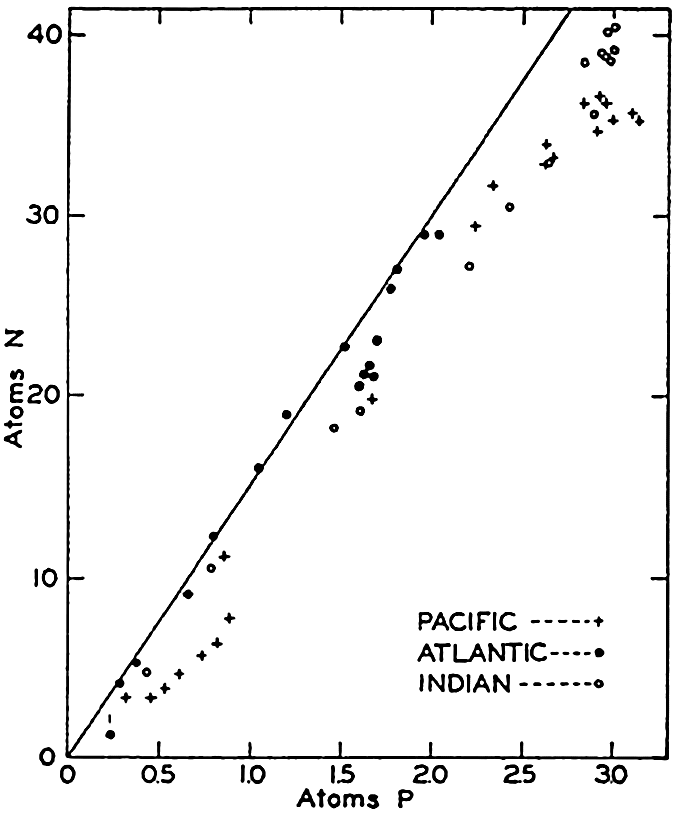
The ratios given above hold very well for the nitrate and phosphate in ocean waters (see fig. 51), but, since they represent the net effect of biological activity, marked deviations from the ratios may be found in individual types of organisms. However, they indicate the order of magnitude of the relationships in marine organisms.
In order to extend the usefulness of these relationships, it is worth while to add the oxygen. As an approximation, it may be assumed that two atoms of oxygen are required for the oxidation of each atom of
In the euphotic layer, for each milligram of phosphorus utilized in photosynthesis, these ratios indicate that the plants will take up 7.2 mg of nitrogen (chiefly nitrate) and 76 ml of CO2 and release the same volume of oxygen. At lower levels, where regeneration is taking place, the consumption of 76 ml of O2 should set free the corresponding amounts of CO2, N, and P. The oxygen saturation value for water of 5° temperature is of the order of 7.0 ml/L; hence it may be seen that, if all of the oxygen has been consumed in subsurface water of approximately this temperature, the NO3-N and PO4-P will be increased by about 50 μg-atoms/L (0.650 mg/L) and 3 μg-atoms/L (0.090 mg/L), respectively. These values are approximately the largest amounts ever encountered in the ocean. If all the waters leaving the surface were saturated with oxygen and completely depleted of nitrate and phosphate, it might be expected that there would be a close agreement in the deeper waters between these substances and the oxygen depletion (difference between the saturation value and the observed content). Such a general relationship exists in waters that have left the surface in lower latitudes, but in higher latitudes water sinking from the surface is saturated with oxygen and contains appreciable amounts of nutrients; hence the relationship between the oxygen depletion and the nutrient content must have the form
where V is a variable amount in the water that sinks from the surface. In those regions in which there is a well-defined layer of minimum oxygen content at subsurface levels, layers of maximal nitrate and phosphate usually exist at or somewhat below the oxygen minimum.The above comments do not necessarily apply to elements composing hard “inorganic” skeletal structures. Both calcium carbonate and silica are utilized by organisms in the euphotic layer and elsewhere, but the ratios of utilization of Ca, C as CO3, and Si with reference to, say, phosphate-P, depend upon the character of the organisms. As pointed out elsewhere (p. 208), CaCO3 is removed from the surface layers, and the same is true of the SiO2. Although there is generally a depletion of Si in regions where the nitrate-N and phosphate-P are low, the processes of re-solution of calcareous and siliceous structures do not necessarily parallel decomposition and the regeneration of the elements found in the soft parts of the organisms. Therefore, the general distribution of silicon in the sea differs somewhat from that of phosphate and nitrate, and the ratios between Si and N and Si and P are variable.
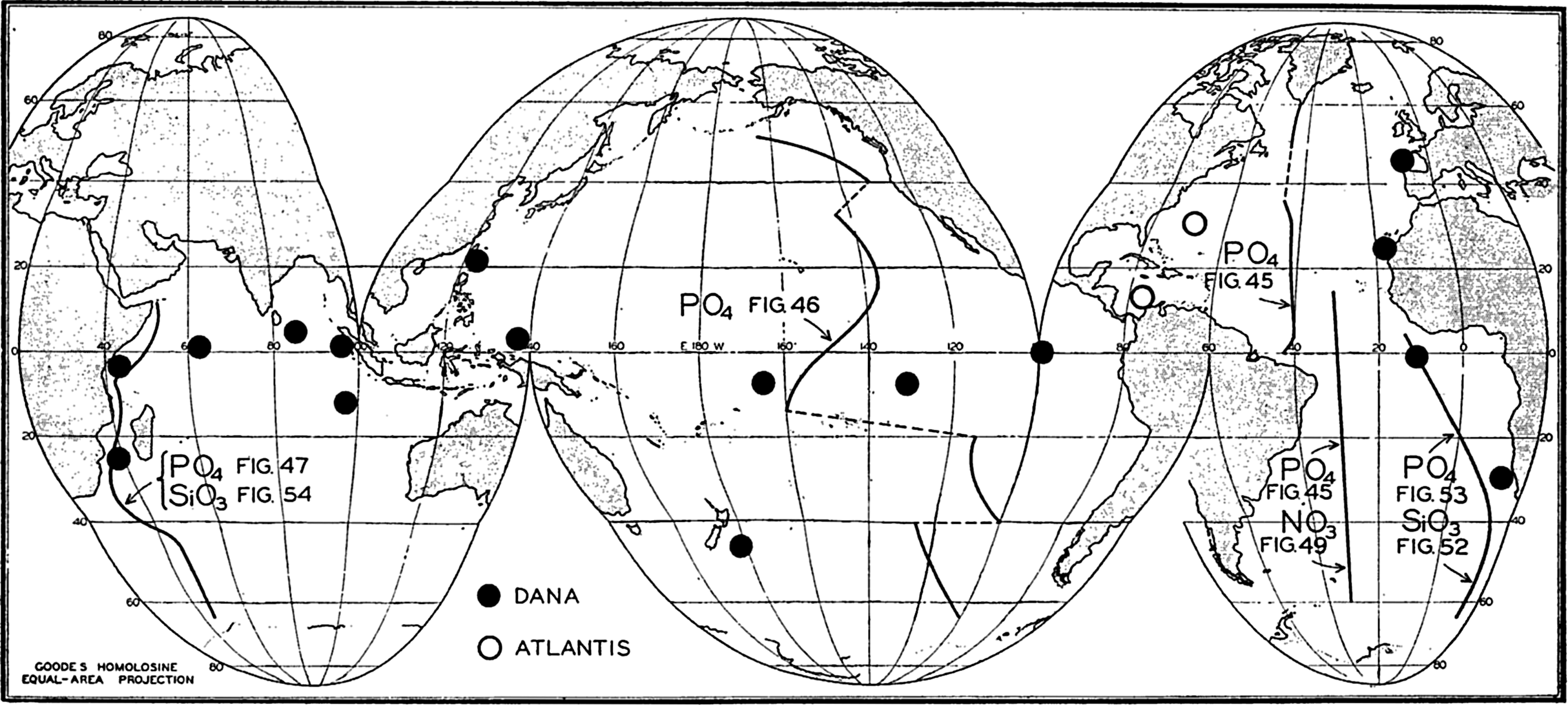
Locations of vertical sections and stations used to illustrate the distribution of phosphate, nitrate, and silicate, in the oceans.
[Full Size]
Distribution of Phosphate, Nitrogen Compounds, and Silicate in the Oceans
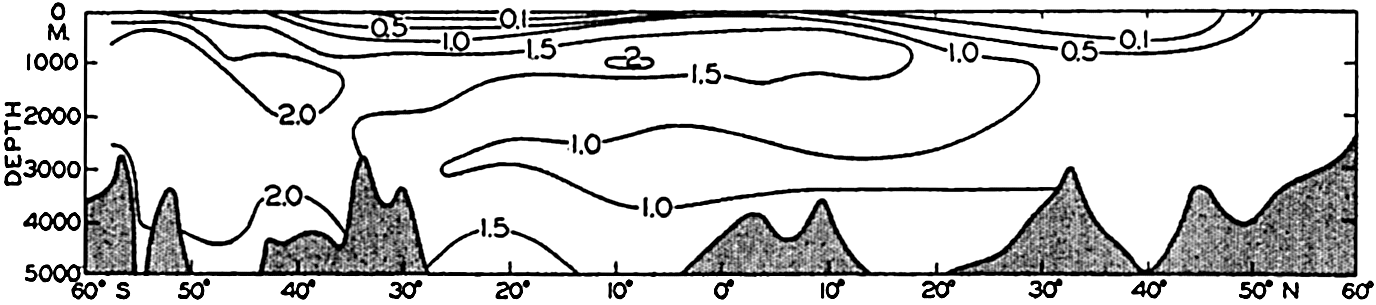
The distribution of phosphate in the three oceans may best be shown by means of longitudinal vertical sections whose locations are indicated in fig. 44. The representations are intended to bring out only the major features of the vertical distribution, and for this reason many of the minor irregularities have been omitted. The section in the Atlantic Ocean (fig. 45) is based on data obtained by the Discovery (Deacon, 1933) in the Southern Hemisphere, by the Atlantis (Seiwell, 1935) in the North Atlantic, and by the Meteor (Defant et al, 1936) in the area to the south of Greenland. The section in the Pacific Ocean (fig. 46) has been constructed from Discovery observations in the Antarctic (Clowes, 1938) and from those of the Carnegie (in press). The section in the Indian Ocean (fig. 47) is based on Discovery observations (Clowes, 1938). Examination of these sections and the vertical distribution curves in figs. 48 and 50 shows that in general the distribution of phosphate and nitrate is characterized by four different layers: (1) a surface layer in which the concentration is low and relatively uniform with depth, (2) a layer in which the concentration increases rather rapidly with depth, (3) a layer of maximum concentration that is usually located somewhere between 500 and 1500 m, and (4) a thick bottom layer in which there is relatively little change with depth. Examination of figs. 45, 46, and 47 shows that the surface layer is thickest in mid-latitudes
Phosphate distribution in a longitudinal section in the central Atlantic Ocean. Units: μg-atoms of phosphorus per 20° liter.
[Full Size]
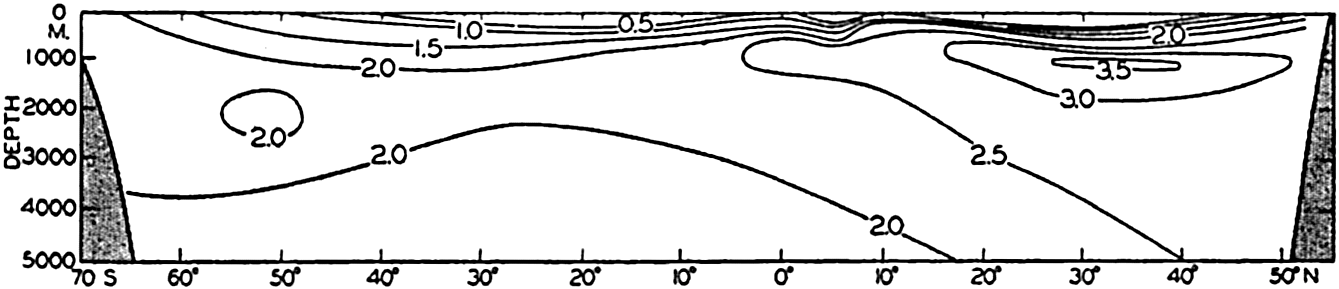
Phosphate distribution in a longitudinal section in the Pacific Ocean. Units: μg-atoms of phosphorus per 20° liter.
[Full Size]
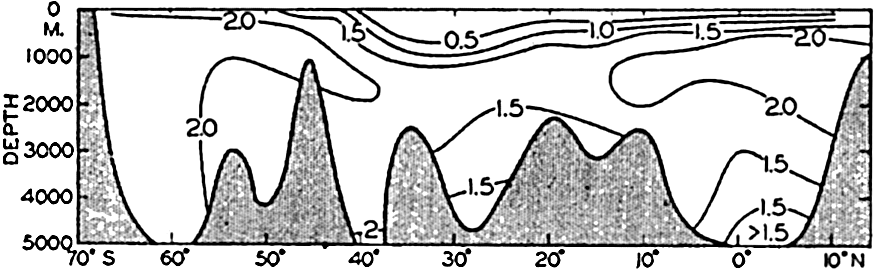
Phosphate distribution in a longitudinal section in the western Indian Ocean. Units: μg-atoms of phosphorus per 20° liter.
[Full Size]
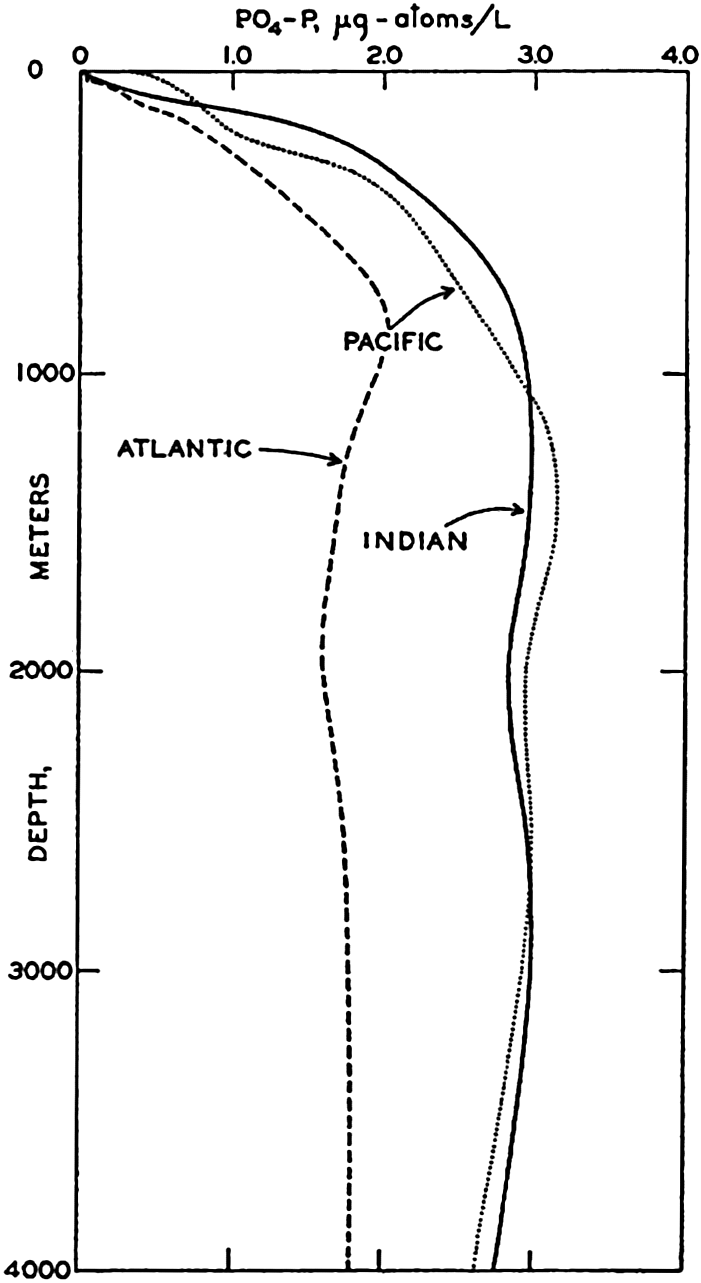
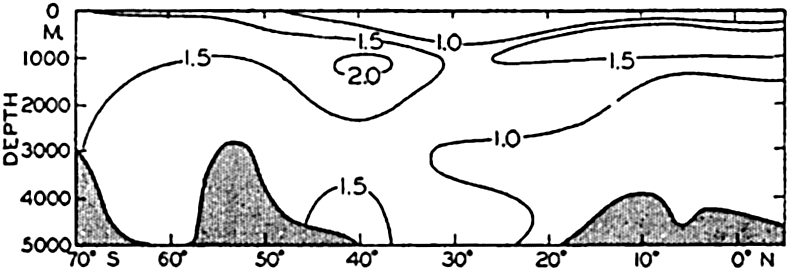
In the Atlantic Ocean the highest phosphate content (about 2 μg-atoms/L) is found in an intermediate layer extending northward from the Antarctic and centered at depths of about 1000 m. At all depths of about 1000 m or more there is a gradual decrease in phosphate concentration from south to north. A layer of minimum concentration containing less than 1.0 μg-atoms/L of phosphorus extends southward beneath the layer of maximum content. The phosphate concentration in the Antarctic is about twice that found in the northern part of the ocean. Regional differences in the phosphate distribution in the upper 50 m of the Atlantic Ocean are shown in fig. 217 (p. 787). Such differences are of importance in the distribution of plankton. The variations in a transverse section across the South Atlantic are illustrated in fig. 218 (p. 788), which shows the variable thickness of the surface layer of low phosphate content.
The distribution of phosphate in the Pacific Ocean (fig. 46) has many features that differ from those in the Atlantic Ocean. As might be expected, the conditions in the Antarctic are rather similar. However, the maximum amounts in the Pacific are found not in the Southern Hemisphere, as in the Atlantic, but north of the Equator, where the amounts present are about twice those found in the Antarctic (3.5 μg-atoms/L and about 2.0 μg-atoms/L, respectively). Furthermore, there is no clearly defined layer of minimum phosphate content beneath the maximum. The deeper waters of the Pacific are in general higher in phosphate than those of the Atlantic Ocean. The difference in the character of the distribution in the two oceans is related to the nature
The amounts of phosphate in the Indian Ocean (fig. 47) are greater than those in the Atlantic but somewhat less than those in the Pacific Ocean. The intermediate maximum in southern latitudes corresponds to that in the Atlantic, and the maximum in the equatorial region corresponds to that in the North Pacific, as it is related to the low oxygen content of the water. The minimum layer, at depths of about 3500 m, is clearly defined in all latitudes north of 40°S.
The differences between the phosphate concentrations in the three oceans are brought out in fig. 48, which is based on data collected by the Dana (Thomsen, 1931) on a voyage around the world and by the Atlantis in the western North Atlantic (Rakestraw and Smith, 1937). Observations from six stations (locations shown in fig. 44) in each ocean have been averaged and the values corrected for salt error (p. 182), so that they are somewhat higher than the values given in the sections. Too great emphasis should not be placed on the absolute values, as the purpose of the illustration is only to show the character of the vertical distribution in the three oceans, and to emphasize the higher phosphate content of the Pacific and the Indian Oceans in contrast to that of the Atlantic Ocean. Data from individual stations in moderate and low latitudes usually show a well-defined intermediate maximum.
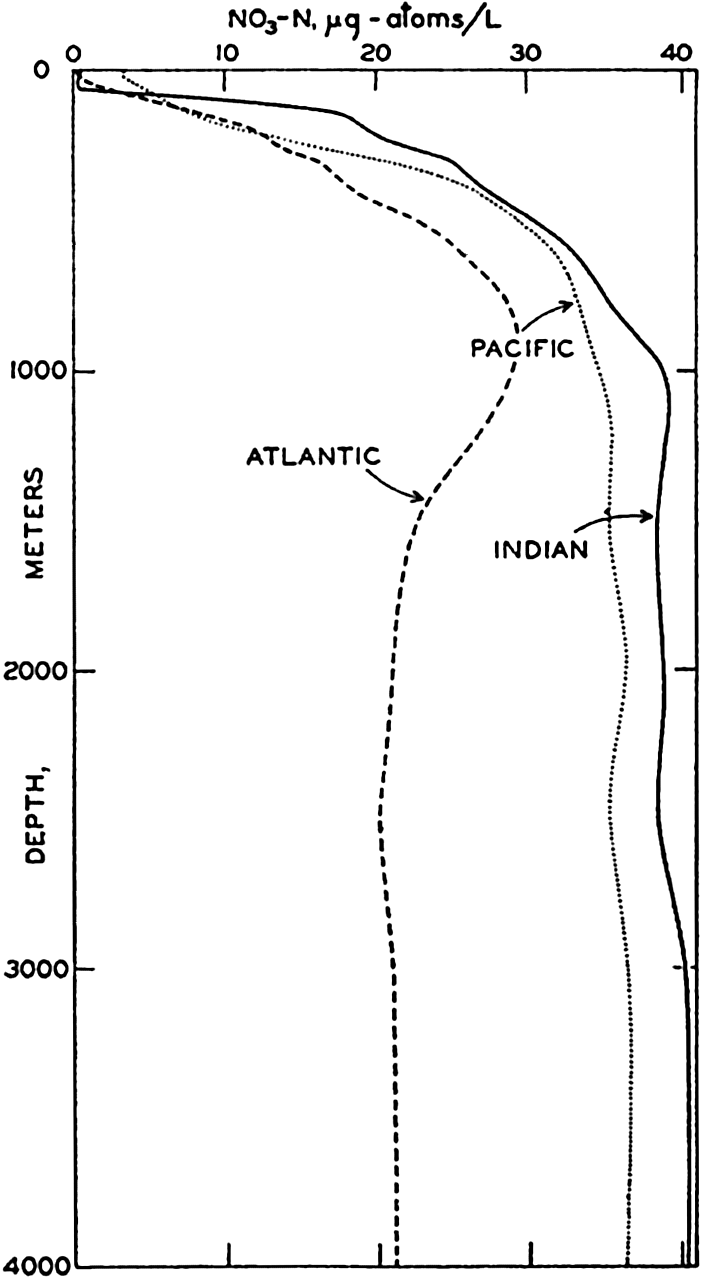
The lack of nitrate observations from many parts of the sea makes it impossible to prepare sections comparable to those for phosphate. Considerable data have been collected by the Dana (Thomsen, 1931, 1937), the Discovery (Discovery Reports, 1932; Deacon, 1933), the
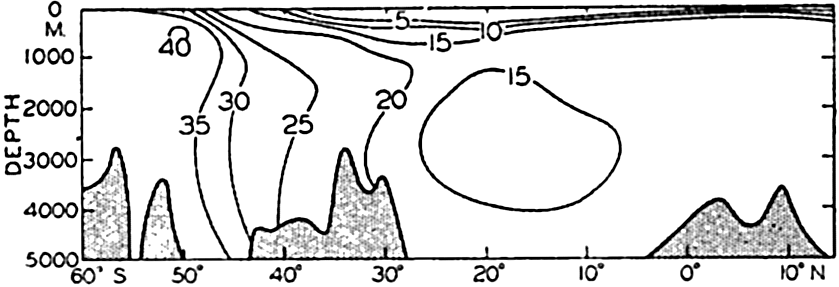
Distribution of nitrate in a longitudinal section in the central Atlantic Ocean. Units: μg-atoms of nitrogen per 20° liter.
[Full Size]
Curves for the nitrate distribution in the three oceans based on observations of the Dana and Atlantis and comparable to those shown for phosphate are given in fig. 50. Too much emphasis should not be placed upon the absolute values, but the curves clearly demonstrate the characteristic features of the vertical distribution
It has repeatedly been emphasized that there is a close parallelism between the concentrations of nitrate and phosphate. This relationship has been demonstrated by plotting against each other in fig. 51 the average data for phosphate and nitrate presented in figs. 48 and 50. It is immediately seen that there is a good linear relationship between the two substances. The straight line represents the “normal” ratio of nitrogen to phosphorus of 15:1 atoms proposed by Cooper (1938a). Because of this relationship, it is possible to predict with a fair degree of accuracy the concentration of either nitrate or phosphate when either one is known, and, as pointed out previously (p. 237), a relationship exists between the concentrations of these elements and the oxygen depletion.
The ranges in the various inorganic forms of nitrogen have been given (p. 181) as
The nitrate is the most abundant form of inorganic nitrogen, and, as shown in fig. 51, the low values occur at and near the surface, the high values in deeper water. The distribution of nitrite and ammonia, which are always in low concentrations, differs from that of nitrate in that the higher values occur in or above the thermocline. Nitrite may also be found near the bottom in shallow water, but it is generally absent from most of the water column. The ammonia content of the deeper waters is relatively uniform and low. Data given by Rakestraw (1936) and by Redfield and Keys (1938) indicate that in deep water, away from shore, the amounts of these substances are small. At one station, half way between Cape Cod and Bermuda, the nitrite was found only at about
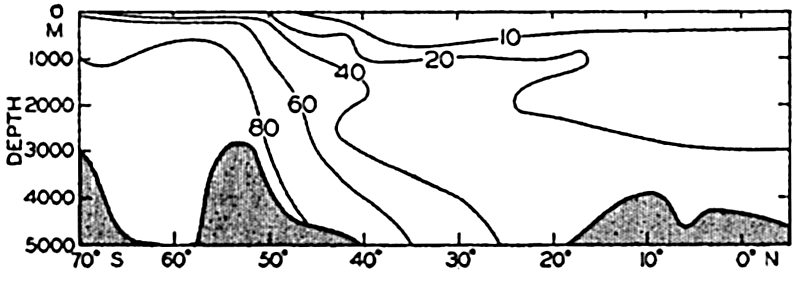
Distribution of silicate in a longitudinal section in the southeastern Atlantic Ocean. Units: μg-atoms of silicon per 20° liter.
[Full Size]
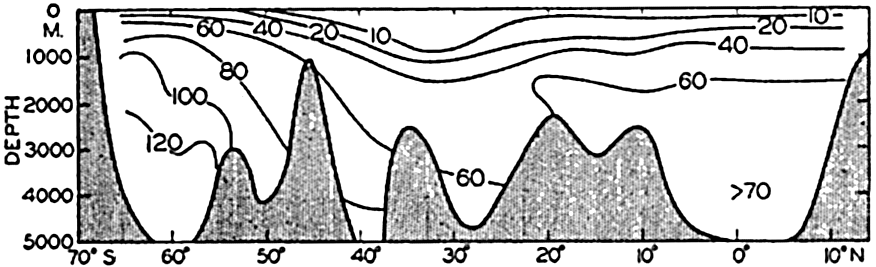
The data available on the distribution of silicate in the oceans are even fewer than those for nitrate. The Discovery (Clowes, 1938) has made numerous observations in southern latitudes, and certain of these data have been used in preparing longitudinal sections in the South Atlantic (fig. 52) and in the Indian Ocean (fig. 54). The locations of these sections are shown in fig. 44. The Carnegie obtained numerous observations in the northeastern and central Pacific and there are scattered observations from other regions, but they are inadequate for the preparation of longitudinal sections. In order to bring out similarities and differences in distribution, the silicate sections should be compared with the corresponding phosphate sections (figs. 47 and 53). It is readily seen that the vertical distribution of silicate differs from that of phosphate and nitrate, as there is no marked intermediate maximum and the concentration increases all the way down to the bottom. The reasons for this difference in the pattern of distribution have already been set forth (p. 237). In the Atlantic Ocean (fig. 52) the silicate content of the deeper water is much less in low latitudes than it is in the
Distribution of phosphate in a longitudinal section in the southeastern Atlantic Ocean. Units: μg-atoms per 20° liter.
[Full Size]
Distribution of silicate in a longitudinal section in the western Indian Ocean. Units: μg-atoms of silicon per 20° liter.
[Full Size]
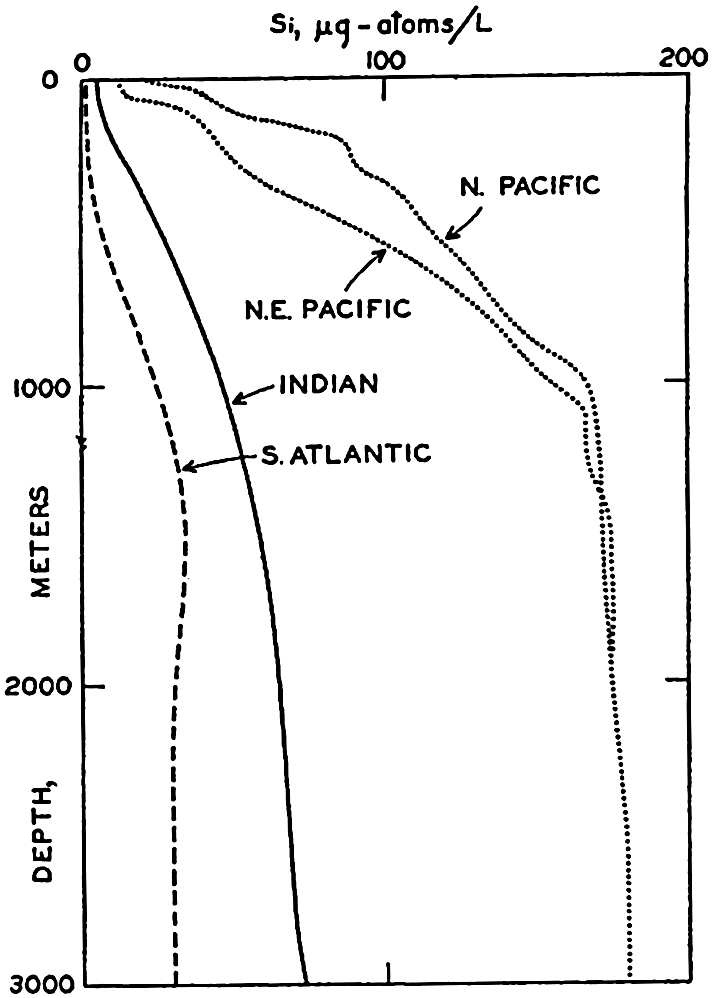
Vertical distribution of silicate at individual localities in the North Pacific, South Atlantic, and Indian Oceans.
[Full Size]
In basins, the distributions of the elements discussed above may be quite different from those characteristic of the open sea. As shown in chapter IV, the conditions in basins depend upon the topography, the character of the renewal processes below sill depth, and the dissolved oxygen content (aeration) of the water. In well-aerated basins, where there is inflow at the surface, the nutrient content is usually low. For example, in the Mediterranean Sea the phosphate and nitrate below sill depth are small when compared with the concentrations in the waters of the Atlantic Ocean (Thomsen, 1931). In the western Mediterranean below about 1000 m the contents of phosphate and nitrate are constant in amounts of 0.6 μg-atoms/L and 11 μg-atoms/L, respectively, which are about one half or less of the amounts in the open Atlantic. The dense water that flows out of the Mediterranean over the sill and mixes with the intermediate waters of the North Atlantic is therefore relatively low in nutrient elements and tends to reduce the phosphate and nitrate contents of the waters at intermediate levels in the eastern North Atlantic.
In basins of low oxygen content, such as the Red Sea, the nitrate and phosphate are relatively high for reasons already stated (p. 237). In stagnant Norwegian fjords, where hydrogen sulphide is present in the water, Ström (1936) found the phosphate to be as high as 10 μg-atoms/L.
Factors Influencing the Distribution of Nutrient Elements
The concept of dynamic equilibrium (p. 160) can be applied to the large-scale distributions of phosphate, nitrate, and silicate discussed above. On this assumption the distribution is stationary, the local change is zero, (∂s/∂t = 0), and there must be a balance between the effects of diffusion, advection, and the net effects of biological processes. Although the concept of stationary distribution is applicable to the deeper waters and probably to the upper layers in low latitudes, it is not valid for the upper several hundred meters in localities where seasonal variations are conspicuous.
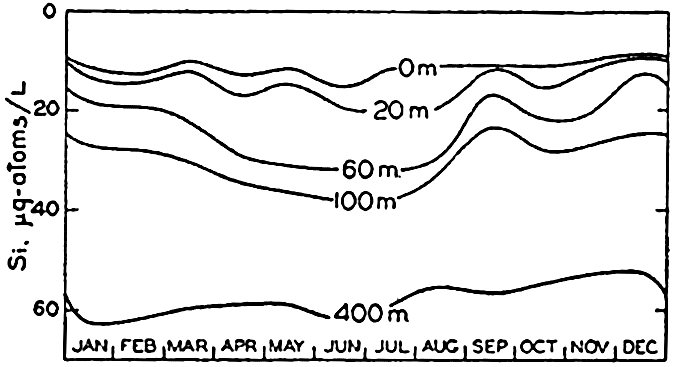
In the surface layers—that is, in the euphotic zone—biological processes will generally lead to a net utilization of the nutrient elements, and, if the rate of utilization exceeds the rate of supply by diffusion and by advection, the concentrations will decrease. This is the characteristic change that occurs during the spring and summer months in regions where physical conditions limit plant activity during the winter. In such regions the reverse change takes place during the winter, when the supply to the surface layers by diffusion and advection outweighs the utilization and leads to an increase in the nutrient content at and near the surface. Consequently in regions where the temperature, light intensity, and biological or other agencies are unfavorable for plant growth during part of the year, marked seasonal variations may occur in the nutrient distribution in the surface layers. In addition to fluctuations in consumption, the supply of nutrient elements to the surface layers by diffusion and advection may vary during the course of the year. For example, the magnitude of the vertical coefficient of eddy diffusivity, Az, is independent of the gradient in the nutrient concentrations but will be affected by the temperature distribution and the wind conditions. Furthermore, changes due to shifts in currents will affect the distributions, although undoubtedly the most conspicuous effects are those related to vertical advection. In regions of surface convergence, waters low in nutrients may extend for a considerable distance below the euphotic layer. On the other hand, where there is divergence—that is, upwelling—nutrientrich waters are carried upward toward the surface. Divergence may occur in the open ocean, as on the Equator and at the northern boundary of the Equatorial Countercurrent (p. 635), or along continental coasts where the prevailing winds are such that upwelling is induced (p. 501). In coastal areas where upwelling is seasonal or intermittent, the nutrient
Seasonal variations in the silicate content at various depths in Monterey Bay, California. Silicate scale increases downwards.
[Full Size]
From the foregoing comments, it is obvious that seasonal variations in the distribution of the biologically affected elements must be interpreted with great care. Fluctuations in concentration cannot be ascribed to biological processes alone unless it can be established on the basis of other data, such as temperature and salinity observations, that the effects of advection and diffusion can be neglected. In certain regions where there are marked differences between the summer and winter concentrations, estimates of organic production have been made from the depletion of nutrients occurring in the upper layers. Such estimates are minimal unless the effects of regeneration and diffusion are taken into account, and will be invalid if advection is an important factor, as in Monterey Bay. No systematic study of the seasonal variations in the distribution of the nutrients in the open ocean has yet been made, but in certain coastal areas a considerable amount of data has been accumulated that will be taken up under the discussion of the individual elements in the following pages.
Compounds of Carbon, Nitrogen, Phosphorus, and Silicon in the Sea
Organic Carbon. The carbon present as carbon dioxide and the salts of carbonic acid, as well as many of the effects of biological activity on the distribution of CO2, have been discussed in chapter VI. Seasonal changes in the CO2 content of the waters of the English Channel have been described by Cooper (1933). The carbon present in sea water in organic combination will now be considered.
The CO2 that is removed from the water by organisms is utilized partly for the secretion of calcareous structures but chiefly for building up organic compounds. Metabolic activity returns most of the organic carbon to solution as CO2, and bacteria and other microorganisms play an important part in the break-down of excretory products and the detrital material resulting from the death and partial disintegration of plants and animals. If dissolved oxygen is present in the water, the end products will be completely oxidized, but in the absence of oxygen anaerobic bacteria may flourish and hydrogen sulphide and other products of putrefactive decomposition may be formed. The latter conditions apparently occur only in and above the sediments in certain areas and in enclosed basins. The CO2 in calcareous structures returns to solution if the skeletal material dissolves. It should be remembered that, as with other elements, the cycle of carbon in the sea is not completely closed, since there is some loss to the sediments in both calcareous material and in resistant organic matter.
Before proceeding to a discussion of the amounts of carbon occurring in organic combination, either in living organisms or in particulate or dissolved compounds of organic origin, it should be pointed out that the division of the organic matter into various fractions is an empirical one. It has been customary to speak of “net plankton”—usually that which can be removed from the water by filtration through a fine net; “nannoplankton”—that which will pass through the ordinary net but which can be removed by centrifuging or passage through filter paper; and “dissolved organic matter”—that which will pass through the filter. Examination of the literature reveals that a variety of methods have been used to separate those fractions, and consequently the results for different fractions are not always comparable. In the following discussion the term “particulate material” will be used to designate all the material, either living or dead, which is caught by a fine filter that will retain particles of about the size of the larger bacteria. It should be kept in mind that the organisms in sea water, the number of bacteria, and even the inorganic constituents involved in bacterial development undergo rather rapid changes after the collection of samples. Therefore, unless the separation into the required fractions is made immediately, or unless suitable preservatives are added, the results obtained for the
Pütter (p. 912) maintained that marine invertebrates obtained nourishment from the dissolved organic matter in the water. This hypothesis was based on observations which indicated that the amount of organic matter in solution was many hundred times greater than that present as plankton and particulate detritus. Further investigation has tended to reduce the difference between the two fractions of organic matter, because the earlier determinations of dissolved organic material were obtained by inaccurate methods. Additional studies summarized by Krogh (1931) and Bond (1933) also indicate that dissolved organic matter cannot be utilized by animals. Although Pütter's hypothesis has lost its original significance, it has stimulated a great deal of interest in the problem of the dissolved organic material and its utilization, and investigations have shown that dissolved organic material, although unused by animals, can be utilized by bacteria (p. 912).
Although the problem has attracted much discussion and speculation, there are very few trustworthy data concerning the amounts of carbon present in particulate or dissolved material in the sea. It is extremely difficult to determine accurately the carbon in small amounts of organic matter, especially in the presence of large quantities of salts. Methods have been proposed for concentrating the particulate material by filtration or precipitation (von Brand, 1935) and for determining the carbon by a microcombustion method. No method is as yet available for concentrating the dissolved organic material and freeing it from the salt; consequently the existing methods are based on wet combustions with strong oxidizing agents such as permanganate or chromate. (Bond, 1933; Krogh and Keys, 1934.) Two difficulties are inherent in the latter type of determination: (1) many of the inorganic salts present in sea water interfere with the oxidation and usually tend to give high values, (2) there is uncertainty as to the completeness of the destruction of the organic compounds. Some organic materials may be completely converted into carbon dioxide, water, and so on, by such a procedure; other compounds are only partially decomposed, and still others are not attacked at all. As the chemical constitution of the dissolved material is not known, it is difficult to evaluate the accuracy of determinations made by such methods. Determinations made by wet combustion give the “oxygen consumed,” and a further uncertainty arises when it is necessary to convert these values into the amounts of organic carbon present in the samples.
The development of marine bacteriology has offered a new approach to the problem of the amount of organic material (both particulate and dissolved) in sea water. If sea water is placed in clean, stoppered bottles and kept in the dark, bacteria will develop in great numbers
The amount of carbon present in oceanic sea water in inorganic compounds is between 2.1 and 2.5 mg-atoms per liter (25 to 30 mg/L), depending upon the salinity, temperature, and effects of biological activity. Krogh (1931, 1934a,b) has summarized the available data on the amount of organic carbon in sea water. In his later work he reports total organic carbon analyses on six water samples from the Atlantic Ocean. Virtually no variation with depth was found, and Krogh considers the average value applicable to all depths and oceans. The average was 0.2 mg-atoms (2.05 mg) of carbon per liter, which is approximately one tenth of the amount present in inorganic form. From estimates of the amount of plankton, Krogh found the dissolved material to be about three hundred times more abundant than the particulate organic matter. These figures apply to the deeper water of the open ocean. Bond (1933) examined the surface layers in nearshore areas of higher production and found rather different values. His original data, obtained by wet combustion, are expressed in terms of oxygen consumed. In order to make them comparable to those of Krogh, it has been assumed that two atoms of oxygen were required to oxidize one atom of carbon. The recomputed minimum, maximum, and average values are given in table 53. Although Bond's values for the dissolved fraction are approximately the same as those of Krogh for the total carbon, they show a considerable range. Furthermore, it will be noted that the particulate material is relatively more abundant and forms between one tenth and one third of the total.
The results of bacteria1 oxygen-consumption studies are difficult to evaluate because of the variety of techniques that have been used.
| Substance | mg-atoms/L of carbon | ||
|---|---|---|---|
| Minimum | Maximum | Average | |
| Net plankton | 0.008 | 0.06 | 0.03 |
| Nannoplankton | 0.005 | 0.11 | 0.03 |
| Dissolved | 0.13 | 0.25 | 0.20 |
| Total organic | 0.143 | 0.42 | 0.26 |
The oxygen consumptions given above were for water from near the surface in areas relatively rich in plankton. Samples from deeper levels consume about one half as much oxygen. As shown previously (p. 236), there are relatively constant ratios between carbon, nitrogen, and phosphorus in the organic material. This fact has been used in certain studies in which the organic nitrogen (determined by the Kjeldahl method) is used as a measure of the amount of organic matter. The relative amounts of organic nitrogen and phosphorus in sea water are in fair agreement with the amounts of carbon given above. The organic nitrogen content of bottom samples (p. 1010) has been widely used as a measure of their content of organic matter. In sediments the ratio of
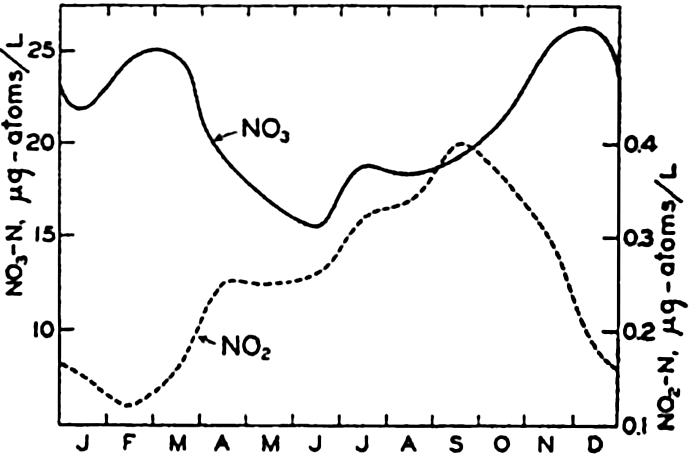
Annual cycle in the nitrate and nitrite content of the surface waters at Friday Harbor, Washington.
[Full Size]
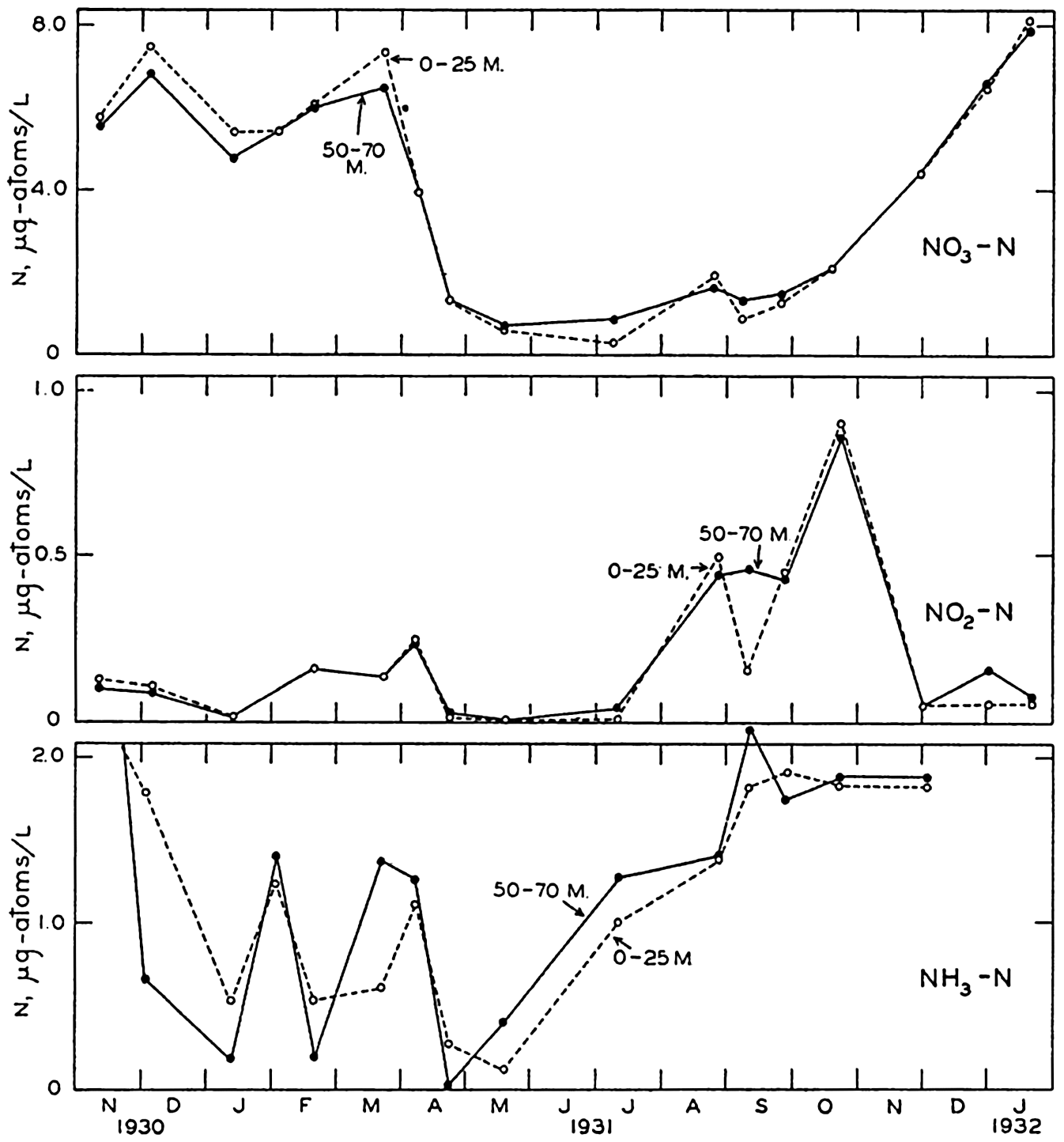
Seasonal variations in nitrate, nitrite, and ammonia in the surface layer (0–25 m) and in the bottom layer (50–70 m) in the English Channel during the period November, 1930, to January, 1932. (After Cooper, 1937b.)
[Full Size]
Nitrogen Compounds and Their Seasonal Variation. In certain coastal areas, sufficient data are available to examine the seasonal changes in the distribution of nitrate, nitrite, and ammonia. Only selected cases of seasonal variations will be given, but additional references may be found in the works cited. Phifer and Thompson (1937) give the results of nearly five years' studies of the surface conditions at Friday Harbor, on the San Juan Channel. The averages of the monthly means for NO3 and NO2 for the period 1931 to 1935 are shown in fig. 57. It should be noted
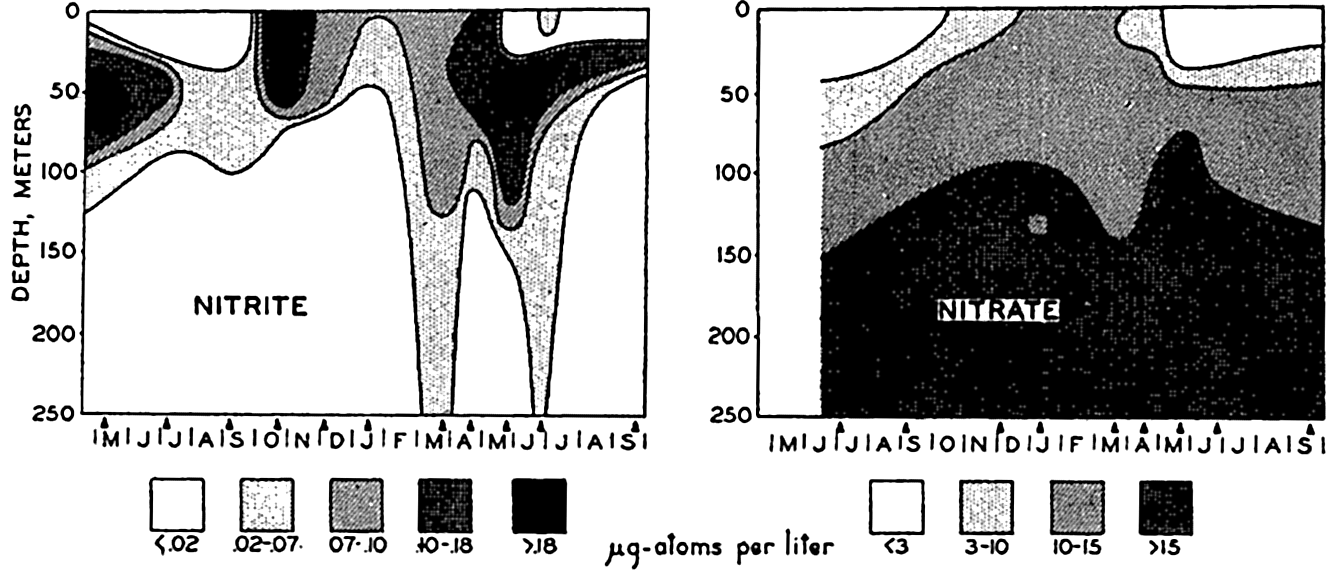
Seasonal variations in the vertical distribution of nitrite and nitrate in the Gulf of Maine during the period May, 1933, to September, 1934. (After Rakestraw, 1936b.)
[Full Size]
The nitrogen in particulate organic material may be determined on the separated material which has been concentrated by filtration or
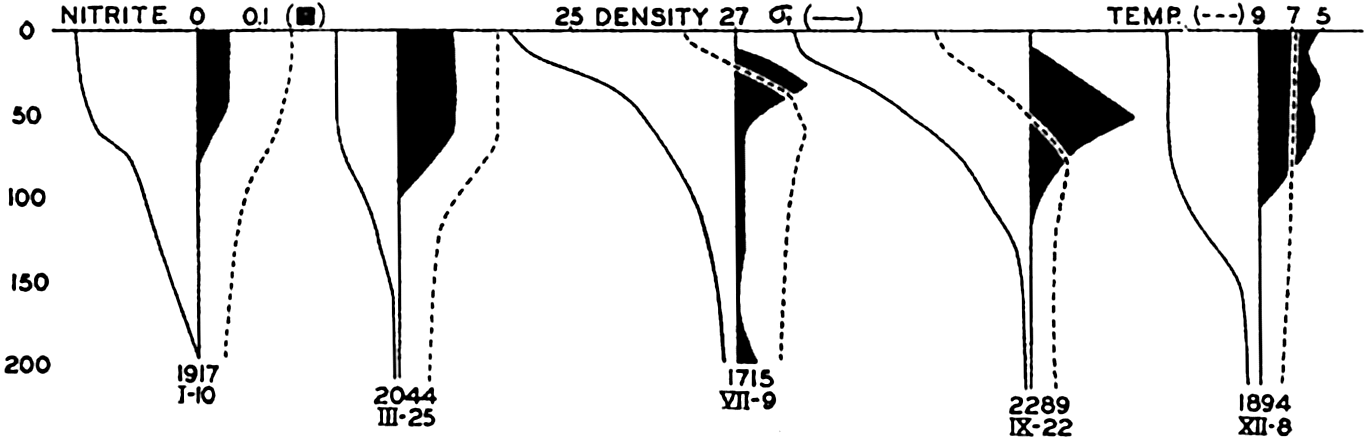
Vertical distribution of nitrite, as related to density (σt) and temperature. (After Rakestraw, 1936.)
[Full Size]
The total organic nitrogen, including both particulate and dissolved material, has been investigated by Robinson and Wirth (1934a,b). Kjeldahl analyses on unfiltered oceanic sea water showed about 7.2 μg-atoms/L of organic nitrogen near the surface, about half this amount at intermediate depths, and a slight increase again toward the bottom. In nearshore water the values near the surface were about twice as high as those in the oceanic samples. Moberg and Fleming (1934), using a similar method, found about 10 μg-atoms/L, on an average, of organic nitrogen in the surface layers off southern California and somewhat higher values at greater depths.
Figures 57 and 58 show that at Friday Harbor about 10 μg-atoms/L of NO3-N disappear during the summer, and that the change in the English Channel is approximately the same. As NH3 and NO2 are rarely present in comparable amounts, we must conclude that the nitrogen is in organisms, organic debris, dissolved organic compounds, or in some unrecognized inorganic form.
Interesting experiments by von Brand, Rakestraw, and Renn (1937, 1939) on the regeneration of nitrate from marine plankton in vitro indicate that the formation of ammonia from organic matter probably takes place without the formation of intermediate compounds. The results
These experiments are extremely interesting, although regenerative processes in vitro apparently differ considerably from those in the sea. This result might well be expected from the peculiar laboratory conditions and the fact that the water was enriched with organic material. It may also account for the fact that the NH3 and NO2 reached relatively high values (the same as the NO3), and that during the regeneration the stages of production of NH3, NO2, and NO3 were clearly defined (cf. figs. 57 and 58). Furthermore, it is interesting to note the great increase in the amount of nitrogen present as particulate material that was apparently resistant to the action of the bacteria present. At the end of the experiment, approximately 50 per cent of the nitrogen was in this form. Such a “waste” of nitrogen does not take place in the sea. It is also of interest to note that diatoms would flourish if the medium were placed in the light when either NH3 or NO2 were abundant and before the NO3 had been produced. This supports the theory that marine plants can use any of these inorganic forms bf nitrogen equally well.
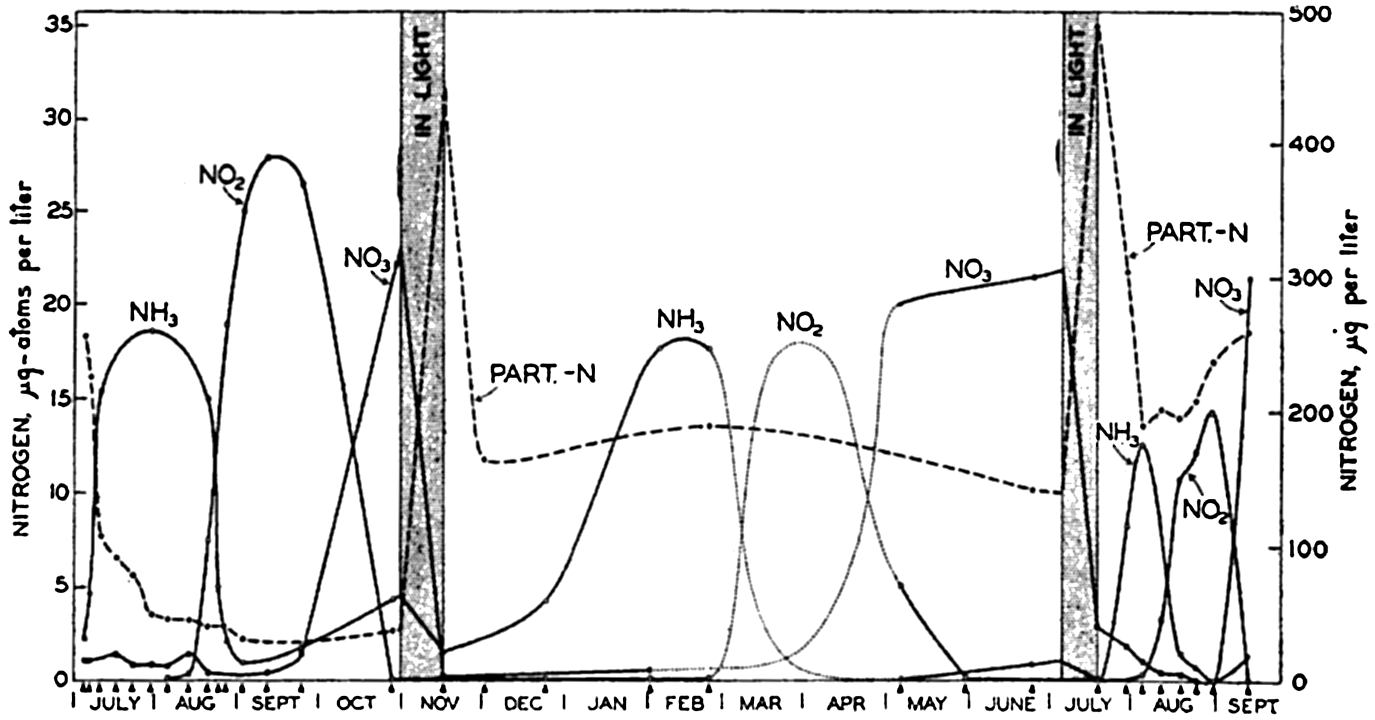
Experiment on the utilization and regeneration of nitrate. When medium was placed in light, it was innoculated with diatoms. Data from von Brand, Rakestraw, and Renn, 1939.
[Full Size]
Nitrogen Cycle in the Sea. The chemically bound nitrogen in sea water is known to occur in living organisms, in dissolved and particulate material of organic origin, and as ammonia, nitrite, and nitrate.
Bacteria play important roles in the mineralization of organic nitrogen by acting upon detrital material, excreta, and dissolved organic matter, and in the intertransformation of NH3, NO2, and NO3 (chapter XVIII). Pure culture studies with different species of marine bacteria show that a variety of transformations can be made under laboratory conditions, but these observations must be applied to the sea with caution because the organisms may not be capable of carrying on similar processes in the natural environment. The very large literature bearing on the nitrogen cycle in the sea has been reviewed by Cooper (1937b), who has considered all the various modes of transformation that are possible and from them has selected the more probable ones. In general, it is believed that the nitrogenous material gives rise to ammonia, which in turn is converted to nitrite and then to nitrate. The ammonia may be formed by the hydrolysis of protein material, amino acids, amines, and purine compounds such as urea, or through bacterial action on them.
The oxidation of ammonia to nitrite releases a large amount of energy and hence needs only to be activated in some way. The following agencies have been suggested:
Photochemical oxidation induced by direct sunlight. This reaction was first observed in sea water by ZoBell (1933), but, as pointed out by Cooper, can be effective only within the upper meter or so of water, owing to the rapid absorption of the shorter wave lengths that activate the reaction.
Chemical oxidation by the free oxygen in the water in the presence of surface catalysts. This reaction is of unknown significance.
Bacterial oxidation. Nitrifying bacteria are present in bottom sediments, and forms isolated by Zobell (1935b) converted ammonia to nitrite. However, the conversion occurred at a much higher oxidation-reduction potential than is ordinarily found in the sediments. Studies of the decomposition of marine plankton in vitro show a conversion of ammonia to nitrite, but no nitrifying bacteria could be isolated (von Brand, Rakestraw, and Renn, 1937). Nonetheless, the fact that nitrifying bacteria cannot be readily detected in sea water is not definite proof of their absence. It is well known that many marine bacteria are difficult to culture, and development of new techniques may establish their presence. Carey (1938) has shown that they can be isolated from
― 257 ―water rich in plankton. The presence of nitrite, which can sometimes be detected near the sea bottom, as in the English Channel in certain seasons, may indicate either oxidation of ammonia or reduction of nitrate. Although it has not been established, bacteria are probably the most important agency in the oxidation of ammonia to nitrite.
Oxidation of nitrite to nitrate also releases energy, and, as in the oxidation of ammonia, purely chemical or photochemical processes may be important. Bacteria capable of making the transformation are abundant in sediments, but they are difficult to isolate from the water column. Development of suitable techniques may also clarify this problem. Cooper has pointed out that in sea water saturated with oxygen the nitrate in equilibrium with nitrite will be of a tremendously greater order of magnitude. Hence, the detection of nitrite in the water column may indicate active production of this substance, which is present only as a transitional stage in the regeneration of nitrate. Brandt's hypothesis (p. 768) was based on the discovery of marine bacteria which under laboratory conditions were capable of denitrification. However, it is now considered that under the conditions prevailing in the sea there is little or no loss of fixed nitrogen, although it has been shown that reduction of NO3 to NO2 may occur. This reduction may also be carried out by diatoms, as has been observed in pure-culture experiments (ZoBell, 1935a).
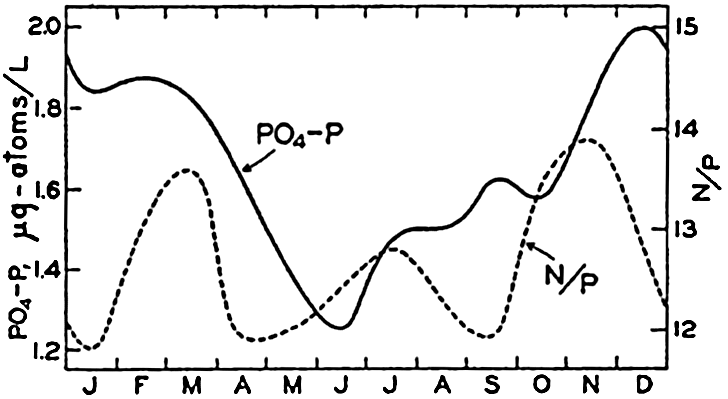
Annual cycle in the phosphate content of the surface waters at Friday Harbor, Washington, and the monthly N/P ratios.
[Full Size]
Organic Phosphorus and Seasonal Variations in Phosphate. For certain coastal areas, sufficient data exist to show the nature of the seasonal variations in phosphate. Friday Harbor, the English Channel, and the Gulf of Maine have been selected as examples.
In fig. 62 are presented the averages based on about four years' observations of the monthly mean values for PO4-P at Friday Harbor (Phifer and Thompson, 1937). Highest values occur during the winter, and lowest values during the summer season, when phytoplankton growth has been great. The monthly ratios of N:P are somewhat lower than the normal ratio proposed by Cooper, and their variability during the course of the year indicates that proportionally more nitrate than phosphate is utilized. Neither of these substances can be considered as limiting the amount of phytoplankton produced at this locality. Phosphate data from the English Channel (Cooper, 1938b) for the period November, 1930, to January, 1932, are presented in fig. 63 as
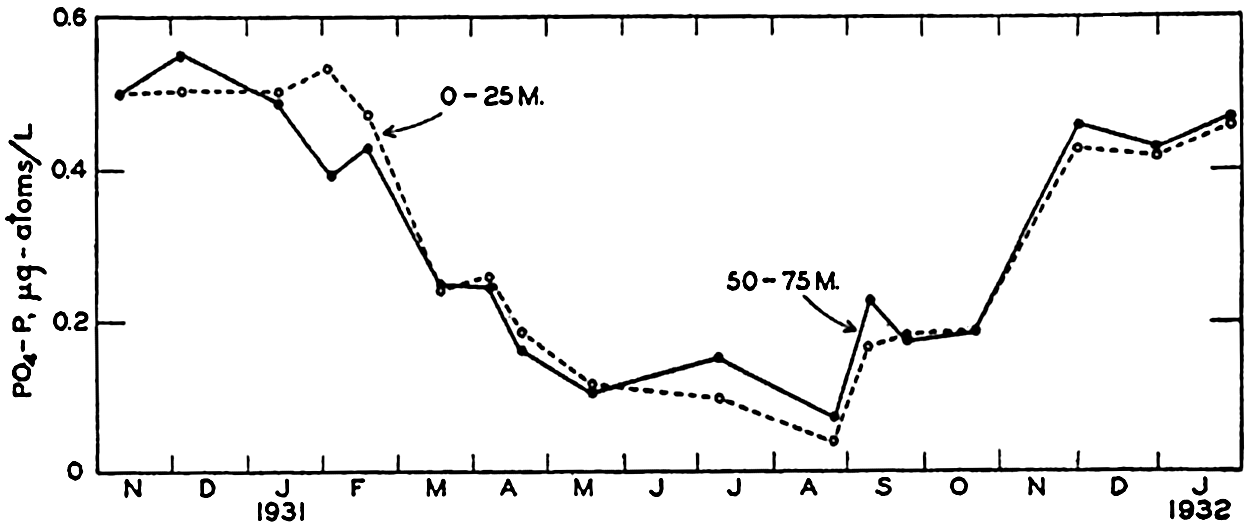
Seasonal variations in the phosphate content of the surface layer (0–25 m) and the bottom layer (50–75 m) in the English Channel during the period November, 1930, to January, 1932.
[Full Size]
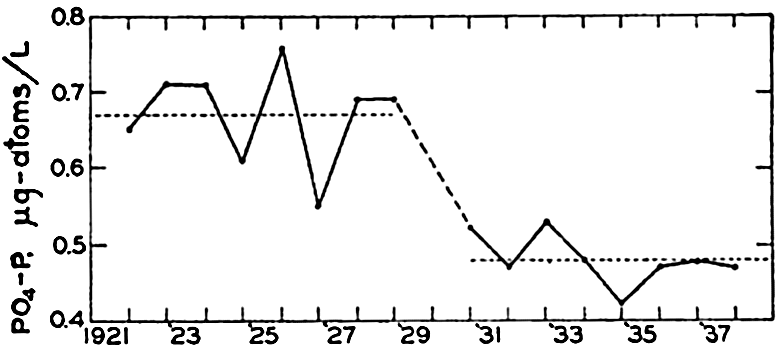
Cooper (1938b) has assembled phosphate data for the English Channel covering a period of eighteen years. In fig. 64 are entered the winter maxima for the average PO4-P content of the water column near Plymouth. As the phosphate content of the water during the winter is a measure of potential production for the following spring and summer, his data indicate a drop in fertility after 1929 or 1930. The changes are considered to be associated with the circluation, which may undergo random or periodic fluctuations.
In the preceding discussion, emphasis has been placed on the cyclic nature of the seasonal variations, and it is obvious that the conditions in any area may not repeat themselves if disturbances, such as shifts in the circulation, bring about changes.
The seasonal variations in the PO4-P and in the various organic phosphorus fractions in the Gulf of Maine have been studied by Redfield, Smith, and Ketchum (1937). During one year, five series of samples were collected at various depths between the surface and the bottom. These
| Form of phosphorus | Interval of depth, meters | μg-atoms/L of phosphorus | ||||
|---|---|---|---|---|---|---|
| May 18, 1935 | Aug. 20, 1935 | Nov. 8, 1935 | Feb. 26, 1936 | May 14, 1935 | ||
| Phosphate | 0–60 | 0.60 | 0.68 | 0.65 | 1.03 | 0.64 |
| 60–120 | 1.11 | 0.91 | 1.08 | 1.02 | 1.25 | |
| 120–180 | 1.31 | 1.22 | 1.25 | 1.11 | 1.51 | |
| 180–240 | 1.61 | 1.39 | 1.22 | 1.51 | 1.60 | |
| Dissolved organic | 0–60 | 0.08 | 0.34 | 0.29 | 0.07 | 0.14 |
| 60–120 | 0.02 | 0.29 | 0.31 | 0.14 | 0.36 | |
| 120–180 | 0.01 | 0.17 | 0.29 | 0.17 | 0.15 | |
| 180–240 | 0.00 | 0.20 | 0.37 | 0.03 | 0.10 | |
| Particulate organic | 0–60 | 0.15 | 0.10 | 0.10 | 0.05 | 0.12 |
| 60–120 | 0.06 | 0.05 | 0.05 | 0.05 | 0.07 | |
| 120–180 | 0.04 | 0.03 | 0.08 | 0.03 | 0.02 | |
| 180–240 | 0.04 | 0.03 | 0.08 | 0.04 | 0.06 | |
| Total (Average for whole water column) | 0–240 | 1.26 | 1.36 | 1.44 | 1.31 | 1.51 |
Earlier work on organic phosphorus in the sea has been summarized by Cooper (1937a), who also reports observations from the English Channel. Cooper points out that many determinations of the total “organic phosphorus” (both particulate and dissolved) probably include arsenite-arsenic that has been oxidized to arsenate when the organic matter was destroyed and that has not been reduced again. Arsenate will give the same reaction as phosphate in the colorimetric estimation, and, because the arsenic present as arsenite will not affect the inorganic phosphate analyses, the determination of “organic phosphorus” will be too high by about 0.2 μg-atoms/L (the concentration of arsenic). In the determinations in the Gulf of Maine this source of error was
The maximum values reported by Redfield et al in the Gulf of Maine for particulate and dissolved organic phosphorus are
If multiplied by 16, the ratio of N:P in organisms, the corresponding values of particulate and dissolved organic nitrogen would be 3.4 and 9.3 μg-atoms/L respectively, which agree with determinations of nitrogen in the corresponding fractions (p. 254).Phosphorus Cycle in the Sea. The cycle of phosphorus in the sea is rather similar to that of nitrogen except that only one inorganic form, phosphate, is known to occur. As shown above, phosphorus can be found in organisms, in particulate and dissolved organic compounds, and as phosphate. Probably only the phosphate is utilized by plants, and the dissolved organic fraction, which can originate as a metabolic product and from excreta, and the decomposition of organic material must be intermediate stages in the regeneration of phosphate. The roles that bacteria play are not yet known.
Studies of the decomposition of plankton material in vitro have led to some interesting results. Cooper (1935) added zooplankton and phytoplankton material of known phosphorus content to sea water and determined the rate at which the PO4-P was formed. The PO4 appeared more rapidly in the zooplankton samples than in the diatom material. Furthermore, the PO4 produced in the zooplankton samples was in excess of that originally present as PO4 plus that added in the particulate material. This excess was formed from dissolved organic phosphorus originally present in the water. The PO4 in the diatom experiments never rose to the level of the original PO4 plus that added, even after an interval of about five months. The initial rate of appearance of PO4 was rapid, and in the zooplankton experiments the transformation was nearly complete in about two weeks. The difference in behavior of the plant and animal material cannot yet be accounted for. Seiwell and Seiwell (1938) found that zooplankton decomposition at 22° to 25°C (probably higher than the temperature of Cooper's experiments) was such that the formation of PO4 was most rapid during the first day or two after death. This stage was often followed by a period when the rate of utilization of PO4 by microorganisms in the experiments exceeded that of formation. On the basis of these experiments it is sometimes considered that the PO4 must be regenerated more rapidly than the NO3. However, field evidence offers little support for this theory. Examination
Redfield, Smith, and Ketchum (1937), on the basis of the material summarized in table 54, have calculated the manner in which the various phosphorus compounds vary in the different layers, assuming (1) that all utilization of phosphate takes place in the upper 60 m, (2) that all downward transport of phosphorus is due to the settling of organisms and particulate material, and (3) that the upward transport is all due to eddy diffusion. From their examination it was concluded that decomposition and regeneration took place throughout the column of about 240 m of water.
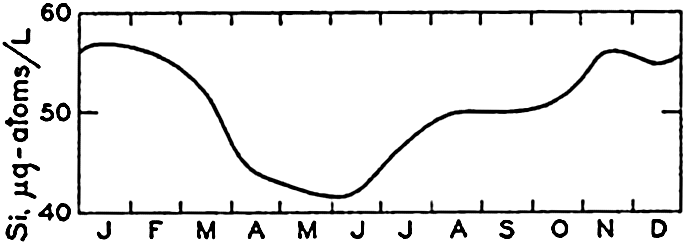
Seasonal Variation in Silicate. Observations on the seasonal cycle in silicate-silicon have been carried out in many localities, of which Friday Harbor and the English Channel have been selected as examples. The data presented are comparable to those given for NO3-N and PO4-P. Fig. 65 shows the average monthly values for Friday Harbor based on more than four years' observations. The highest values occur during the winter and the lowest values during the early summer. The range in silicon is 15 μg-atoms/L. The ranges in SiO3-Si, NO3-N, and PO4-P are in the proportions of 20:14.7:1. Data for the English Channel (Cooper, 1933) are shown in fig. 66. It will be noted that the concentration of silicon in the English Channel is much lower than it is at Friday Harbor, being in general only about 1/25 as great. The range in silicon
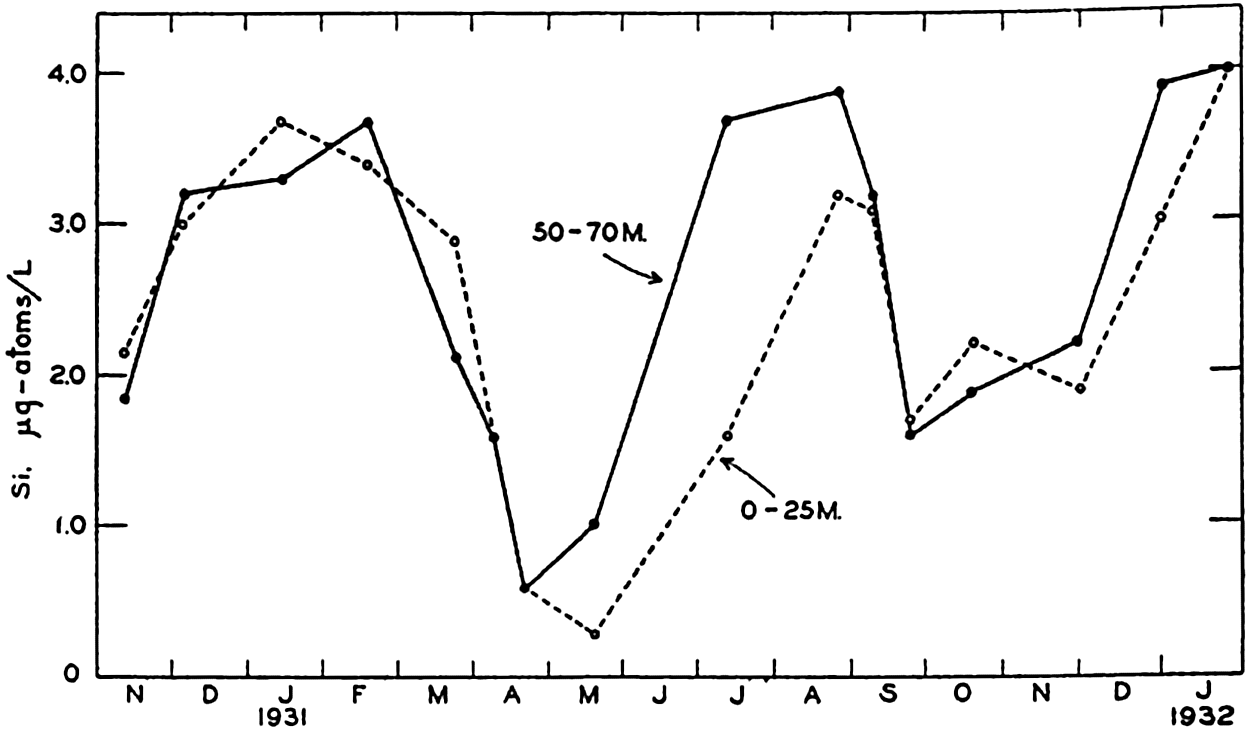
Seasonal variations in the silicate content of the surface layer (0–25 m) and the bottom layer (50–70 m) in the English Channel during the period November, 1930 to January, 1932.
[Full Size]
A factor that may complicate the seasonal changes in the silicon concentration in nearshore areas is the amount of siliceous material carried in by river waters, which is generally several times greater than that found in sea water; dilution will therefore tend to raise the concentration of silicate (Hutchinson, 1928).
The Silicon Cycle in the Sea. The depletion of the silicon in the surface layers is the result of biological activity and the sinking of the organisms or of their skeletal remains. The silicon removed from the water by diatoms or other organisms may return to solution after the death of the organism or may be deposited on the sea bottom. Unlike their roles in the cycles of nitrogen and phosphorus, bacteria are probably not directly involved in the re-solution of silicon from skeletal material. The skeletal material that sinks to the bottom forms either a temporary or permanent constituent of the sediments. Siliceous sediments are found in higher latitudes where the bottom material contains a very large proportion of diatom frustules, while in other regions radiolarian skeletons make up a large part of the sediment (chapter XX).
In the English Channel the silicon distribution is more erratic than that of PO4 and of NO3, and there may be large differences in the amounts at various levels. High values often occur near the surface and immediately over the bottom. The high surface values are ascribed to the effects of river water, the high bottom values to the re-solution of skeletal material that has settled there. The accumulation of dissolved silicates at a marked thermocline is also evidence of re-solution of slowly settling debris.
Estimates of plankton production in the English Channel based on the difference between the winter maxima and summer minima in PO4-P, NO3-N, and SiO3-Si (Cooper 1933, 1938b) yield values from the SiO3 data which are approximately 1/15 of those obtained from the other elements. Cooper has attributed the smaller value to the rapidity with which the silicon passes through its cycle. The difference in the amount of silicon utilized in the English Channel and at Friday Harbor was noted above, and the fact that the concentration is reduced in the English Channel to a very low level may indicate that in that locality it does influence the production of diatoms.
King and Davidson (1933) found that the quantity of SiO3-Si in solution affected the growth of diatoms in laboratory cultures. They also followed the changes in dissolved silicon after the death of the diatoms and found that complete solution took place in about five months. Marine phytoplankton were similarly studied, and it was found that samples which were boiled dissolved more slowly than those unboiled. The authors suggest the possible existence of an enzyme that hastens solution.
Bibliography
Barnes, C. A., and T. G. Thompson. 1938. Physical and chemical investigations in Bering Sea and portions of the North Pacific Ocean. Univ. of Washington, Pub. in Oceanogr., v. 3, no. 2, 79 + App. 163 pp., 1938. Seattle. 
Bond, R. M.1933.
“A contribution to the study of the natural food-cycle in aquatic environments”
. Bingham Oceanogr. Coll., Peabody Museum, Bull., v. 4, art. 4, 89 pp., 1933. 
Carey, Cornelia L.1938.
“The occurrence and distribution of nitrifying bacteria in the sea”
. Jour. Marine Research, v. 1, p. 291–304, 1938. 
Carnegie Results. Physical and chemical data and results of dynamic computations for Carnegie deep-sea stations, 1928–1929, Tabulated data. Results of Oceanographic and Meteorological Work on Board the Carnegie on Cruise VII, 1928–1929. (In press.) Carnegie Institution of Washington. 
Clarke, F. W., and W. C. Wheeler. 1922.
“The inorganic constituents of marine invertebrates”
. U. S. Geol. Surv., Prof. Paper, no. 124, p. 1–62, 1922. 
Clements, F. W., and R. C. Hutchinson. 1939.
“The ash constituents of Australian fish”
. Australian Jour. Exper. Biol. and Med. Sci., v. 17, p. 89–92, 1939. 
Clowes. A. J.1938.
“Phosphate and silicate in the southern oceans”
. Discovery Repts., v. 19, p. 1–120, 1938. 
Cooper, L. H. N.1933.
“Chemical constituents of biological importance in the English Channel, November, 1930, to January, 1932”
. Marine Biol. Assn. U. K., Jour., v. 18, p. 677–753, 1933. Plymouth. 
Cooper, L. H. N.1934.
“The determination of phosphorus and nitrogen in plankton”
. Marine Biol. Assn. U. K., Jour., v. 19, p. 755–59, 1934. Plymouth. 
Cooper, L. H. N.1935.
“The rate of liberation of phosphate in sea water by the breakdown of plankton organisms”
. Marine Biol. Assn. U. K., Jour., v. 20, p. 197–200, 1935. Plymouth. 
Cooper, L. H. N.1937a.
““Organic” phosphorus in sea water from the English Channel”
. Marine Biol. Assn. U. K., Jour., v. 21, p. 673–78, 1937. Plymouth. 
Cooper, L. H. N.1937b.
“The nitrogen cycle in the sea”
. Marine Biol. Assn. U. K., Jour., v. 22, p. 183–204, 1937. Plymouth. 
Cooper, L. H. N.1938a.
“Redefinition of the anomaly of the nitrate-phosphate ratio”
. Marine Biol. Assn. U. K., Jour., v. 23, p. 179, 1938. Plymouth. 
Cooper, L. H. N.1938b.
“Phosphate in the English Channel, 1933–38, with a comparison with earlier years, 1916 and 1923–32”
. Marine Biol. Assn. U. K. Jour., v. 23, p. 181–95, 1938. Plymouth. 
Deacon, G. E. R.1933.
“A general account of the hydrology of the South Atlantic Ocean”
. Discovery Repts., v. 7, p. 171–237, 1933. 
Defant, A., G. Böhnecke, H. Wattenberg. 1936.
“Die ozeanographischen Arbeiten des Vermessungsschiffes Meteor in der Dänemarkstrasse und Irmingersee, Berlin Universität, Institut f. Meereskunde, Veröff., N.F., A. Geogr.-naturwiss”
. Reihe, Heft 32, 152 pp., 1936. 
Discovery Reports. 1932.
“Station list, 1929–1931”
. Discovery Repts., v. 4, p. 1–230, 1932. 
Fleming, Richard H.1940.
“The composition of plankton and units for reporting populations and production”
. Sixth Pacific Sci. Congr., Calif., 1939, Proc.v. 3, p. 535–540, 1940. 
Gilson, H. C.1937.
“The nitrogen cycle, John Murray Exped., 1933–34”
, Sci. Repts., v. 2, no. 8, p. 21–81, 1937. 
Hutchinson, A. H.1928.
“A bio-hydrographical investigation of the sea adjacent to the Fraser River mouth”
. Roy. Soc. Can., Trans., v. 22, Sect. 5, p. 293–310, 1928. 
Jowett, W. G., and W. Davies. 1938.
“A chemical study of some Australian fish”
. Australia, Council for Sci. and Industr. Research, Pamphlet 85, 40 pp., 1938. 
Keys, Ancel, E. H. Christensen, and A. Krogh. 1935.
“The organic metabolism of sea water with special reference to the ultimate food cycle in the sea”
. Marine Biol. Assn. U. K., Jour., v. 20, p. 181–96, 1935. Plymouth. 
King, E. J., and Viola Davidson. 1933.
“The biochemistry of silicic acid. IV. Relation of silica to the growth of phytoplankton”
. Biochem. Jour., v. 27, p. 1015–21, 1933. 
Krogh, August. 1931.
“Dissolved substances as food of aquatic organisms”
. Conseil Perm. Internat. p. l'Explor. de la Mer, Rapp. et Proc.-Verb., v. 75, p. 7–36, 1931. 
Krogh, August. 1934a.
“Conditions of life in the ocean”
. Ecol. Monographs, v. 4, p. 421–29, 1934. 
Krogh, August. 1934b.
“Conditions of life at great depths in the ocean”
. Ecol. Monographs, v. 4, p. 430–39, 1934. 
Krogh, August, and Ancel Keys. 1934.
“Methods for the determination of dissolved organic carbon and nitrogen in sea water”
. Biol. Bull., v. 67, p. 132–44, 1934. 
McCance, R. A., and M. Masters. 1937–38.
“The chemical composition and acid base balance of Archidoris brittanica.”
. Marine Biol. Assn., U. K., v. 22, p. 273–79, 1937–38. Plymouth. 
Moberg, E. G., and R. H. Fleming. 1934.
“The distribution of nitrogen compounds in the sea near southern California”
. Fifth Pacific Sci. Congr., Canada, 1933, Proc., v. 3, p. 2085–88, 1934. 
Phelps, Austin. 1937.
“The variation in the silicate content of the water in Monterey Bay, California, during 1932, 1933, and 1934”
. Amer. Phil. Soc., Trans., v. 29, p. 153–88, 1937. 
Phifer, Lyman D., and T. G. Thompson. 1937.
“Seasonal variations in the surface waters of San Juan Channel during the five-year period, January 1931 to December 30, 1935”
. Jour. Marine Research, v. 1, p. 34–53, 1937–38. 
Rakestraw, Norris W.1936.
“The occurrence and significance of nitrite in the sea”
. Biol. Bull., v. 71, p. 133–67, 1936. 
Rakestraw, N. W., and H. P. Smith. 1937.
“A contribution to the chemistry of the Caribbean and Cayman Seas”
. Bingham Oceanogr. Coll., Peabody Museum, Bull., v. 6, art. 1, 41 pp., 1937. 
Redfield, A. C.1934. On the proportions of organic derivatives in sea water and their relation to the composition of plankton. p. 176–92 in James Johnstone Memorial Volume, Liverpool, Univ. Press, 348 pp., 1934. 
Redfield, A. C., and Ancel Keys. 1938.
“The distribution of ammonia in the waters of the Gulf of Maine”
. Biol. Bull., v. 74, p. 83–92, 1938. 
Redfield, A. C., H. P. Smith, and B. Ketchum. 1937.
“The cycle of organic phosphorus in the Gulf of Maine”
. Biol. Bull., v. 73, p. 421–43, 1937. 
Robertson, J. D.1939.
“The inorganic composition of the body fluids of three marine invertebrates”
. Jour. Exp. Biol., v, 16, p. 387–97, 1939. 
Robinson, R. J., and H. E. Wirth. 1934a.
“Report on the free ammonia, albuminoid nitrogen and organic nitrogen in the waters of the Puget Sound area, during the summers of 1931 and 1932”
. Conseil Perm. Internat. p. l'Explor. de la Mer, Journal du Conseil, v. 9, p. 15–27, 1934. 
Robinson, R. J., and H. E. Wirth. 1934b.
“Free ammonia, albuminoid nitrogen and organic nitrogen in the waters of the Pacific Ocean off the coasts of Washington and Vancouver Island. Conseil Perm”
. Internat. p. l'Explor. de la Mer, Journal du Conseil, v. 9, p. 187–95, 1934. 
Rogers, C. G.1938. Textbook of comparative physiology. 2nd ed.. New York, McGraw-Hill, 715 pp., 1938. 
Seiwell, H. R.1935.
“The cycle of phosphorus in the western basin of the North Atlantic. I. Phosphate phosphorus”
. Papers in Physical Oceanogr. and Meteorol., v. 3, no, 4, 56 pp., 1935. 
Seiwell, H. R., and G. E. Seiwell. 1938.
“The sinking of decomposing plankton in sea water and its relationship to oxygen consumption and phosphorus liberation”
. Amer. Phil. Soc., Proc., v. 78, p. 465–81, 1938. 
Ström, K. M.1936.
“Land-locked waters. Hydrography and bottom deposits in badly ventilated Norwegian fjords with remarks upon sedimentation under anaerobic conditions”
. Norske Vidensk. Ak. i Oslo, 1. Mat.-Naturv. Klasse, no. 7, 85 pp., 1936. 
Thompson, T. G., B. D. Thomas, and C. A. Barnes. 1934. Distribution of dissolved oxygen in the North Pacific Ocean. p. 203–34 in James Johnstone Memorial Volume, Liverpool Univ. Press, 348 pp., 1934. 
Thomsen, Helge. 1931.
“Nitrate and phosphate contents of Mediterranean water”
. Danish Oceanogr. Exped. 1908–1910, v. 3, no. 6, 14 pp., 1931. 
Thomsen, Helge. 1937.
“Hydrographical observations made during the Dana Expedition 1928–1930”
. Dana Repts., no. 12, 46 pp., 1937. 
Vinogradov, A. P.1935.
“Elementary chemical composition of marine organisms. (In Russian) Pt. 1. Laboratoire de Biogeochimie près l'Acad”
. des Sci., U.R.S.S., Travaux, v. 3, p. 63–278, 1935. Leningrad. 
Vinogradov, A. P.1937.
“Elementary chemical composition of marine organisms”
. (In Russian) Pt. 2. Laboratoire de Biogeochimie près l'Acad. des Sci., U.R.S.S., Travaux, v. 4, p. 5–225, 1937. Leningrad. 
Vinogradov, A. P.1938.
“Chemical composition of marine plankton. (In Russian.)”
. Inst. Marine Fisheries and Oceanogr. of the U.S.S.R., Trans., v. 7, p. 97–112, 1938. Moscow. 
von Brand, Theodor. 1935.
“Methods for the determination of nitrogen and carbon in small amounts of plankton”
. Biol. Bull., v. 69, p. 221–32, 1935. 
von Brand, Theodor. 1937.
“Observations upon the nitrogen of the particulate matter in the sea”
. Biol. Bull., v. 72, p. 1–6, 1937. 
von Brand, Theodor. 1938.
“Quantitative determination of nitrogen in the particulate matter of the sea”
. Conseil Perm. Internat. p. l'Explor. de la Mer, Journal du Conseil, v. 13, p. 187–96, 1938. 
von Brand, Theodor, N. W. Rakestraw, and C. E. Renn. 1937.
“The experimental decomposition and regeneration of nitrogenous organic matter in sea water”
. Biol. Bull., v. 72, p. 165–75, 1937. 
von Brand, Theodor, N. W. Rakestraw, and C. E. Renn. 1939.
“Further experiments on the decomposition and regeneration of nitrogenous organic matter in sea water”
. Biol. Bull., v. 77, p. 285–96, 1939. 
Waksman, S. A., and C. E. Renn. 1936.
“Decomposition of organic matter in sea water by bacteria. III. Factors influencing the rate of decomposition”
. Biol. Bull., v. 70, p. 472–83, 1936. 
Webb, D. A., and W. R. Fearon. 1937.
“Studies on the ultimate composition of biological material. Pt. I. Aims, scope and methods”
. Roy. Dublin Soc., Sci. Proc., n.s., v. 21, p. 487–504, 1937. 
ZoBell, Claude E.1933.
“Photochemical nitrification in sea water”
. Science, v. 77, p. 27–28, 1933. 
ZoBell, Claude E.1935a.
“The assimilation of ammonium nitrogen by Nitzschia closterium and other marine phytoplankton”
. Nat. Acad. Sci., Proc.v. 21, p. 517–22, 1935. 
ZoBell, Claude E.1935b.
“Oxidation-reduction potentials and the activity of marine nitrifiers”
. Abstract. Jour. Bacter., v. 29, p. 78, 1935. 
ZoBell, Claude E.1940.
“The effect of oxygen tension on the rate of oxidation of organic matter in sea water by bacteria”
. Jour. Marine Research, v. 3, p. 211–23, 1940. 
ZoBell, Claude E., and D. Q. Anderson. 1936.
“Observations on the multiplication of bacteria in different volumes of stored sea water and the influence of oxygen tension and solid surfaces”
. Biol. Bull., v. 71, p. 324–42, 1936.