6.5.2—
Metabolic Reactions of the Peroxisome
It was generally accepted that the enzymes of the glycollate pathway were soluble proteins of the cytosol and the failure of several attempts to localize these enzymes, particularly glycollate oxidase, in discrete organelles confirmed this idea. The first successful localization of enzymes of glycollate metabolism in a discrete organelle was achieved by Tolbert and coworkers at Michigan State University (Tolbert et al., 1968). They demonstrated that sucrose density gradient centrifugation of spinach homogenates separated three bands of particles: broken chloroplasts, mitochondria, and small bodies distinctly separated from, and denser than the other organelles. Electron microscopic examination of these bodies showed them to be organelles bounded by a single unit membrane and since they closely resembled peroxisomes from animal cells in size and morphology, they were referred to as leaf peroxisomes. The most important finding was that these peroxisomes contained the bulk of the activity of the glycollate oxidase, catalase and hydroxypyruvate reductase (NAD-glyoxyllate reductase) of the gradient, while cytochrome c oxidase was specifically located in the mitochondrial fraction. Peroxisomes were later
isolated from the leaves of nine other plant species including tobacco, maize, and sugarcane, and all contained the same enzyme complement (Tolbert et al., 1969). These studies therefore demonstrated that processes of glycollate oxidation and peroxide breakdown were localized together in an organelle discrete from the chloroplast. Electron microscope studies, particularly by Newcomb, have since revealed the presence of peroxisomes in leaves of many plant species (e.g. Frederick & Newcomb, 1969).
Further studies in Tolbert's laboratory have shown that other enzymes of the glycollate pathway are also localized in the peroxisome. Two aminotransferases, glutamate-glyoxyllate aminotransferase catalysing the conversion of glyoxyllate to glycine, and serine-pyruvate aminotransferase catalysing the conversion of serine to hydroxypyruvate, were found in peroxisomes isolated from leaves of various species. Serine hydroxymethyltransferase is the only enzyme of the glycollate pathway not found in the peroxisome, and is probably located in the mitochondrion. Supply of [14 C] glycollate and [14 C] glyoxyllate to isolated peroxisomes gave rise only to [14 C] glycine and while oxygen uptake occurred, no 14 CO2 release was detected (Kisaki & Tolbert, 1969).
The glycollate pathway appears to require enzymic steps located in three subcellular organelles (Fig. 6.8). Glycollate, formed in the chloroplast is
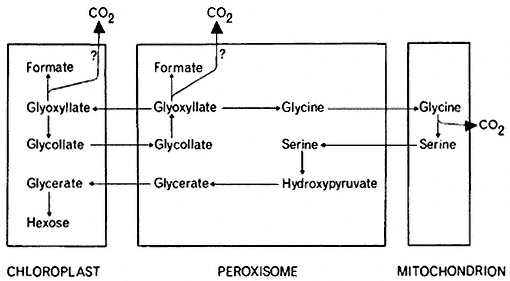
Figure 6.8
The distribution of the reactions of the glycollate
pathway among organelles of the leaf cell.
oxidized to glyoxyllate in the peroxisome. The glyoxyllate may then be exported to the chloroplast where it could be reduced to glycollate by the action of NADP-dependent glyoxyllate reductase which is located specifically in the chloroplast (Tolbert et al., 1970). Such a coupling of alternate oxidation and reduction reactions. a 'glycollate-glyoxyllate shuttle', has been proposed as a mechanism for controlling the levels of reduced NADP in the chloroplast, but no unequivocal evidence for such a reaction in vivo has been found. Glycine is formed from glyoxyllate in the peroxisome and then transferred to the
mitochondrion where it is converted to serine, with a concomitant loss of carbon dioxide. The conversion of serine to glycerate can then be accomplished by enzymes localized in the peroxisome. Further metabolism of glycerate to hexose appears to be confined to the chloroplast since these reactions would be initiated by the formation of phosphoglyceric acid, a reaction catalysed by phosphoglycerate phosphatase which is located in the chloroplast.
The metabolism of glycollate by leaf tissue results in the release of carbon dioxide, but the exact site of this CO2 release in the cell is still a controversial question. It has been proposed that CO2 is evolved by the mitochondria during the conversion of glycine to serine, since [14 C] glycine is as good a precursor as [14 C] glycollate for 14 CO2 evolution in leaves and the 14 CO2 evolved is derived from the carboxyl groups of these compounds. The presence of catalase in the peroxisomes is thought to minimize the non-enzymatic oxidation of glyoxyllate by H2 O2 to formate and carbon dioxide and no evolution of CO2 by isolated peroxisomes from glycollate or glyoxyllate could be detected by some workers (Kisaki & Tolbert, 1970). This loss of CO2 from glycine, which amounts to only 25% of the total carbon passing through the glycollate pathway, would not account for the large losses of CO2 which occur as a result of the photorespiration of glycollate in leaves. However the amount of catalase present in the peroxisome may not preclude the non-enzymatic oxidation of glyoxyllate and it has been shown that both [14 C] glycollate and [1-14 C] glyoxyllate can be decarboxylated by peroxisomal fractions at pH 8.0 (Halliwell & Butt, 1974). An enzyme is also present in chloroplasts which catalyses the decarboxylation of glyoxyllate to formic acid and CO2 (Zelitch, 1972). Present evidence indicates therefore that three subcellular organelles have the capacity to decarboxylate components of the glycollate pathway and each may contribute to the production of the carbon dioxide in photorespiration (Fig. 6.8).