PART ONE
THE DIAGNOSIS
The machine itself makes no demands and holds out no promises: it is the human spirit that makes demands and keeps promises.
Lewis Mumford Technics and Civilization
1
The Diagnostic Framework
The gains in technics are never registered automatically in society; they require equally adroit inventions and adaptations in politics; and the careless habit of attributing to mechanical improvements a direct role as instruments of culture and civilization puts a demand upon the machine to which it cannot respond….
No matter how completely technics relies upon the objective procedures of the sciences, it does not form an independent system, like the universe; it exists as an element in human culture and it promises well or ill as the social groups that exploit it promise well or ill. The machine itself makes no demands and holds out no promises: it is the human spirit that makes demands and keeps promises.
Lewis Mumford Technics and Civilization
The Problem
Medical devices, the "technics" of this book, pervade our experiences with health care throughout our lives—from fetal monitoring equipment and ultrasound imaging before birth to life-support systems and even suicide machines when death is near.[1]
Lewis Mumford, Technics and Civilization (New York: Harcourt Brace Jovanovich, 1934).
Medical care has become increasingly dependent on technology, and medical devices are the epitome of the trend. The modern hospital is a wonderland of complicated machinery, and the rhythmic beeping of heart monitors evokes a life-and-death drama. Tens of thousands of Americans depend upon artificial body parts for survival and to improve the quality of their lives—fromhips to intraocular lenses to heart pacemakers. Even doctors' offices are full of new devices. Physicians use lasers to remove cataracts without the need for hospitalization; ultra-sound monitors take pictures of unborn babies. The allure of medical innovation is powerful, holding out the possibility of a perfect outcome, an amelioration of pain, a delay in our inexorable decline toward death.
Spending on medical devices reflects this trend. This multi-billion dollar industry producing thousands of products may account for as much as 40 percent of the health care bill, which has grown to close to 12 percent of the gross national product.[2]
Forecasts placed health care as rising from 11.9 percent of the GNP in 1990 to 13.1 percent in 1995. Alden Solovg, "Recession Prospects Mixed Bag for Health Care," cited in Medical Benefits 6 (30 August 1989).
(See figure 1.) The demand seems insatiable. We have come to expect a steady stream of new "miracles." Experts predict bloodless laser surgery in the near future and a genetic engineering revolution at the turn of the century. "At the current rate of innovation," an analyst recently projected, "by the year 2000 close to 100,000 new or enhanced medical devices will be introduced into the marketplace. Health care will be a $1.5 trillion industry."[3]Russell C. Coile, "Advances in the Next Decade Will Make Today's Technology Seem Primitive," cited in Medical Benefits 6 (30 August 1989).
Demand is fueled by our belief in equitable access to medical care. When a medical innovation is considered beneficial, there is pressure to distribute its benefits to all who need it. News stories of people denied access to high-cost treatments because of their inability to pay generate public sympathy and, often, outrage. Until very recently, the ideal of the highest quality care for everyone has been rarely questioned, albeit unattained.
Medical technology is not without critics who challenge any unquestioning belief in its value. Some argue that in our love affair with technology we have sacrificed the caring, or service, side of medicine.[4]
Stanley Joel Reiser, Medicine and the Reign of Technology (Cambridge: Cambridge University Press, 1978).
The public is also intolerant of technologies that cause harm. The dangers of the Dalkon Shield, an intrauterine device (IUD) that injured thousands of women, received widespread publicity. In a recent controversy, the government has charged that a medical device producer marketed heart valves it knew to be defectively designed, leaving thousands in daily fear that their implanted devices will fail and kill them.[5]The risks of the Bjork-Shiley heart valve, an implanted disk that controls the flow of blood through the heart, received publicity in 1990. The valve has a tendency to fail in some cases, leading to the deaths of recipients. The legal and regulatory issues raised by this medical device are discussed in chapter 6.
Some blame medical technology for the escalating costs of care as well. There is no question that some technology is expensive, but other products reduce costs through early diagnosis or
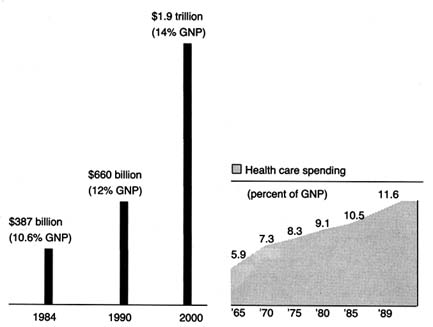
Figure 1. Forecasted health spending.
Sources: (a) M. S. Freeland et al., Health Care Financing Review 6 (Spring 1985): 1–20;
and K. W. Tyson and J. C. Merrill, Journal of Medical Education 59 (1984): 773–81;
(b) Health Care Financing Administration.
shorter hospital stays. Pressures to control costs have led to calls for elimination of wasteful or unnecessarily expensive equipment. Exposure of fraud in the cardiac pacemaker industry and of unnecessary implants of intraocular lenses lend credence to this charge.[6]
Cardiac pacemakers are discussed in chapters 4 and 5; intraocular lenses are studied in chapter 7.
Even highly desirable technologies are controversial. There can be tension between providing access to expensive lifesaving devices to a few and more widespread access to lower-cost benefits to many. In a recent policy decision, Oregon medical officials declared that the Medicaid program, which serves the poor in that state, would no longer pay for liver transplants, a desirable high-cost, lifesaving technology, in order to use the funds to cover prenatal care for a larger number of low-income pregnant women. The state also recommended a priority list to determine which treatments would be covered under the program.[7]
See Karen Southwick, "Oregon Blazing a Trail with Plan to Ration Health Care," Healthweek, 12 March 1990, 30, 33. To implement its plan, Oregon needs waivers from some of the federal Medicaid requirements. In expanding coverage for poor families by restricting benefits, Oregon would violate a requirement that families receiving federal aid automatically receive full Medicaid coverage as well. Waivers can be granted administratively or through Congress. There is much political controversy about the rationing scheme. By the end of 1990, Oregon was revising its final priority list, and Congress was in a "wait-and-see" mode. Virginia Morell, "Oregon Puts Bold Health Plan on Ice," Science 249 (3 August 1990): 468-471.
This isone example of the many explicit and implicit tradeoffs that are made as public and private budgets are stretched to the limit.
Medical device innovations—with lifesaving promises, potential risks, and often high price tags—are deeply embedded in the broad debates about health policy. The insatiable demand, fed by a profusion of new technologies and the profits they represent, has been called the medical arms race. Increasingly, government has been called upon to manage it.
This book explores the "adroit inventions and adaptations in politics" that have accompanied the profusion of medical devices in our society. As government has become inextricably linked to health services, public policies affecting medical devices have proliferated. These policies reflect diverse values and arise from many different institutions. Because of the pervasiveness of public policy, the medical device industry can only be understood in relation to the policy environment. Indeed, the industry grew and matured in response to public policy incentives. Those responses, in turn, brought new layers of public policy.
A medically related analogy will help to structure the discussion. Policy proliferation is analogous to polypharmacy, an increasingly familiar condition to the health care profession. Polypharmacy occurs when a patient takes a number of prescription drugs. Each prescription may have been given for a legitimate ailment, but the interactions between the drugs can harm the patient. Attentive doctors routinely request that patients put all their drugs in a brown paper bag and bring them in for review. The review evaluates what is known about the drugs and what is known about the particular patient. There may be new information about a drug product, and there may also be changes in the patient's underlying condition. A physician must look for possible interactions between the drugs. Some reactions may be previously unknown or unexpected; some predictable and even tolerable. Some may dissipate the efficacy of other prescriptions and require modifications of dosage, and some may be fatal.
This book provides a "brown-bag" review of all the policies that directly and indirectly affect the medical device industry. The industry is the patient; the present policies are the prescriptions to which the patient reacts and responds. The book begins the diagnosis by asking key questions: What is the impact
of these multiple prescriptions on the patient? On the medical device industry? Are our prescriptions producing desirable outcomes without adverse and unexpected side effects? Is the result good health—in this case, meaningful innovation that is safe, efficacious, efficient, and cost effective? If not, what revisions in the treatment are appropriate? What is the patient's prognosis?
To answer these questions, we must take the patient's history, review each prescription to determine when and why it was offered, how it has changed over time, and how it relates to other prescriptions. After extensive review of these factors, the conclusion is mixed. It would, of course, be dramatic to report that government policy has destroyed device innovation; that is, the side effects are worse than the treatment. The reality is less definitive and more elusive. Until the 1990s, we have managed to muddle along, balancing myriad policy goals. Innovation flourished in the 1950s and 1960s because the dominant policies promoted both discovery of new products and their widespread distribution. Innovation has survived in the 1970s and 1980s despite safety and cost-control policies that inhibit innovation. The momentum of innovation was sustained in part because these policies were relatively unsuccessful in accomplishing their goals. Regulation was not fully implemented as intended, product liability had only sporadic effects on certain devices, and cost-containment strategies could not combat the pressure for distribution of benefits. If these policies had been successful, the industry would have been more adversely affected.
What does the future hold? We can expect renewed efforts to impose greater regulation and more effective cost controls in the 1990s. There is clear evidence of overdiffusion that government may try to control. As the marketplace attracts more equipment and as technicians and specialists arise to operate it, there can be supply-induced demand. In other words, if a facility has an MRI machine, it will find the patients necessary to operate the machine at a profit.
Policies to control supply costs can be on a collision course with the competing desire for more innovative devices. When public policies clash, there can be serious adverse reactions for innovation. The departure of most childhood vaccine producers in the wake of product liability suits and the impact of regulation
and liability on innovation in contraceptive research and development have been well documented.[8]
An excellent study on the impact of policies on contraceptive research is Luigi Mastroianni, Jr., Peter J. Donaldson, and Thomas T. Kane, eds., Developing New Contraceptives: Obstacles and Opportunities (Washington, D.C.: National Academy Press, 1990).
These cases represent potential adverse effects of public policy that we must seek to avoid with other devices.Muddling through the 1990s will not be acceptable. If we allow the situation to drift, we could become passive observers of unwanted outcomes. However, prevention of harmful effects is possible. Reforms that affirmatively balance competing values while eliminating conflicts and redundancies are presented here. Incremental accommodations can relieve some of the stresses on the system. But we must recognize the limits of medical device technology to cure all ills and the limits of public policy to solve social problems. Indeed, wise policy must address the larger moral questions of how we want to live and how we want to die: difficult questions we tend to avoid.
Medical devices are sufficiently important to warrant this case study on its own merits. However, this book also presents some of the broader concerns of public policy. It contributes to our understanding of policy proliferation. In recent years, the government has developed a propensity to intervene in the economy. For example, federal agricultural policy has been described as "a complex web of interventions covering output markets, input markets, trade, public good investments, renewable and exhaustible natural resources, regulation of externalities, education, and marketing and distribution of food products."[9]
Gordon C. Rausser, "Predatory Versus Productive Government: The Case of U.S. Agricultural Policies," Journal of Economic Perspectives 2 (Winter 1992).
A historical review of these public policies reveals the tension in the government's arguably contradictory actions.[10]Rausser defines some government policy as productive (reducing transaction costs and correcting market failures) and other policy as predatory (redistributing wealth without concern for growth or efficiency). His work seeks to explain and reconcile the perceived conflicts between the two approaches.
Public policy toward tobacco also illustrates this conflicting approach.[11]Congress protects the tobacco industry with a variety of favorable economic policies while simultaneously inhibiting the sale of tobacco products through television advertising limits and warning label requirements. Officials in several administrations have used their positions to condemn the marketing and the use of tobacco, and states and localities have severely restricted or banned smoking in public places.
Additional examples include nuclear power, the oil industry, and automobiles.[12]The government protects the automobile industry with negotiated trade restrictions while also regulating automobile design to promote safety and environmental goals. Other federal policies regarding gasoline pricing and supply, highway construction, and alternative forms of transportation all affect the infrastructure upon which the automobile depends. The 1989 Alaskan oil spill revealed both redundancies and regulatory gaps between federal and state authorities and illustrated the problems that can arise when various government institutions impose overlapping or conflicting demands.
Some degree of policy proliferation is inevitable in the American system. A pluralistic and democratic society tends toward incrementalism and compromise—features that encourage the proliferation of smaller interventions rather than comprehensive unilateral policies. Separation of powers at the federal level increases the likelihood of multiple sources of intervention, with each branch employing different tools and involving different constituencies. Our national government also shares power with the fifty states.[13]
There is a vast literature on federalism. For an overview, see David B. Walker, Toward a Functioning Federalism (Cambridge, Mass.: Winthrop Publishers, 1981). For a discussion of federalism and health, see Frank J. Thompson, "New Federalism and Health Care Policy: States and the Old Questions," Journal of Health Politics, Policy and Law 11 (1986): 647-669.
Thus, multiple layers of regulation have becomethe norm, not the exception, which is particularly true in the health care delivery and regulation system because of its complexity.
This book on medical device technology may have more general relevance to other areas of the economy. Of course, perfect generalizations cannot always be drawn from a specific case; each area has individual characteristics, and the variety of prescriptions will inevitably differ. However, the search for patterns and relationships in the evolution of various policies, and the study of private-sector responses to them, may be instructive for other areas of business-government relations. This chapter will first familiarize the reader with the patient and then establish the framework for the diagnostic process.
The Patient: The Medical Device Industry
Defining a Medical Device
The term medical device is often used synonymously with medical products, medical equipment and supplies, or medical technology. As a working definition, that of the Federal Food, Drug, and Cosmetic Act seems appropriate: a medical device is "an instrument, apparatus, implement, machine, contrivance, implant, in vitro reagent, or other similar or related article" that is intended for use in "the diagnosis of disease or other conditions [or the] cure, mitigation, treatment, or prevention of disease [or] intended to affect the structure or any function of the body of man, which does not achieve any of its principal intended purposes through chemical action within or on the body of man or other animals and which is not dependent upon being metabolized for the achievement of any of its principal purposes."[14]
21 U.S.C.sec. 321(h). This definition appears in the 1976 Medical Device Amendments to the Food, Drug, and Cosmetic Act, discussed at great length in chapter 5.
The definition is less complicated than it may first appear. Basically, devices include health care items other than drugs. Devices used for diagnosis of disease include X-ray machines, scopes for viewing parts of the body, including stethoscopes and bronchoscopes, and heart monitoring equipment, to name a few. Devices used for cure or treatment include scalpels and more complicated surgical tools, lithotripsy devices that crush kidney stones with ultrasonic waves, and balloon catheters that
clean out plaque formations on arteries. Examples of products that affect the structure or function of the body include artificial lenses to correct impaired vision, diaphragms or intrauterine devices used for birth control, and cardiac pacemakers to regulate heart rhythms.
The term medical devices can be distinguished from medical products, which includes both drugs and medical devices despite the distinct biomedical properties of each. Medical devices do include, but are not limited to, all nonpharmaceutical medical equipment and supplies. The term medical technology is even broader; the Office of Technology Assessment (OTA) has defined medical technology as "drugs, devices, and medical and surgical procedures used in medical care, and the organizational and supportive systems within which such care is provided."[15]
The Office of Technology Assessment is a research arm of Congress and produces technical reports and evaluations at its request.
Thus, devices are only one part of medical technology.Medical devices include thousands of products currently produced by over 3,500 U.S. firms. The term encompasses all supplies and equipment used in hospitals from bedpans to sophisticated monitoring devices, diagnostic products from X-rays and lab kits to complex innovations such as magnetic resonance imaging and ultrasound, and nonpharmaceutical treatment products from bandages to laser surgery equipment. Outside the hospital, medical devices are found in physicians' offices, from stethoscopes and blood pressure cuffs to automated desk-top blood analyzers and portable electrocardiograph (EKG) machines. Medical devices are also used in the home, including over-the-counter articles such as pregnancy test kits and heating pads.
Use of the definition crafted by the Food and Drug Administration (FDA) is noteworthy. First, in that this book explores the complex interrelationship between the government and the producers of devices, it is interesting that it was the FDA, a government agency, that first struggled with a precise definition of medical devices in order to formulate regulatory policy. Indeed, government helped to shape the industry by defining who was in or out for regulatory purposes. It is also interesting to note that the definition is residual, defining medical devices as products that are essentially nondrugs—reflecting the fact that the FDA regulated drugs long before medical devices were considered an
important part of health care.[16]
For a discussion of the history of the FDA, see chapter 2.
As the number and complexity of these nondrugs grew, a residual, catch-all category had to be carved out. This history explains why federal regulation treats the device category as a stepchild to the more clearly identifiable drug category.Inevitably, as in most definitions, there remain some gray areas. For example, the swimming pool that one may use to alleviate back pain generally would not be considered a device, at least for regulatory purposes. Sometimes confusion arises between the treatment process and the product because in many cases the treatment cannot be given without a particular medical device. For example, a patient cannot receive electroshock treatment without an electroshock device. Thus, some efforts to regulate the device were thinly disguised attempts to ban the treatment.
Unfortunately, the FDA definition, which works well enough for regulatory purposes, is different from the categories traditionally used by government to collect data on the industry that produces medical devices. The Census of Manufactures contains comprehensive industry statistics compiled by the U.S. Department of Commerce, Bureau of the Census. These data group medical products into five Standard Industrial Classifications (SIC codes), including designations for surgical and medical instruments, surgical appliances and supplies, X-ray, electromedical, and electrotherapeutic apparatus, dental equipment, and ophthalmic goods (see table 1). The data in these classifications capture an estimated 50 to 75 percent of products defined as medical devices by the FDA.[17]
Foote, "From Crutches to CT Scans," 4.
In the aggregate, these data, while imperfect, are sufficient to evaluate trends in the industry.Distinguishing Medical Products from Other Consumer Goods
Health care, like education, has never been officially considered a fundamental right under the U.S. Constitution. However, many of our health policies reflect the view that health care, like education, is a form of entitlement, a human right, rather than a privilege for the few. Social welfare programs have been designed to ensure that the elderly, the disabled, and the poor receive adequate, or at least minimal, health care services.
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Clearly, medical products, including both drugs and devices, are deeply embedded in and inextricably linked to health care.
The social benefits associated with medical products distinguish them from other consumer goods. Economists use the term merit goods to describe products that have greater significance to society than other consumer goods.[18]
Karl A. Fox, Social Indicators (New York: John Wiley, 1974).
Unlike toasters, lawnmowers, or home computers, medical products, and their availability and affordability, raise humanitarian as well as economic issues. As a result, tension has existed between the economics of medical care and the social side of health. For example, at the turn of the century, entrepreneurial scientists took out patents and channeled the profits into further research, their supporting institutions, or their own pockets. However, traditionalmedical ethics made this practice questionable for health innovations. "Medicine was ensnared, as usual, in complications resulting from its peculiar combination of business and social service."[19]
Richard H. Shyrock, American Medical Research, Past and Present (New York: The Commonwealth Fund, 1947), 140.
We have not resolved this "peculiar" combination today. A recent article on financial issues for end-stage renal disease (ESRD) patients asks: "[I]s it a good idea to motivate patients … to choose their kidney dialysis units on the basis of price as well as medical quality, personal convenience, and other important factors?"[20]
Randall R. Bovbjerg, Philip J. Held, and Louis H. Diamond, "Provider-Patient Relations and Treatment Choice in an Era of Fiscal Incentives: The Case of the End-Stage Renal Disease Program," Milbank Quarterly 65 (1987): 177-202, 177.
It is difficult to imagine advising consumers of automobiles or financial services to ignore price solely in the interests of quality or convenience. In essence, important and long-standing social values undergird our views of health care and are reflected in the public policies that have emerged.Distinguishing Medical Devices from Drugs
Although pharmaceuticals provide the closest analogy to medical devices, we should not assume that drug and device issues are identical. Both drugs and devices are essential for the treatment and diagnosis of disease, but there are very important differences as well. Recalling the FDA definition, we know that drugs and devices operate through different biomedical mechanisms—most drugs are metabolized, and devices are not. The size and the composition of the marketplace and the range of producers differ in the two industries.
Of course, drugs and medical devices are often used in concert; for example, syringes and intravenous equipment deliver drugs directly into the body. Innovative skin patches allow gradual absorption of chemicals through the skin for various medical treatments, such as for motion sickness. Drugs and devices may offer alternative treatments. Patients may choose chemotherapy, a drug treatment, over surgery, a procedure, to eradicate cancer; women can choose among birth control pills (drugs), diaphragms, and IUDs (devices) to prevent pregnancy.
The governmental distinction between drugs and medical devices is more than a biomedical nicety. Device technologies range from lasers to computer systems to implanted materials. Because of that diversity, the nature of the medical equipment
and the costs associated with its purchase and use vary much more widely than for drugs. That is not to say that all pharmaceuticals are cheap. Indeed, recent introductions, particularly products based on biotechnology, carry high price tags. Tissue plasminogen activator (TPA), a drug for treatment of stroke victims, can cost as much as $5,000 a dose.[21]
In 1990, one dose of TPA cost $2,200, in contrast to the drug it, claims to replace, streptokinase, which cost $200 a dose. U.S. sales of TPA in 1989 were nearly $200 million. Karen Southwick, "Analysts Say TPA Use May Drop in Wake of Study," Healthweek, 26 March, 1990, 45.
Recent controversies over the high price of zidovudine (AZT), one of the few effective drugs for AIDS patients, raises the same issue. However, price variability in devices is still much broader. Devices range from simple products, such as crutches and bandages, to high-cost capital equipment, such as laboratory blood analyzers, lithotripsy equipment, and magnetic resonance imaging machines that cost several million dollars. Unlike drugs, such equipment may require maintenance, specially designed facilities, and specially trained operators and are subject to depreciation and deterioration. Other devices, such as components for kidney dialysis, raise important questions of reuse not relevant for drugs.In addition, the medical devices market may differ from the drug market. While hospital pharmacies generate sales, physicians generally order prescription drugs for individual patients, and consumers can purchase over-the-counter (OTC) drugs directly. The primary purchasers of medical equipment are hospitals, which rank ahead of physicians, ambulatory care centers, and individuals. Hospital purchasing patterns are extremely sensitive to changing reimbursement policies by third-party payers.
There are significant distinctions between drugs and devices on the supply side as well. Because of the range of technologies embedded in medical device production, a widely diverse group of firms consider themselves part of the medical device industry. In pharmaceuticals, the top one hundred companies market 90 percent of the drugs.[22]
Grabowski and Vernon, Regulation of Pharmaceuticals, 18.
The device industry is not nearly as concentrated: currently, over 3,500 device companies produce several thousand products. These companies range from those also known for drugs, such as Johnson & Johnson and Pfizer, to electronic giants, such as General Electric and Hewlett-Packard. In addition, there are many smaller companies concentrating exclusively on medical devices, such as Alza Corporation, whichspecializes in innovative drug delivery systems, and Medtronic, the leader in heart pacemakers and heart valves. Others have diversified. For example, SpaceLabs, now owned by Bristol-Myers/Squibb, applied technologies that were developed for the space program to produce a variety of monitoring and data-recording devices.
With all this diversity, it helps to consider medical device producers as a single industry. While this industry is many industries, in one sense, because no substitutability exists among all products, there is a commonality of buyers—hospitals, clinics, laboratories, physicians, and patients. However, there is a commonality in use in a broad sense—the products affect the function of the body and/or treat disease. Finally, it is the peculiar set of government health policies that have shaped the performance of this industry. All companies are, to some extent, offspring of the same set of policies, and this brings us full circle to the FDA definition of medical devices. The industry is unified by its relationship to government, both because it is defined by government and because of its symbiotic relationship to it.
The Process: A Diagnostic Framework
As this study explores the interaction between innovative medical devices and government, an understanding of both innovation and public policy is required.
Innovation Defined
Put most simply, innovation has been defined as certain technical knowledge about how to do things better than the existing state of the art.[23]
David J. Teece, "Profiting from Technological Innovation: Implications for Integration, Collaboration, Licensing, and Public Policy," Research Policy 15 (December 1986): 285-305.
Innovation can be seen broadly as a process leading to technical change, or the concept can be more narrowly applied to a specific new product or technique.The value of innovation to sustain economic growth and competitiveness and to improve the quality of life is not in question. Yet a clear understanding of how and why innovation occurs remains elusive. There is extensive literature, primarily in economics, on ways to model innovation.[24]
For example, in The Sources of Innovation (New York: Oxford University Press, 1988), Eric von Hippel questions the assumption that manufacturers are the primary source of innovation. He presents studies to show that the sources of innovation vary greatly, often coming from the suppliers of component parts or the product users. He then develops a theory of the functional sources of innovation. In another recent work, Economic Analysis of Product Innovation: The Case ofCT Scanners (Cambridge: Harvard University Press, 1990), Manuel Trajtenberg presents a method to estimate the benefits from product innovations that accrue to the consumer over time, focusing particularly on the interaction between innovation and diffusion. For those interested in pursuing the study of innovation, see also Nathan Rosenberg, Inside the Black Box: Technology and Economics (Cambridge: Cambridge University Press, 1982); and Edwin Mansfield, Industrial Research and Technological Innovation: An Econometric Analysis (New York: Norton, 1968). For a thoughtful effort to identify the components of innovation, see James J. Zwolenik, Science, Technology, and Innovation, prepared for the National Science Foundation (Columbus, Ohio: Battelle Columbus Labs, February 1973).
Despite these continuingefforts, however, other scholars of innovation have concluded that the forces that make for innovation are so numerous and intricate that they are not fully understood.[25]
John Jewkes et al., The Sources of Innovation (London: Macmillan, 1969).
It is generally agreed, however, that certain stages are essential to the innovative process. Figure 2 illustrates the relevant stages.
The innovative process begins with science, which involves the systematic study of phenomena purely to add to the sum total of human knowledge.[26]
Shyrock, American Medical Research, 2.
Drawing on a science base, technology is directed toward use and has two phases: invention and development. Invention is the first confidence that something should work and involves testing to demonstrate that it does work. Development encompasses a wide range of activities by which technical methods are applied to the new invention so that the task is more precisely defined, the search more specific, and the chances of success more susceptible to measure. In John Jewkes's words, "Invention is the stage at which the scent is first picked up, development the stage at which the hunt is in full cry."[27]Jewkes, Sources, 28.
Finally, at the end of the development stage, technical considerations give way to market concerns. Experts have broken this stage into adoption and diffusion. Once a product has been adopted by the relevant decision makers, diffusion rates depend on a variety of market factors.
For purposes of simplicity, this book distills the stages into two main categories. Science, invention, and development lead to the creation of the product itself—described here as discovery . The second stage—decisions to adopt and use a product—is captured by the term distribution .
While simplification is helpful for discussion, it is important not to forget the complexity of the process. The existence of one element in the innovation stream does not ensure forward movement. Scientific understanding does not always produce applied technical development. However, the converse appears to be true: the sequence of innovation cannot occur without all elements. There can be no invention without science. For example, despite commitment to find a cure for cancer or AIDS, the necessary products await greater scientific understanding of the diseases. Moreover, the process is not perfectly linear. For many innovations, early prototypes are tried by physicians, who may
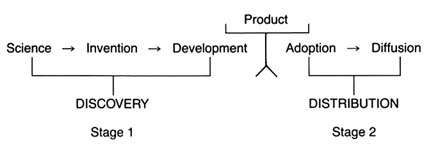
Figure 2. The stages of innovation.
encourage inventors to modify the product. Indeed, device innovation often is an iterative process involving inventors, physicians, and, occasionally, patients as well.
This innovative process can be illustrated by example. Infant mortality resulting from premature birth is a serious medical problem. Assume that physicians observed that pure oxygen is beneficial, but excessive oxygen may lead to blindness or death in the premature infant. The discovery stage would require knowledge of human physiology and of the role of oxygen. At the invention stage, the innovator would design a prototype for a mechanism to deliver the oxygen in appropriate amounts. That would require engineering expertise, such as a meter to measure oxygen flow and so on. The development stage would involve the production of the product.
Once the product is designed and constructed, the distribution stage becomes relevant. The inventor has to persuade physicians and/or hospitals to adopt the new invention, and diffusion measures how many products had been purchased to treat premature infants. Clearly, important factors arise that help or hinder the progress of the oxygen device along the innovation continuum. Questions of funding (who supports the inventor?), natural resources (is there an adequate supply of oxygen for the products?), and costs (can hospitals afford to buy the invention?) arise.
We can understand much of the medical device industry in relation to the traditional model of innovation. The industry is a business, subject to many of the same economic forces that confront all highly innovative industries. We need to know how technology is transferred, how firms are organized to facilitate
competitiveness, what economic strategies work, and so forth. However, in the medical device industry, as in many other areas of the economy, government policy has intervened at virtually every stage of the innovative process. Thus, to understand innovation, we must understand something of the public policy process as well.
Public Policy
Policymaking is the process of setting goals for the public good and implementing strategies to attain them. Every public policy is the outcome of an institutional decision. Public institutions, such as regulatory agencies or state courts, are themselves creatures of the political process. They have characteristics derived from their unique history and organization. In response to political, administrative, or legal pressures, public institutions can change. To understand public policy, then, we must be well grounded in the literature on bureaucracies, the judiciary, and the legislative process.[28]
The literature emerges primarily from the fields of political science, history, and law. It is impossible to provide a complete bibliography, but a good place to start an inquiry on how government agencies work is James Q. Wilson, Bureaucracy: What Government Agencies Do and Why They Do It (New York: Basic Books, 1989) and his earlier book, The Politics of Regulation (New York: Basic Books, 1980). See also James O. Freedman, Crisis and Legitimacy: The Administrative Process and American Government (Cambridge: Cambridge University Press, 1978). For discussion of how bureaucrats make decisions, see Eugene Bardach and Robert Kagan, Going By the Book: The Problem of Regulatory Unreasonableness (Philadelphia: Temple University Press, 1982); and Graham Allison, Essence of Decision (Boston: Little, Brown, 1971). For an understanding of the legislative process, begin with Eric Redman, The Dance of Legislation (New York: Simon and Schuster, 1973); and Hedrick Smith, The Power Game (New York: McGraw-Hill, 1988). For introduction to the judiciary, see Robert A. Carp and Ronald Stidham, Judicial Process in America (Washington, D.C.: Congressional Quarterly Press, 1990). For a discussion of litigation, see Jethro K. Lieberman, The Litigious Society (New York: Basic Books, 1981).
The public policies that affect medical device innovation must be understood in institutional, political, and legal contexts. History helps to establish the political and social context in which the policy intervention was introduced. For medical devices specifically, the institutions that set public policy include the National Institutes of Health (NIH), the FDA, the Medicare and Medicaid bureaucracies (the Health Care Financing Administration at the federal level for Medicare and many various departments of health in each state for Medicaid administration), the state and federal courts, and a variety of other assessment and regulatory entities.
Each policy must also be understood in terms of the social values that motivated the initial intervention. Like the concept of innovation, the concept of value is elusive. The term has been given so many meanings by economists, philosophers, and the public that no precise definition emerges.[29]
For an excellent discussion of values in relation to public policy, see generally William W. Lowrance, Modern Science and Human Values (New York: Oxford University Press, 1985).
For the purposes of our discussion, however, value is used to reflect social preferences—preferences communicated by the public or interest groups to decision makers for implementation through the public policy process.For example, the public values product safety. The public policy that manifests safety is government regulation of certain products, including medical devices. How well that value is carried out depends upon the institutional commitment to it (the politics of the FDA), the structure and the jurisdiction of the institution (what the law empowers FDA to do and not do), and the response of the regulated industry. As we begin to think about reform and change, it is imperative not to lose sight of the underlying values the original policies represent. The relevant questions include: Can we achieve those values more efficiently in other ways? Are the values outdated or superseded by newer ones?
Again, for clarity, we must begin to simplify the complicated policy environment. Because we are interested in the impact of these policies on innovation, we can categorize them in relation to that process. Public policies, regardless of source or structure, tend either to promote innovation by accelerating the progress of a new product along the innovation stream or to inhibit the flow of innovation through barriers along the route.
The Matrix
The development of a matrix helps to illustrate the interrelationship between public policy and innovation. In figure 3, the stages of the innovation continuum (from discovery to distribution) are on the horizontal axis. The two identified types of public policy (promote or inhibit) appear on the vertical axis.
Each box in the matrix contains examples of policies that reflect the coordinates. Thus, policies in box 1 promote innovation at the stage of discovery, and policies in box 4 inhibit innovation at the distribution stage. The matrix provides the organizing framework for the discussion in subsequent chapters.
Chapter 2 chronicles the evolution of innovation in the private sector and the preconditions for subsequent policy interventions. In the first half of this century, medical device technology and government institutions were quite independent and engaged in little meaningful interaction. Yet there were signs of change. Technological development was taking place in the private sector, and inventors overcame significant barriers to produce
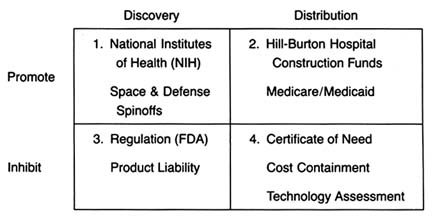
Figure 3. The policy matrix.
a variety of innovations. Public attitudes about the government's role in the innovative process underwent important and perceptible shifts, and public institutions were established that later would play pivotal roles in the device industry. Interaction came later. In terms of both technology development and government policy, World War II provided the transition to the modern environment.
Part II (chapters 3 through 8) follows the chronology of the boxes in the matrix. Policy trends are easily identified, as the numbers reflect the last four decades. In general, the policies of the 1950s (box 1) were dominated by promotion of innovation at the discovery stage, and the 1960s (box 2) saw promotion at the distribution stage, with consequent benefits for both discovery and distribution. The 1970s and 1980s did not completely reverse these trends, in that the policies initiated earlier continued. However, new concerns led to efforts to inhibit innovation. In the 1970s (box 3), significant efforts to increase regulation inhibited discovery; in the 1980s (box 4), concern about cost containment led to policies to inhibit product distribution. The chronology is not exact because some of the regulatory antecedents appeared before the policies were enforced. (The FDA had authority to regulate medical devices as early as 1938; the extension of authority with regulatory teeth, however, came in 1976.)
Subsequent policies did not replace earlier ones. Rather, policies were layered one on top of the other, so that public policy affected innovation at every step. To help us understand the proliferation of policies, each intervention is discussed in relation to the politics of its creation and evolution, the interest groups involved, the goals of the policymakers, and the changes over time.
Each chapter in part II relates to a box in the matrix presented in figure 3. Chapter 3 addresses how government policy promotes discovery (box 1). The NIH is the primary federal organization charged with supporting biomedical research, which has been accomplished through grants to researchers, primarily in universities. Some recent initiatives, most notably the Artificial Heart Program (AHP) at NIH, which is modeled on the experiences of the space program, have targeted specific device technologies. Additionally, chapter 3 looks at government sponsored research in space and defense that has had some interesting effects on medical device technology. Also discussed are recent political efforts to realign the key research institutions—universities, government scientists, and the industry—so that medical technology is transferred from the basic science of the laboratory into the hands of product producers. The chapter evaluates these three initiatives in relation to the medical device industry.
Chapter 4 focuses on policies that promote distribution (box 2). Public policy has played a pivotal role in shaping the size and the composition of the medical device market. Federal and state governments have developed a complex set of policies to pay for health care services. After years of disinterest in health services, federal spending on the growth of the hospital infrastructure began after World War II. Chapter 4 describes how the public role expanded significantly with the enactment of Medicare and Medicaid in the 1960s. The primary health policy goal undergirding public payment is to increase access to health care for those previously excluded, including the elderly, the disabled, and the indigent. Although these programs did not directly address the medical device industry, their impact on that industry was dramatic. Government programs continue to inject billions of dollars into the medical marketplace every year, and
medical technologies are a primary beneficiary. The design of these payment policies dramatically and idiosyncratically affects the size of the market for particular medical technologies.
A few examples illustrate the point. According to some estimates, in 1982 the government paid for over 41 percent of all medical expenditures.[30]
U.S. Department of Commerce, Bureau of the Census, Statistical Abstract of the United States, 1985 (Washington, D.C.: GPO, 1984), table 143.
The payment structure favored hospital based technologies over nonhospital products. Intensive care units, full of new life-support and monitoring equipment, were virtually unknown in 1960; in 1984, they accounted for 8 percent of all hospital beds. Congress extended Medicare coverage for all end-stage renal disease (ESRD) patients in 1972. Kidney dialysis, virtually nonexistent in 1960, was used by 80,000 patients in 1984 at a cost to the government of $1.8 billion.[31]U.S. Congress, Office of Technology Assessment, Federal Policiesand the Medical Devices Industry (Washington, D.C.: GPO, October 1984).
The 1970s brought a new set of concerns to the policy arena—primarily product safety. Chapter 5 explores how the government inhibited discovery of medical devices through safety regulations (box 3). Regulation of medical devices is the primary vehicle for reducing risks of adverse reactions to these products. The federal government and, to a lesser extent, the states have recognized that certain medical products present unacceptable risks and require government intervention through safety and efficacy regulation.
Food and drug regulation dates back to the turn of the century. Congress extended the jurisdiction of the FDA to cover medical devices in 1938; the FDA acquired significantly more extensive regulatory powers under the 1976 Medical Device Amendments to the Federal Food, Drug, and Cosmetic Act, one of the many pieces of consumer protection legislation of the 1970s. The stated goal of these amendments was to "provide for the safety and effectiveness of medical devices intended for human use."[32]
Preamble to Medical Devices Amendment, Public Law 94-295, 90 Stat. 539.
The FDA's jurisdiction is over producers, and its regulations affect firms at the development stage. Cardiac pacemakers and intrauterine devices (IUDs) are used in this chapter to illustrate the impact of regulation on device technology. Because the law focuses on perceived risks associated with medical devices, the riskier the product, the more likely it will encounter the inhibiting forces of the FDA.Chapter 6 continues the discussion of policies that inhibit device discovery (box 3). Although it has antecedents in early
common-law rules, there was an explosion in product liability suits in the 1970s. Product liability seeks to inhibit the manufacture and use of devices if they are determined to be unsafe. However, state courts use completely different tools from FDA regulators to accomplish this substantially similar goal. Liability law in general has a less well-recognized, but clearly related, health mission. Its goal is to compensate individuals injured by defective products and to deter others from producing harmful products. It functions as a part of the health care system in that income to pay for medical costs, as well as noneconomic damage, is transferred from the producers of products to the consumers of products. It is essentially a form of insurance coverage for risk. The law includes both a compensatory and a safety function. The product liability system applies to all consumer products; medical products are included in this broad net. Product liability law, in terms of both the costs and availability of insurance and the consequences of lawsuits, can have a significant, indeed a crippling, impact on some producers.
Chapter 7 analyzes the series of recently imposed mechanisms that inhibit distribution of medical devices (box 4). These policies focus on cost containment rather than on safety, though some seek to control costs through evaluation of product quality. Concern about health care costs in the 1970s and 1980s has affected the momentum of federal and state payment programs. Efforts to restructure the system to control costs have had substantial effects on some segments of the medical device marketplace. Cost-containment strategies began with state based Certificate of Need programs and expanded to a variety of cost-control forms through technology assessment mechanisms. The goal of assessment processes is to ensure that only the "best" technologies are distributed—other technologies should be abandoned. Chapter 7 focuses on federal efforts to institutionalize technology assessment beyond the existing policymaking bodies. In addition, the new payment system under Medicare, known as the Prospective Payment System (PPS), was instituted to control the wildly escalating costs of Medicare. This program has created a new set of idiosyncratic effects on medical device technology.
Chapter 8 introduces the emerging issues of a global marketplace. These issues do not appear within the matrix because it is
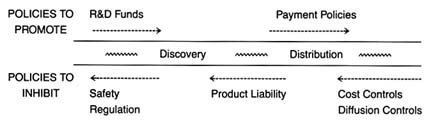
Figure 4. Policies affecting medical device innovation.
unclear whether the international market and the policies of foreign governments regarding medical technology will help domestic medical device producers (that is, promote distribution) or pose a competitive threat to domestic producers (that is, inhibit distribution). Through a brief look at three major markets—Japan, the emerging European Community, and China—these challenges will be assessed.
Part III provides a prognosis. Returning to the innovation continuum, the discussion in chapter 9 falls into three sections (figure 4). The first analyzes the present policy environment at both the discovery and the distribution stages, with illustrations of creative industry strategies that respond to policy incentives. The second section discusses pending policy reforms and their possible effect on the flow of new products. The third section discusses interactions among the various policies that are the inevitable consequences of policy proliferation. Some ways to improve the policy process are discussed.
Chapter 10 looks to the future. The contributions of medical devices to the fight against disease have been critical. However, there are dangers posed by misuse and overuse. The medical arms race must be managed with an understanding of the economic, political, and moral dimensions of medical technology.
The development and application of an analytical framework to the medical device industry only scratches the surface. Further empirical work is necessary to improve our understanding of medical technologies, of the process of innovation, and of the impact of government institutions on the private sector. It is hoped that the issues raised in this book will encourage this research. We must strive for rational policy reform grounded in an understanding of the values and goals of our health care system.
Finally, this study makes clear that policy proliferation is inherent in our political system and reflects the complexity of technology. The system has many benefits, but it also has costs and limitations. It is useful to step back and view the whole landscape to observe the dynamic interactions between the public and the private sectors. In the case of our patient—medical devices—reform may indeed be a matter of life and death.
2
Preconditions for Interaction
When my grandfather George had a stroke he was led into the house and put to bed, and the Red Men sent lodge brothers to sit with him to exercise the curative power of brotherhood…. Ida Rebecca called upon modern technology to help George. From a mail-order house she ordered a battery-operated galvanic device which applied the stimulation of low-voltage electrical current to his paralysed limbs…. In Morrisonville death was a common part of life. It came for the young as relentlessly as it came for the old. To die antiseptically in a hospital was almost unknown. In Morrisonville death still made house calls.
Russell Baker Growing Up
Journalist Russell Baker's description of his grandfather's death is typical of American health care in the 1920s.[1]
Russell Baker, Growing Up (New York: New American Library, 1982), 36-38.
The technological changes that characterized agricultural and industrial production had not yet come to medicine.[2]Stephen Toulmin, "Technological Progress and Social Policy: The Broader Significance of Medical Mishaps," in Mark Siegler et al., eds., Medical Innovation and Bad Outcomes: Legal, Social, and Ethical Responses (Ann Arbor: Health Administration Press, 1987), 22.
Physicians had limited knowledge, and commonly prescribed therapies were improved diets, more exercise, and cleaner environments, not the highly technological interventions customary today.[3]Selma J. Mushkin, Lynn C. Paringer, and Milton M. Chen, "Returns to Biomedical Research, 1900-1975: An Initial Assessment of Impacts on Health Expenditures," in Richard H. Egdahl and Paul M. Gertman, eds., Technology and the Quality of Health Care (Germantown, Md.: Aspen Systems, 1978).
Doctors held out little hope for treatment of most illnesses; consumers like Baker's grandmother often turned to folk remedies or miracle cures. Hospitals were shunned as places where destitute people without family went to die.[4]Charles E. Rosenberg, Caring for Strangers: The Rise of America's Hospital System (New York: Basic Books, 1987). This is a comprehensive study of American hospitals from 1800 to 1920.
Government played a negligible role in health care, and any costs of treatment would havebeen borne by the Baker family, or the treatment foregone if money were not available.
During the first four decades of the twentieth century, innovation in health sciences did occur. Important breakthroughs included improved aseptic surgery techniques, sulfa drugs, and vaccines to treat, and prevent the spread of, infectious diseases. The nascent medical device industry assisted in the advancement of medical science. There were increasingly sophisticated scopes for observation of internal bodily functions and advances in the laboratory equipment that permitted precise measurement of biochemical phenomena. However, quack devices proliferated in addition to legitimate innovations. Many fraudulent devices promising miracle cures capitalized on new discoveries in the fields of electricity and magnetism. The galvanic device purchased to treat Baker's grandfather was typical of popular quack products of the time. Unfortunately for the industry as a whole, these quack devices contributed to the public perception that medical devices were marginal to advanced medical care.
This chapter illustrates how individuals and firms overcame structural and scientific barriers to innovation in the private sector. Various private individuals and corporations managed to bridge these gaps. As a result, the medical device sector enjoyed modest and steady growth.
What is most interesting to the modern reader is the limited role that government played in medical technology innovation. However, seeds of the subsequent multiple roles of government were sown in this early period. By the eve of World War II, several public institutions had been created that would later significantly affect device development. The National Institutes of Health (NIH) would promote medical discovery; the Food and Drug Administration (FDA) would inhibit it. Government involvement in medical device distribution, however, did not occur until somewhat later. World War II accelerated the process of device innovation because of government involvement in the war effort; it both created a demand for medical innovations and overcame the public's reluctance to accept a government role in health policy.
The foundation for subsequent business-government interaction
in medical device innovation had been laid. To use our analogy, the preconditions for later prescriptions to treat our patient were set; full-blown medical intervention awaited the 1950s.
Taking the Patient's History: Industry Overview, 1900–1940
Barriers to Device Innovation in the Private Sector
Throughout the eighteenth and nineteenth centuries there was no continuous scientific tradition in America. Medical science depended upon foreign discoveries, which were dominated by Britain in the early nineteenth century, France at midcentury, and Germany in the last half of the century. The lack of research has been attributed to the dearth of certain conditions and facilities essential to medical studies.[5]
Richard H. Shyrock, American Medical Research, Past and Present (New York: The Commonwealth Fund, 1947).
The absence of dynamic basic science obviously limited the possibility of technological breakthroughs.However, the early part of the twentieth century witnessed a rise in private sector commitment to basic medical science. Philanthropists, intrigued by possibilities of improvements in health sciences, began to endow private research facilities. The first was the Rockefeller Institute, founded in New York City in 1902. A number of other philanthropic foundations were established in the Rockefeller's wake, including the Hooper Institute for Medical Research, the Phipps Institute in Philadelphia, and the Cushing Institute in Cleveland. This period has been called the "Era of Private Support."[6]
Ibid., 99.
These efforts improved the scientific research base in America, but it was still weak.Compounding this weakness were barriers between basic science and medical practice. During the nineteenth century, practicing physicians had little concern for, or interest in, medical research. Most doctors practiced traditional medicine, relying on their small arsenal of tried-and-true remedies. However, in the first decades of the twentieth century, medicine began to change profoundly, which led to organizational permutations through which the medical profession became more scientific
and rigorous.[7]
Paul Starr, The Social Transformation of American Medicine (New York: Basic Books, 1982), 79-145. This comprehensive study of American medical practice is a classic.
University medical schools began to grow as the need for integration of medical studies and basic sciences was acknowledged. Support for medical schools tied research to practice and rewarded doctors for research based medical education. By the end of this period, then, many of the problems associated with lack of basic medical science research had begun to be addressed. However, by modern standards, the commitment to research was extremely small.As our discussion of innovation revealed, basic scientific research must be linked to invention and development. In other words, there must be mechanisms by which technology is transferred from one stage in the innovation continuum to another. In the nineteenth century, there were serious gaps between basic medical science and applied engineering, which is an essential part of medical device development. American engineering education rarely involved research.[8]
Leonard S. Reich, The Making of American Industrial Research: Science and Business at GE and Bell, 1876-1926 (Cambridge: Cambridge University Press, 1985), 24.
Engineers were trained in technical schools but had few university contacts thereafter. Furthermore, manufacturers had little patience for the ivory towers of university science; they looked instead for profits in the marketplace. Engineering practitioners emphasized applying knowledge to the design of technical systems. Thus, engineers based in private companies were cautious about the developments of advanced technology, preferring to make gradual moves in known directions.[9]Ibid., 240.
Producers were slow to take advantage of any advances in research in the United States or abroad. Established industries tended to ignore science and to depend on empirical inventions for new developments.[10]John P. Swann, American Scientists and the Pharmaceutical Industry: Cooperative Research in Twentieth-Century America (Baltimore: Johns Hopkins University Press, 1988).
Patents for medical products did accelerate at the turn of the century, but a majority of them represented engineering shortcuts, not true innovations.[11]Shyrock, American Medical Research, 145.
A few very large corporations addressed this problem through the creation of in-house research laboratories that combined basic science and applied engineering. These institutions focused primarily on incremental product development, but innovative technologies did appear. Thus, while the gap between universities and corporations did not close, some firms became research-oriented through the establishment of independent laboratories. Companies with large laboratories included American
Telephone and Telegraph and General Electric (GE). One major medical device, the X-ray, emerged from GE's research lab. It will be described at greater length shortly.
Further barriers to product development arose from the uneasy relationship between product manufacturers and both university researchers and medical practitioners. A rash of patents were applied for following the development of aseptic surgery. Between 1880 and 1890, the Patent Office granted about 1,200 device patents. The debate that raged over patents of medical products illustrates the tension between health care and profits. Universities generally resisted patenting innovations for several reasons. They valued the free flow of scientific knowledge among scholars. They faced hard questions raised about how to allocate profits of the final result because of the interconnectedness of basic scientific research. They were also concerned that a profit orientation within academia would discourage the practice of sharing scientific knowledge for the betterment of all.[12]
The debate about the appropriate role of academic scientists within universities and about private sector profits continues to this day. For a discussion of the current debate and the relevant public policy on these issues, see chapter 3.
Innovations produced by medical inventors raised additional ethical problems. Often the physician-inventors stood by while commercial organizations exploited remedies based on their work. The medical profession had a traditional ethic against profiting from patents, and the debates about the ethics of patenting medical products continued through this period.[13]
Shyrock, American Medical Research, 143.
For example, the Jefferson County Medical Society of Kentucky stated that it "condemns as unethical the patenting of drugs or medical appliances for profit whether the patent be held by a physician or be transferred by him to some university or research fund, since the result is the same, namely, the deprivation of the needy sick of the benefits of many new medical discoveries through the acts of medical men."[14]Ibid., 122.
Dr. Chevalier Jackson, a noted expert in diseases of the throat, developed many instruments to improve diagnostic and surgical techniques. In his autobiography in 1938, he wrote:
When I became interested in esophagoscopy, direct laryngoscopy, and a little later in bronchoscopy, the metal-working shop at home became a busy place. In it were worked out most of the mechanical problems of foreign body endoscopy. Sometimes the finished instrument was made. At other times only the models were made to demonstrate to the instrument maker the problem and the method
of solution. This had the advantage that the instrument was ever afterward available to all physicians. No instrument that I devised was ever patented. It galled me in early days, when I devised my first bronchoscope, to find that a similar lamp arrangement had been patented by a mechanic for use on a urethroscope, and the mechanic insisted that the use of such an arrangement on a bronchoscope was covered. Caring nothing for humanity, the patentee threatened a lawsuit unless his patent right was recognized during the few remaining years it had left to run.[15]
Chevalier Jackson, The Life of Chevalier Jackson: An Autobiography (New York: Macmillan, 1938), 197.
Not all physicians and scientists shared these views, but there was clearly a conflict between the perception of medical technology for the public good and the pursuit of profits, one that created a possible barrier to a full exploitation of the marketplace.
The last barrier to innovation in the medical field was the absence of a sizable market for the available products. In 1940, on the eve of World War II, total spending on health care in the United States amounted to $3.987 billion, or $29.62 per person per year. Health care spending accounted for only 4 percent of GNP. Patients paid directly for about 85 percent of costs. A small number of people received public assistance in city or county hospitals; there was virtually no private insurance coverage.[16]
Department of Commerce, Bureau of the Census, Historical Statistics of the United States, Colonial Times to 1970, bicentennial edition, pt. 2, ser. 221, 247 (Washington, D.C.: GPO, 1975).
There was no buffer for individuals and families in hard times, and health care was often sacrificed when family income was limited.The market for medical technology remained small by modern standards. The number of hospital beds was low in comparison to present population-to-bed ratios. Doctors' offices had little technological equipment. Fortunately, the small market did not deter researchers in some cases. For example, a 1914 study by engineers at General Electric concluded that new X-ray tubes would be more expensive than earlier ones and that the market would be too small to justify manufacture. Nonetheless, the management insisted that production proceed, noting that "the tube should be exploited in such a way as to confer a public benefit, feeling that it is a device which is useful to humanity and that we cannot afford to take an arbitrary or even perhaps any ordinary commercial position with regard to it."[17]
Reich, American Industrial Research, 89, citing George Wise, The Corporations' Chemist (Unpublished manuscript, 1981), 237.
Because of this altruistic view, General Electric soon dominated
the X-ray market and later turned a tidy profit on the enterprise. Obviously, however, most producers and inventors could not afford to ignore "ordinary commercial" considerations.
Bridging the Gaps
Innovation did occur despite the severe limitations in the private sector. Several examples of device innovation illustrate how these limitations were overcome.
Individual Initiative
One way to overcome institutional barriers to technology transfer was for an individual to fulfill several essential roles. Arnold Beckman, the founder of Beckman Instruments, was a scientist, engineer, and entrepreneur and thus had expertise in all the necessary stages of innovation.[18]
Most of the information on Arnold Beckman and the founding of his company appears in Harrison Stephens, Golden Past, Golden Future: The First Fifty Years of Beckman Instruments, Inc. (Claremont, Calif.: Claremont University Center, 1985).
As an assistant professor of chemistry at the California Institute of Technology in the early 1930s, he was called upon to solve technical problems that involved chemistry and then engaged in entrepreneurial activities with the results. His first business venture was to create a special ink formula that would not clog the inking mechanism in a postal meter. Because existing companies would not make the special formula, Beckman founded the National Inking Appliance Company in 1934.Beckman soon undertook additional projects with applications for science and medicine. At the request of agricultural interests, he invented a meter to measure the acidity of lemon juice that had been heavily dosed with sulfur dioxide. Using electronic skills he had developed at Bell Labs as a graduate student, Beckman designed the first acidimeter to measure the acidity or alkalinity of any solution containing water. He formed Beckman Instruments in 1935 to produce these acidimeter and discovered a market among some of his former professors at a meeting of the American Chemical Society.
In recalling the founding of his company, Beckman noted, "We were lucky because we came into the market at just the time that acidity was getting to be recognized as a very important
variable to be controlled, whether it be in body chemistry or food production."[19]
Stephens, Golden Past, 14.
In 1939 he quit teaching to run the business fulltime. Beckman's substantial contribution to the war effort, and the benefits the firm reaped as a consequence, will be discussed in a subsequent section. His company later became one of the major medical technology firms in America.[20]Much of the history of Arnold Beckman's contributions were summarized in Carol Moberg, ed., The Beckman Symposium on Biomedical Instrumentation (New York: Rockefeller University, 1986). This volume celebrates the fiftieth anniversary of the founding of Beckman Instruments.
Beckman's career illustrates how a multifaceted individual, with scientific, technical, and marketing skills, could successfully produce scientific instruments despite the significant barriers to innovation in the early twentieth century.Industrial Research Laboratories
Industrial research laboratories also served as bridges between science and technology and the market. Generally set apart from production facilities, these laboratories were staffed by people trained in science and advanced engineering and who were working toward an understanding of corporate related science and technology.[21]
Reich, American Industrial Research, 3.
By setting up labs, large firms could conduct scientific research internally and profit from the discoveries. These laboratories were possible only after the period of consolidations and mergers in the late 1890s and early 1900s that led to the growth of industrial giants that had sufficient resources to fund them. Research was vital, and product development formed a large part of their competitive strategy. These large firms were well aware of scientific developments in Europe, particularly in the fields of electrochemistry, X-rays, and radioactivity, and they knew they had to stay abreast of technological change.[22]Ibid., 37.
General Electric Company, founded in 1892, created a research laboratory that was firmly established within the company by 1910. William Coolidge led research efforts on the X-ray tube, which had been discovered in Germany in the 1890s. It was widely known that X-rays had medical value in that they allowed doctors to observe bone and tissue structure without surgery. Two-element, partially evacuated tubes generated the X-rays. As they operated, the tubes produced more gas, changing the pressure and making them erratic. Coolidge substituted tungsten for platinum. It could be heated to a greater temperature without
melting, while emitting less gas than platinum to achieve higher-powered and longer-lived tubes.
By 1913, the laboratory was manufacturing Coolidge's X-ray tubes on a small scale, selling 300 that year and 6,500 in 1914.[23]
Kendall Birr, Pioneering in Industrial Research: The Story of the General Electric Research Laboratory (Washington, D.C.: Public Affairs Press, 1957).
When the government began to place large orders of tubes for portable X-ray units for military hospitals during World War I, GE began to make significant profits. After the war, GE's management decided that the company would become a full-line X-ray supplier. GE bought the Victor X-ray company, switched its production of Coolidge tubes to Victor, and soon held a dominant position in the new and increasingly profitable medical equipment supply business.[24]The government's role in World War I represents the beginning of a transition to government involvement in medical device innovation. In this case, the government's demand for X-ray equipment as a purchaser significantly benefited the firm.
Problems of Medical Quackery
Along with these exciting breakthroughs in medical technology, a very different side of the industry flourished. The popularity of fraudulent devices, and concern about the consequences of that fraud, tainted public perception of the industry. Many people associated medical devices with quack products.[25]
The term quack dates to the sixteenth century and is an abbreviation for quacksalver. The term refers to a charlatan who brags or "quacks" about the curative or "salving" powers of the product without knowing anything about medical care. From the Washington Post, 8 July 1985, 6.
When medical science offers no hope of treatment or cure, people have traditionally turned to charms and fetishes for help.[26]
Warren E. Schaller and Charles R. Carroll, Health Quackery and the Consumer (Philadelphia: W. B. Saunders, 1976), 228. This volume contains many descriptions of a variety of fraudulent devices. It makes for amusing reading, but deceptive activities left a lasting legacy on the medical device industry.
The desperate search for cures was a major factor in the success of all forms of health fraud, including tonics, drugs, potions, and quack devices. Indeed, it is estimated that the public spent $80 million in 1906 on patent medicines of all kinds. The proliferation of quack devices helped to generate subsequent government intervention to protect the public.Innovative developments in scientific fields, such as electromagnetism and electricity, were often applied to lend credence to these health frauds (see figure 5). At the turn of the century, Dr. Hercules Sanché developed his Electropoise machine to aid in the "spontaneous cure of disease."[27]
James H. Young, Medical Messiahs (Princeton: Princeton University Press, 1967), 243.
This device, consisting of a sealed metal cylinder attached to an uninsulated flexible cord that, in turn, attached to the wrist or ankle, supplied, according to the inventor, "the needed amount of electric force to the system and by its thermal action places the body in a condition to absorb oxygen through the lungs and pores."[28]Ibid.
The Oxydonor was a subsequent "improvement" that added a stick of
Figure 5. A quack device.
Source: The Bakken Archives.
carbon to the sealed, and incidentally hollow and empty, cylinder and "cured all forms of disease." The success of the Oxydonor spawned a whole cadre of imitations. There was a death knell for these "pipe and wire" therapies when the inventor of the Oxypathor was convicted of mail fraud. Evidence collected at the trial revealed that the company had sold 45,451 Oxypathors at $35 each in 1914. Considering that as late as 1940 per capita spending on health was only $29.62, the consequences of wasted expenditures seem serious.
Another fraud was based on the new field of radio communications. Dr. Albert Abrams of San Francisco introduced his "Radionics" system, which was based on the pseudomedical theory that electrons are the basic biological unit and all disease stems from a "disharmony of electronic oscillation." Dr. Abrams's diagnoses were made by placing dried blood specimens into the Radioscope. Operated with the patient facing west in a dim light, the device purported not only to diagnose illness but also to tell one's religious preference, sex, and race. At the height of its use, over 3,500 practitioners rented the device from Abrams for $200 down and $5 a month.[29]
Schaller and Carroll, Health Quackery, 226.
One of Abrams's followers was Ruth Drown. She marketed the Drown Radio Therapeutic Instrument, which she claimed could prevent and cure cancer, cirrhosis, heart trouble, back pain, abscesses, and constipation. The device used a drop of blood from a patient, through which Drown claimed she could "tune in" on diseased organs and restore them to health. With two drops she could treat any patient by remote control. A larger version of her instrument was claimed to diagnose as well as to treat disease. Thousands of Californians patronized her establishment.[30]
Drown v. U.S., 198 F.2d 999 (1952). Drown was prosecuted for grand larceny and died while awaiting trial. See Joseph Cramp, ed., Nostrums and Quackery and Pseudo-Medicine, vols. 1-3 (Chicago: Press of the American Medical Association).
Unfortunately for legitimate medical device producers, the prevalence of device quackery tainted the public's perception of the industry. Indeed, initial state and federal efforts to regulate the industry were in response to problems of fraud. Device fraud continues, especially for diseases like arthritis and cancer and for weight control.[31]
Concern about fraudulent drugs and devices surfaces periodically. Congress has held hearings investigating health frauds, particularly frauds against the elderly. See, for example, House Select Committee on Aging, Frauds Against the Elderly: Health Quackery, 96th Cong., 2d sess., no. 96-251 (Washington, D.C.: GPO, 1980). In 1984, the FDA devoted only about one-half of 1 percent of its budget to fighting quack products. Under pressure from the outside, it set up a fraud branch in 1985 to process enforcement actions. See Don Colburn, "Quackery: Medical Fraud Is Proliferating and the FDA Can't Seem to Stop It," Washington Post National Weekly Edition, 8 July 1985, 6. For a description of past and present device quackery, see Stephen Barrett and Gilda Knight, eds., The Health Robbers: How to Protect Your Money and Your Life (Philadelphia: George F. Stickley, 1976).
Fraud in the industry can be seen as a market failure that subsequent government prescriptions sought to correct.Industry Status in 1940
Despite the successes of some firms and some individuals, the medical device industry remained relatively small even as late as 1937. The Census of Manufactures, the best source of information about producers, categorized all industrial production into Standard Industrial Codes. Before 1937, the Census contained only one primary industrial category for medical products. This was SIC 3842—surgical appliances and supplies—which included items such as bandages, surgical gauze and dressings, first aid kits, and surgical and orthopedic appliances. Both the number of producers and the value of their sales in SIC 3842 were quite small until the 1930s. For example, in 1914 there were 391 establishments that shipped products valued at about $16.5 million. The number of producers rose to 445 during World War I, dropped to below 300 until the midtwenties, fell further to 250 during the depression, and reached 323 in 1937. Sales reflected the same ebb and flow, dropping from $71 million in 1929 to $51 million in 1933, and recovering to $77 million by 1937.
In the late 1920s a second classification—SIC 3843—was added for dental products and supplies. In the 1937 data, three important classifications emerged. SIC 3693 included X-ray and therapeutic apparatus. Sales of these products had occurred before this time; however, they were buried in other, nonmedical industrial categories. The industries that produced these products engaged primarily in manufacturing X-ray tubes and lamps for ultraviolet radiation. By the 1950s, the name of the code changed to X-ray, electromedical, and electrotherapeutic apparatus, reflecting the growing innovation in this field. By 1937, SIC 3693 accounted for $17 million in sales with forty-six companies producing equipment. A second new code was SIC 3841—surgical and medical instruments—which included products such as surgical knives and blades, hypodermic syringes, and diagnostic apparatus such as ophthalmoscopes. In 1937, thirty-nine companies were producing devices in this code. In that year, records began to be kept for a fifth medical product category, SIC 3851, ophthalmic goods, which included eyeglass frames, lenses,
and industrial goggles and included seventy-nine companies selling about $43 million worth of goods in 1937. In all five relevant SIC codes, there were only 588 establishments with total product shipments valued at $200 million.[32]
In addition to the extensive data gathered by the Census, see R. D. Peterson and C. R. MacPhee, Economic Organization in Medical Equipment and Supply (Lexington, Mass.: D. C. Heath, 1973).
As we have seen, there was some innovation and modest, though inconsistent, growth in the medical device industry from 1900 to 1940. New products emerged despite barriers to technology transfer at the various stages of the innovation continuum. However, innovation was frequently serendipitous, dependent on individuals and not institutionalized sufficiently to ensure a steady stream of progress. Creative entrepreneurs and industrial laboratories did play a role in overcoming obstacles for some technologies. On the other hand, the device industry was plagued with quackery and fraud.
Evolution of Public Institutions, 1900–1940
With the exception of government procurement during World War I, the device industry grew with minimal government involvement. However, intervention was beginning during this period.
There were perceptible shifts in the public's attitude about the appropriate role of the federal government in economic and social issues. Early institutions and policies emerged that would both promote and inhibit medical innovation. How these policies evolved would influence, to a large extent, the contours of the emerging medical device industry.
Government Promotes Discovery
The general aversion toward government involvement in basic research gave way to acceptance of a public role—a change reflected in the creation of the National Institutes of Health (NIH). At the turn of the century, medical practitioners vehemently rejected any government intervention in their domain. In 1900, a well-known physician appeared before a Senate committee and declared that all the medical profession asked of Congress was no interference with its progress.[33]
Shyrock, American Medical Research, 77
Even as late as 1928, Atherton Seidell, a famous chemist, stated, "We [Americans]naturally question governmental participation in scientific matters because we feel that anything having a political flavor cannot be above suspicion."[34]
Quoted in Harden, Inventing the NIH, 3.
But, in the same year, scientist Charles Holmes Herty said, in regard to support of basic science, "I have changed my mind completely, and I feel that the Government should lead in this matter."[35]Ibid., 92.
Early government activities were primarily in the area of public health, not research. The roots of government involvement lie in the federal Hygienic Laboratory, founded in 1902 to implement the Biologics Control Act, one of the earliest pieces of U.S. health legislation. Under this act, the Hygienic Lab inspected vaccine laboratories, tested the purity of the products and issued licenses.[36]
Ibid., 28.
In addition, the Public Health Service was established in 1912 to address applied health issues. Neither institution had real involvement in basic sciences until World War I. On the eve of the war, the relationship between science in the private sector and science in government was in a state of equilibrium, with the private sector supporting basic research as well as some applied work and the federal government devoting its limited revenues to practical scientific work through the Hygienic Lab and the Public Health Service.[37]Ibid., 93.
The war made the government conscious of the value of science, particularly as German superiority in chemical warfare became apparent. Thus, after the war, there was more talk of public and private sector cooperation to promote science. The proposals put forward in this period, however, advocated the control and direction of research under private auspices with very limited government participation. President Coolidge did not believe in marked expansion of the governmental initiatives to promote health; he viewed medicine and science as the provinces of the private sector. In 1928, Coolidge vetoed a bill to create a National Institute of Health that would provide fellowships for research funded by private donations.[38]
Ibid., 127.
He stated, "I do not believe that permanency of appointment of those engaged in the professional and scientific activities of the Government is necessary for progress or accomplishment in those activities or in keeping with public policy."[39]Ibid., 132.
However, the private sector was not willing to come forward single-handed with substantial funds to support this undertaking. One historiannoted that this lack "was to make the concept of government funding less noxious as the years passed and the original hope dimmed"[40]
Ibid., 91.
With the private sector sufficiently unresponsive, a law establishing the National Institute of Health finally passed in 1930. However, NIH expansion into a large-scale facility was nearly twenty years in the future. The NIH remained small but not totally inactive. Although Congress refused to appropriate the maximum amount during the 1930s, the NIH did receive increased monies to expand research into chronic diseases. In 1937, Congress authorized the National Cancer Institute (NCI) with legislation that sped through Congress in record time and that was unanimously supported by the Senate.[41]
For a thorough discussion of the politics of the NIH's role in cancer research, see Richard A. Rettig, A Cancer Crusade: The Story of the National Cancer Act of 1971 (Princeton: Princeton University Press, 1977).
However, most of the NIH's accomplishments up to World War II were responses to public health emergencies. As late as 1935, the president's Science Advisory Board concluded that no comprehensive, centrally controlled research program was desirable except for certain problems related to public health. By 1937, total federal research expenses, spread out among many federal agencies, amounted to only $124 million, and much of that was allocated to natural sciences and nonmedical technology.[42]
Shyrock, American Medical Research, 78-79.
Despite limited funding, the NIH represented a significant change in attitude toward the role of government. During this period, the NIH research orientation was set as well. Much of its early work continued to be in the science of public health, and research support could have moved in a more applied direction. However, the NIH preferred to expand into basic research, primarily in conjunction with universities, and avoided support of applied research. In chapter 3, we shall see how the political commitment to the NIH and its research orientation affected medical device innovation.
Government Inhibits Discovery
Government promotion of medical discovery emerged alongside institutions that would later inhibit discovery. This inhibiting role for government arose in response to problems of fraud and quackery.
Fraud in food and medicine sales was nothing new, but during the 1880s and 1890s concern grew about dangerous foodstuffs in the market. Attention focused on the sale of diseased meats and milk, on adulterated food products such as a combination of inert matter, ground pepper, glucose, hayseed, and flavoring that was sold as raspberry jam. Many nostrums contained dangerous, habit-forming narcotics sold to an uninformed and unsuspecting public.[43]
Oscar E. Anderson, "Pioneer Statute: The Pure Food and Drug Act of 1906," Journal of Public Law 13 (1964): 189-196. See also Oscar E. Anderson, The Health of a Nation: Harvey W. Wiley and the Fight for Pure Food (Chicago: University of Chicago Press, 1958).
States did pass laws to regulate the marketing of harmful products, but these laws were relatively ineffectual because a state could not enforce its regulations against an out-of-state manufacturer. The only recourse was to reach producers indirectly through the local retailers who handled the goods. At the federal level, Congress was not inactive; from 1879, when the House introduced the first bill designed to prevent the adulteration of food, to June 30, 1906, when the Pure Food Law was signed, 190 measures relating to problems with specific foods had been introduced.[44]
Thomas A. Bailey, "Congressional Opposition to the Pure Food Legislation, 1879-1906," American Journal of Sociology 36 (July 1930): 52-64.
A number of important interests marshalled considerable opposition to federal intervention. States' rights Democrats from the South believed that the federal government did not have the constitutional authority to intervene in the private sector.[45]
Ibid.
Some food producers and retailers contended that they could not sell their goods if they had to label all the ingredients. The drug industry joined the fray when a Senate bill extended the definition of drugs to include not only medicines recognized by the United States Pharmacopeia,[46]Since 1820, the United States Pharmacopeia Convention (USPC) has set standards for medications used by the American public. It is an independent, nonprofit corporation composed of delegates from colleges of medicine and pharmacy, state medical associations, and other national associations concerned with medicine. When Congress passed the first major drug safety law in 1906, the standards recognized in the statute were those of the USPC. Its major publication, the United States Pharmacopeia (USP), is the world's oldest regularly revised national compendium. Today it continues to be the official compendia for standards for drugs.
but also any substance intended for the cure, mitigation, or treatment of disease. This definition would bring the proprietary medicines within the scope of the law.[47]Proprietary drugs are those drugs sold directly to the public, and they include patent medicines. The term proprietary indicates that the ingredients are secret, not that they are patented.
The Proprietary Association of America, with trade organizations of wholesale and retail druggists, immediately joined the opposition.Commercial interests could not suppress public opinion after disclosures of adulterated foods in the muckraking press stirred up the progressive fervor. The 1905 publication of Upton Sinclair's The Jungle, which contained graphic images of adulterated food, including a contention that some lard was made out of the bodies of workmen who had fallen into cooking vats, provoked reform. Harvey W. Wiley, chief of the Division of Chemistry in
the Department of Agriculture, became a missionary for reform. He organized the "poison squad" in 1902 and found that the volunteers who restricted their food intake to diets that included a variety of food additives, such as boric acid, benzoate of soda, and formaldehyde, suffered metabolic, digestive, and health problems.[48]
Temin, Taking Your Medicine, 28.
The 1906 Pure Food Act was a compromise among many diverse and competing interests. The act was very different in orientation from the subsequent drug regulation; it was intended to aid consumers in the marketplace, not to restrict access of products to the market. The act, in short, made misrepresentation illegal. A drug was deemed adulterated if it deviated from the standards of the national formularies without so admitting on the label. A drug was considered misbranded if the company sold it under a false name or in the package of another drug or if it failed to identify the presence of designated addictive substances. The authority of the federal government was limited to seizure of the adulterated or misbranded articles and prosecution of the manufacturer.[49]
Pure Food Act, 34 Stat. 674 (1906).
It is important to remember that this was not an effort by the federal government to intervene in medical care decisions. Because there were few effective drugs in 1906 and most were purchased by consumers without the aid of physicians, drug regulation was seen as a part of food regulation, not of health care per se. Inclusion of medical devices—products used for health care but not consumed as food or drugs—does not appear to have been considered during the decades in which Congress debated food and drug legislation.
Nevertheless, the 1906 law initiated the subsequent growth of a federal role in consumer protection. The Department of Agriculture's Division of Chemistry administered the law. It was renamed the Bureau of Chemistry in 1901; its appropriation rose by a factor of five, and the number of employees grew from 110 in 1906 to 425 in 1908.[50]
A. Hunter Dupree, Science in the Federal Government: A History of Policies and Activities to 1940 (Cambridge: Harvard University Press, 1957), 179.
The Agriculture Appropriations Act of 1931 established the Food and Drug Administration (FDA) within the department.[51]Bruce C. Davidson, "Preventive 'Medicine' for Medical Devices: Further Regulation Required?" Marquette Law Review 55 (Fall 1972): 408-455.
It had a budget of $1.6 million and over 500 employees at the time of its name change.Although medical devices were overlooked from 1906 to 1931, several important steps had been taken. First, there was
acceptance of the federal government's role in protecting the public from adulterated and misbranded products. There was also a federal institution in place, the FDA, with expertise in regulation. As problems arose subsequently in other product areas, it was easy to expand the scope of the existing institution to cover medical devices.
Expansion came during the 1930s. W. G. Campbell, the chief of the FDA, and Rexford G. Tugwell, the newly appointed assistant secretary of agriculture, decided to rewrite the legislation in the spring of 1933.[52]
Temin, Taking Your Medicine, 38.
A 1933 report by the FDA first raised the problems related to medical devices.Mechanical devices, represented as helpful in the cure of disease, may be harmful. Many of them serve a useful and definite purpose. The weak and ailing furnish a fertile field, however, for mechanical devices represented as potent in the treatment of many conditions for which there is no effective mechanical cure. The need for legal control of devices of this type is self-evident. Products and devices intended to effect changes in the physical structure of the body not necessarily associated with disease are extremely prevalent and, in some instances, capable of extreme harm. They are at this time almost wholly beyond the control of any Federal statute…. The new statute, if enacted, will bring such products under the jurisdiction of the law.[53]
U.S. Department of Agriculture, Report of the Chief of the Food and Drug Administration (Washington, D.C.: GPO, 1933), 13-14.
An interesting evolution in the FDA's orientation from its earlier conception had begun. The FDA moved from the aegis of agriculture, where food was a primary focus, to the Department of Health, Education, and Welfare (HEW), which was concerned with broader issues of health. This shift made device regulation a logical extension of FDA jurisdiction.
Problems of definition arose, however, in the debates over terminology. In early drafts of the 1906 law, drugs were defined more broadly. There was an effort to capture within the regulatory definition both those products used for the diagnosis of disease and products that were clearly fraudulent, such as antifat and reducing potions that did not purport to treat recognized diseases. Some legislators proposed to include therapeutic devices within the definition of drugs. In Senate hearings on the bill, the FDA chief stated that adding medical devices to the
definition of drugs was intended to extend the scope of the law to include not only products like sutures and surgical dressings but also "trusses or any other mechanical appliance that might be employed for the treatment of disease or intended for the cure or prevention of disease."[54]
Davidson, "Preventive 'Medicine,'" 414.
The legislative debates on the definition are enlightening, not for the quality of the debates per se but for their focus. Indeed, the discussion of the device/drug distinction emerged in a confused context. While Senator Copeland, a major supporter of the bill, spoke to an amendment of the drug definition to include drugs used for "diagnosis" of disease as well as for "cure, mitigation, or treatment," his colleague, Senator Clark, objected to the use of the term drug to describe a device. Senator Clark stated that he did not oppose devices being covered by the law, but to treat them as drugs "in law and in logic and in lexicography is a palpable absurdity."[55]
Ibid., 415.
Because he was raising an issue not relevant to the amendment under debate, the matter was not resolved at that time. However, Senator Copeland had no fundamental objection to separate definitions, and the later versions included a separate paragraph defining devices.Subsequent events made this a very significant distinction indeed. The first bill was introduced in Congress in 1933 and did not become law in the subsequent five-year period. The bill finally got attention following a drug disaster. The Massengill Company, a drug firm, wanted to sell a liquid form of sulfanilamide, one of the new classes of sulfa drugs on the market in tablet form. The company dissolved the drug in a chemical solution and marketed it as Elixir Sulfanilamide in September 1937. The chemical solution, diethylene glycol, was toxic; over one hundred people died from the elixir. The FDA exercised its power to seize as much of the preparation as it could find and managed to retrieve most of it. However, the agency did not have the power to prosecute Massengill for causing the deaths; it could only cite them for failing to label the solution properly. The fine imposed for mislabeling was $26,100.[56]
Temin, Taking Your Medicine, 42.
Public pressure for greater FDA authority arose after this incident. The pending legislation expanded the FDA's power to screen all "new drugs" before they could be marketed. This distinction created a new class of drugs quite separate from
medical devices. The concept of premarket control provided more protection for the consumer than prior labeling and information requirements.
However, this extension of power to regulate "new drugs" had an additional effect on medical devices. They were now included in the final version of the law, but because they were defined separately, the new drug provision did not apply to them. Device producers were subject only to the adulteration and labeling requirements under the law. The emphasis of the lawmakers continued to be on fraudulent devices, not on control of legitimate devices that might have both therapeutic benefit and harmful characteristics.
The FDA's procedural powers over devices were limited to seizure of the misbranded product and prosecution of the producer. It could not initiate regulatory action until a device had entered interstate commerce and then only if it deemed the product improperly labeled ("misbranded") or dangerous ("adulterated").[57]
21 U.S.C. sec. 351-352.
Once the FDA considered a product misbranded or adulterated, it could initiate a seizure action and seek to enjoin the manufacturer from further production of the device.Seizure was the tactic most frequently employed against device producers in subsequent years. The agency had to file a libel action in a district court, alleging a device to be in violation of the law. The FDA seized the devices before trial. The seizure was upheld only if the FDA could prove its charges at trial. The proceeding affected the specific device seized. Only after the agency succeeded in the initial action could it make multiple seizures or move the court to enjoin further production. Of course, multiple seizures were impractical because it was extremely difficult to trace the ultimate location of the condemned devices and virtually impossible to seize them all. Device manufacturers could evade injunction by making insignificant changes in their products and marketing them as "new devices."[58]
See Comptroller General of the United States, Lack of Authority Limits Consumer Protection: Problems in Identifying and Removing from the Market Products Which Violate the Law, B-164031(2) at 18-25 (1972).
The number of trials for fraudulent devices remained small and affected only quack producers at the margins of the industry.When the federal government addressed problems with medical devices, its powers were limited to procedures which had been inadequate for the regulation of harmful drugs. Despite these problems, however, the 1938 law did set an important
precedent. A large, popular federal institution existed to protect the public from harmful products used for health purposes. Devices were generally ignored for nearly forty years, while federal power over the drug marketplace expanded from 1938 to 1962. Not until 1976 did the FDA get jurisdiction to regulate some devices as stringently as new drugs.
Government and Device Distribution
As we have seen, government institutions, primarily the NIH and the FDA, were in place to intervene in the discovery phase of the medical device innovation process. However, the entry of government into the distribution phase lagged behind these developments.
At the beginning of the twentieth century, not much health care was available for the consumer to buy. As we have seen, doctors had a very limited arsenal of treatment options. There were few drugs and fewer medical devices. There was only a primitive understanding of the biological processes of the human body and little that a doctor could do to alleviate illness. Hospitals were used only by the "deserving" poor, who could not be cared for at home but were not candidates for the almshouse. The dismal conditions in hospitals between 1870 and 1920 have been exhaustively documented.[59]
See Rosenberg, Caring for Strangers.
The public sector played only a minor role in support of hospital care. Local governments often supported public hospitals, and some private institutions received local aid. There were no other sources of public funds for hospitals. Although suffering financial woes, a New York City hospital in 1904 would not seek federal support, "No one, in those days, proposed going hat in hand to Albany or Washington."[60]
Starr, Transformation, 148-180.
There were no intermediaries in the costs of sickness for anyone who was not destitute. In the first decades of the twentieth century, Europe began to provide state aid for sickness insurance, but there was no government action to subsidize voluntary expenditures in the United States.[61]
Ibid., 237. Starr attributes European activity to political instability not present in the United States. The American government remained very decentralized, and there was not the political instability that Europe encountered.
There were some progressive reformers that advocated health insurance, both private and public, as early as 1912. However, employers strongly opposed insurance schemes, as did the physicians and labor unions. Themedical profession vigorously decried a health insurance referendum in California, alleging that the proposal was linked to sinister forces in Germany.[62]
Ibid., 245-253.
However, important developments in diagnosis and treatment began to change the attitude of the public toward the desirability of health care. The advent of aseptic surgery and the X-ray machine gave the ill some hope that surgery could cure them. The number of hospitals grew, from 4,000 in 1909 to 7,000 in 1928, and the number of hospital beds expanded from 400,000 to 900,000 as a result. Costs of care began to rise as well. These costs were associated with some new capital equipment, including European innovations for diagnosis and the growth of laboratories.
Unfortunately, the depression in the 1930s meant declines in visits to doctors and hospitals and a consequent drop in the incomes of physicians. Hospitals suffered financially, and at the same time demand increased for free services. Some public welfare payments for medical care were seen as a temporary solution; these continued after the depression.
Important changes were occurring in the market for medical care, but they did not extend to acceptance of federal and state involvement in that market. People had begun to see the value of health care. The middle class no longer was satisfied with attention from the family physician, and the hospital offered important services not available at home. At the same time medical science held out hope, however, the depression made health care inaccessible to large numbers of Americans. There was a perceived need for more health care, but few resources were available to pay for it. Pressure for greater access, particularly by the middle class, would ultimately bring the government into the marketplace. However, the idea of government subsidies met with considerable resistance, and health care took a back seat to other pressing social needs, such as public assistance and welfare.
As America emerged from the Great Depression and approached World War II, significant changes had occurred in the relationship between government and innovation. The NIH was established to promote basic medical science research, which indirectly affected the medical device industry. The federal government
also had made significant steps toward direct regulation of product producers in the 1938 Food, Drug, and Cosmetic Act. The government had not, as yet, committed itself to payment for health care. This development lay ahead.
World War II Accelerates Interaction
As the nation mobilized for war in the 1940s, the federal government became involved in many activities previously left to the private sector. Government leadership in the war effort changed the public perception about its basic role in science and medicine.
Wartime Innovation
The federal government had an effect on all stages of the innovation process in medical devices during the war. Government spending promoted basic science as well as technological invention and development. Government also became a major consumer of both medical technology and military technology, greatly expanding the market for products produced by firms with medical technology expertise.
President Roosevelt established the Office of Scientific Research and Development (OSRD) in 1941, and it had two parallel committees on national defense and medical research. The Committee on Medical Research (CMR) mounted a comprehensive program to address medical problems associated with the war. The government gave 450 contracts to universities and 150 more to research institutes, hospitals, and other organizations. In total, the office spent $15 million and involved some 5,500 scientists and technicians. Government supported achievements included a synthetic atabrine for malaria treatment (which replaced the quinine seized by Japan), therapeutically useful derivatives of blood, and the development of penicillin.[63]
Ibid., 340.
The OSRD was unique because it was organized as an independent civilian enterprise and managed by academic and industrial scientists in equal partnership with the military. In contrast with World War I, where scientists served as military officers under military commanders, the work of OSRD was fully
funded by the government, but scientists worked in their own institutional settings. The research contract model proved to be a flexible instrument in the subsequent partnership between government and private institutions during the postwar period.[64]
Harvey Brooks, "National Science Policy and Technological Innovation," in Ralph Landau and Nathan Rosenberg, eds., The Positive Sum Strategy: Harnessing Technology for Economic Growth (Washington, D.C.: National Academy Press, 1986), 119-167, 123.
Government also let contracts for development of wartime technologies. Some of these efforts benefited device companies directly because they had technologies that could be channeled for military use. Other government efforts promoted technologies that would later prove useful in medical device technology. In addition, the government was a ready market for military and medical supplies. Government purchasing enriched many companies in the instrument business, such as Beckman and GE. Government policy helped to establish a technology base for postwar development and allowed firms to take advantage of the postwar boom.
In addition, federal government spending and greater need for health care for service personnel injured in combat stimulated the medical technology market. The federal government provided medical services for all military personnel—60 percent of all hospital beds were used by the military. Thus government also became a large consumer of medical supplies and equipment.
Medical Device Successes in Wartime
The war provided an impetus to innovation in medical device technology. Three profiles of successful firms—Beckman Instruments, Baxter Travenol, and General Electric—illustrate the effect of government on innovation.
Beckman
Beckman Instruments provides an excellent example of the impact of the war on medical device technology. The National Technical Laboratories, as the firm was called at the time, did not make weapons but did make important military products. Its contribution to the war effort is reflected in sales data: gross sales were thirty-four times larger in 1950 than in 1940.
One key product was Beckman's "Helipot," a unique instrument
for use in radar systems. The U.S. military requested meters built to military specifications for the radar program and able to withstand strong mechanical shocks. Beckman recalled, "I began to get calls from lieutenants and captains and finally from generals and admirals. There were ships that couldn't sail because they didn't have Helipots for their radars."[65]
Stephens, Golden Past, 34.
Beckman himself redesigned the instrument. In the first year of production, the new model accounted for 40 percent of the firm's total profits.Because wartime disrupted supplies of essential products, new markets opened and creativity was welcomed and rewarded. Beckman's spectrophotometer, which used a quartz prism and a newly developed light source and phototube, is a good example. This model, introduced in 1941, could accurately measure the vitamin content of a substance. The war had cut off the supply of cod liver oil, which was a rich source of vitamins A and D, from Scandinavia. Before the Beckman instrument, there was no way to efficiently measure the vitamin content of other foods to plan healthy diets. The Beckman spectrophotometer determined vitamin content precisely in one or two minutes.
Rubber supplies had been cut off by the bombing of Pearl Harbor, and the nation desperately needed a substitute. Supported by the federal Office of Rubber Reserve, Beckman developed infrared spectrophotometers that could detect butadiene, a major ingredient in synthetic rubber. Later on, Beckman was also involved in a government project with the Massachusetts Institute of Technology, working under the Atomic Energy Commission, to develop a recording instrument to monitor radioactivity levels in atomic energy plants.
These new technologies frequently proved to have medical applications. With friends from the California Institute of Technology, Beckman produced oxygen meters for the navy. An anesthesiologist heard about the meter in its development stage and was interested in its use to measure oxygen in infant incubators. If oxygen supplies to a baby are too low, the infant will not thrive; if oxygen levels are too high, it can become blind. This doctor treated his own grandchild with a Beckman meter, feeding oxygen from a tank into a cardboard box and thereby saving the baby's life. However, during the war, hospitals could not
afford oxygen meters. It was twelve years later that hospitals began to purchase them in large numbers.
Baxter Travenol
Baxter Travenol provides another wartime success story. Although the medical theory underlying intravenous (IV) therapy was clearly understood at the outset of the twentieth century, only large research and teaching hospitals could prepare solutions and equipment properly. Even carefully prepared solutions caused adverse reactions, such as severe chills and fevers, because pyrogens produced by bacteria remained in the solutions after sterilization.
In 1931, Idaho surgeon Ralph Falk, his brother, and Dr. Donald Baxter believed they could eliminate the pyrogen problem through controlled production in evacuated containers. When reactions continued to occur in patients, the doctors discovered that pyrogens were present in the rubber infusion equipment used by hospitals. They used disposable plastic tubing to eliminate this source of bacteria and worked with a glass manufacturer to produce a coating that resisted the contamination caused by the deterioration of the bottles that held the solutions.
In 1939 the fledgling company pioneered another medical breakthrough—a container for blood collection and storage. It was the first sterile, pyrogen-free, vacuum-type blood unit for indirect transfusion. It allowed storage of blood products for up to twenty-one days, making blood banking practical for the first time. The enormous demand for IV equipment and blood transfusions during World War II was a boon for Baxter. Its solutions were the only ones approved for wartime use by the U.S. military. Sales dropped dramatically after the war and rose again several years later during the Korean War. These fluctuating fortunes stabilized, and twenty-five years of uninterrupted company growth occurred after 1955 onward.[66]
This information comes from Baxter Travenol Laboratories Public Relations Department. The publication is entitled "The History of Baxter Travenol" and is unpaginated.
General Electric
General Electric was involved in every facet of the war, including building engines for planes, tanks, ships, and submarines. It
provided electrical capacity for large-scale manufacturing and also built power plants, testing equipment, and radio equipment.[67]
John Anderson Miller, Men and Volts at War: The Story of General Electric in World War II (New York: McGraw-Hill, 1947).
GE's activities extended to medical care for combat forces. Innovation was integral to that effort, as well as to the engines of war. "All along the story was the same. The war production job was one of prodigious quantities of all kinds of equipment. But it was also a job of constantly seeking ways to improve that equipment. Only the best was good enough, and the best today might be second best next week."[68]Ibid., 11.
General Electric's war-inspired medical equipment innovations included portable X-ray machines for use on ships and relatively inaccessible stations such as Pacific island hospitals. X-ray machines were also used to screen inductees for tuberculosis, and GE created cost-saving features, including machines that used smaller films. In addition, it built refrigeration and airconditioning systems for blood and penicillin storage. Government purchasing expanded market size. The army bought hundreds of electrocardiograph machines, ultraviolet lamps, and devices for diagnosis and therapy treatments.[69]
Ibid., 189.
The war affected innovation in dramatic ways. At the discovery stage, medical device innovation was stimulated and encouraged. Many technological innovations in materials science, radar, ultrasound, and other advancements had significant medical implications in the postwar period. Government purchasing stimulated the distribution of devices as well. The number of device producers and the value of their shipments grew in every SIC code.
Just as important, but less visible, were the institutional changes that occurred. Before the war, major public institutions had been formed that presaged government intervention in the discovery phase, most notably the NIH and the FDA. Wartime demands also accelerated the general public's acceptance of government involvement in scientific research and new technology. All these forces led to significant government activity in all phases of medical device innovation. The patient soon received extensive treatment.