Chapter 6—
Microbodies
6.1—
Introduction
Most of the organelles of the plant cell were detected by the classical techniques of light microscopy and were described by the turn of this century. Microbodies however were discovered relatively recently with the advent of the electron microscope. They were first recognized in the early 1950's as small spherical bodies in electron micrographs of mammalian kidney and liver tissue and similar organelles were reported in plant tissues in the early 1960's. Later in that decade they were isolated from plant tissues and from studies of their biochemical functions were recognized as being of major importance in plant cell metabolism. It is perhaps interesting to point out that the physiological significance to plant cell metabolism of major organelles, such as mitochondria and chloroplasts, was recognized long before their component biochemical reactions had been studied in detail, while the discovery and isolation of microbodies allowed some already well known metabolic pathways to be ascribed to a specific organelle.
Two types of microbody with identical structures but with distinct physiological functions have been isolated from plant tissues. They have been called peroxisomes and glyoxysomes to distinguish their separate functions in cell metabolism. Peroxisomes occur in the leaves of higher plants and are closely associated with chloroplasts. They are the sites for the oxidation of glycollic acid, a product of carbon dioxide fixation. The oxidation of this compound, results in a release of carbon dioxide and oxygen uptake which is called photorespiration. Glyoxysomes on the other hand, occur abundantly during the germination of those seeds which store fats as a reserve material, and contain the enzymes necessary for the breakdown of fatty acids to acetyl-CoA and the synthesis of succinate from acetyl-CoA.
All microbodies appear to contain flavin-dependent oxidases and catalase. The oxidation of a substrate by a flavin-linked oxidase is accompanied by the uptake of oxygen and the production of hydrogen peroxide, which is broken down by catalase to oxygen and water.
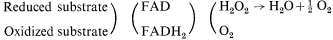
Microbodies, therefore, contribute to the uptake of molecular oxygen by the cell, but unlike mitochondria they do not contain any electron transport system which would be needed for the recovery of energy as ATP.
6.2—
Structure and Occurrence
Microbodies are usually spherical, but can be ellipsoidal or dumbell shaped, and range from 0.2 to 1.5 µm in cross sectional diameter. Because of the low contrast between the matrix of the microbody and the cytosol they are not detectable by light microscopy but in electron micrographs they can be seen to have a single limiting membrane enclosing a granular matrix of moderate electron density (Fig. 6.1). The core of the microbody commonly contains
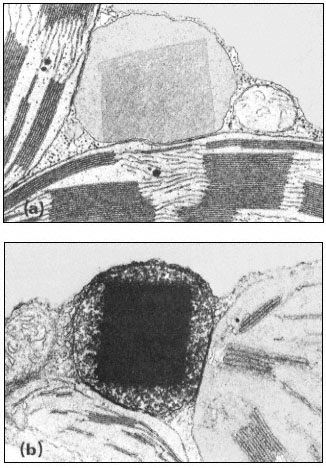
Figure 6.1a
A portion of a tobacco leaf cell showing a microbody with a crystalline
inclusion appressed to two chloroplasts. A mitochondrion lies
to the right of the microbody. (Magnification × 33,000).
b. A portion of a tobacco leaf cell, incubated in DAB medium, showing a heavy
deposition of osmium throughout the crystalline inclusion of the microbody.
(Magnification × 30,500). (Reproduced with permission from Frederick and
Newcomb (1969). Original prints supplied by Professor E. H. Newcomb.)
fibrillar inclusions or a single large crystalline inclusion which may be formed by a reorganization of the fibrillar material. These crystalline bodies have been shown to have catalase activity (Frederick & Newcomb, 1969). No ribosomes or any form of nucleic acids have been detected in the microbody and hence they are not considered to be capable of self-replication. Since they are found in many tissues in association with the endoplasmic reticulum they are thought to be formed by this structure. Microbodies are therefore structurally very simple and cannot be confused in electron micrographs with any organelle except perhaps lysosomes from which they can be distinguished by cytochemical techniques.
The presence of catalase is a distinguishing feature of microbodies and this can be detected cytochemically using the dye 3,3'-diaminobenzidine (DAB). In fixed sections, catalase remains active and in the presence of hydrogen peroxide the dye is oxidized to give an osmiophyllic electron-dense product in the core of the microbody.
Microbodies have been seen in electron micrographs of many plant tissues. They have been found in the leaves of angiosperms, gymnosperms and bryophytes where they are numerous and are invariably located near, or appressed to, chloroplasts. Microbodies have also been found in yeast and hyphal fungi and in species representing a wide range of algal phyla. In the fat-storing seeds of higher plants they are numerous at certain stages of germination and are found in the cells in close association with spherosomes which are large lipid storage bodies. In the roots of higher plants a catalase-containing microbody has been found in association with the endoplasmic reticulum but since it does not contain either glycollate oxidase or enzymes of the glyoxyllate cycle, its function in the cell is not clear.
6.3—
Isolation
Microbodies appear to be very fragile, since homogenization of plant tissue damages them to such an extent that their constituent enzymes appear in soluble fractions after removal of chloroplasts and mitochondria by centrifugation. More gentle techniques of tissue breakage however allow microbodies to be isolated at a yield of usually about 10% of the organelles present in the whole tissue as judged by the solubilization of enzymes presumed to be present in the microbody. The method of breakage depends upon the tissue. Leaves are ground for a brief time with a mortar and pestle at 0–4°C in a buffer containing an osmoticum such as sucrose at a concentration of 0.4 to 0.8 M . Other tissues may be chopped or finely sliced with razor blades in a similar medium. The resulting homogenate is squeezed through cheesecloth and the brei centrifuged at a low speed to remove cell debris. The supernatant is then centrifuged at 6,000 to 10,000 g to obtain a pellet containing broken chloroplasts, microbodies and some mitochondria.
Microbodies are separated from the other organelles by layering this resuspended pellet fraction on a discontinuous or continuous density gradient of sucrose ranging in concentration from 1.3 to 2.5 M , and centrifuging in an ultracentrifuge for 3–4 hr at 40,000 g. Microbodies increase in density during this process by a loss of water and ultimately form a band in the gradient at a density of 1.24 to 1.26 g cm–3 . This density is higher than that of other organelles and they are clearly separated from mitochondria which sediment at a desntiy of 1.16 to 1.19 g cm–3 (Fig. 6.2). On the separation of the gradient into fractions
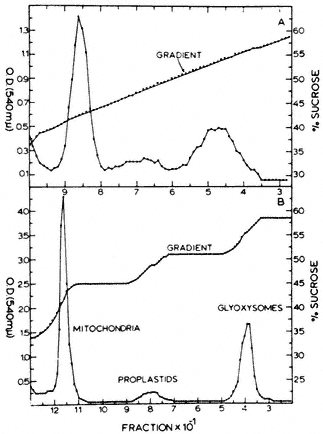
Figure 6.2
The distribution of protein in continuous (A) and
discontinuous (B) sucrose density gradients after
centrifugation of crude particles from castor bean endosperm.
(Reproduced with permission from Cooper & Beevers, 1969a.)
the microbodies are detected by assaying for specific 'marker' enzymes. Typical marker enzymes for peroxisomes are catalase, glycollate oxidase and hydroxypyruvate reductase, while those for glyoxysomes are catalse, ma late synthetase and isocitrate lyase. Mtitochondria are characterized by the presence of cytochrome c or succinic dehydrogenase, and chloroplasts by their chlorophyll pigments.
Much of the activity of the microbody marker enzymes is found in the soluble non-particulate fraction of gradients, indicating that the yield of micro-bodies is low. Despite these low yields, sufficient amounts of intact microbodies have been isolated from several plant tissues to be able to determine their metabolic functions. This has resulted in the distinguishing of at least two types of microbody, peroxisomes and glyoxysomes, with distinctly different enzyme complements and physiological functions.
6.4—
Glyoxysomes
There are many plant species in which lipid is the main storage material in the cotyledons or endosperm of the seed. During the first few days of seed germination there is a dramatic decrease in the lipid content of the seed and sugars, principally sucrose are formed. These sugars are subsequently translocated to the growing embryo or embryonic axis. This lipid to carbohydrate conversion has been correlated with an increase in the activity of the glyoxysomal enzymes, malate synthetase and isocitrate lyase, and as germination proceeds and the lipid reserves are depleted, the number of lipid storage bodies or spherosomes decrease and there is a drop in the activities of malate synthetase and isocitrate lyase. The elucidation of the metabolic processes involved in this process of gluconeogenesis, ultimately led to the isolation of particles in which were localized the crucial enzymes of this pathway. These particles were found to be morphologically similar to animal peroxisomes and were called glyoxysomes.
The breakdown of lipids is initiated by their hydrolysis to fatty acids. Triglycerides are hydrolized to glycerol and fatty acids by the enzyme lipase while phospholipids are hydrolized by phospholipase. The resultant long-chain fatty acids are subsequently degraded by the successive removal of 2-carbon fragments in the process of b -oxidation.
6.4.1—
b -Oxidation
In the process of b -oxidation the removal of each 2-carbon fragment from a long chain fatty acid involves a succession of five reactions (Fig. 6.3). The sequence is initiated by the activation of the substrate by coenzyme-A, catalyzed by the enzyme fatty acid thiokinase in the presence of ATP. The fatty acyl-CoA is then oxidized by the removal of hydrogen from carbons 2 and 3 of the chain and a double bond between 2 and 3 is formed. In this reaction the hydrogen is transferred to FAD. This reacts with molecular oxygen to produce peroxide which is broken down by catalase, and results in the uptake of one mole of oxygen for every two moles of fatty acyl-CoA oxidized. The unsaturated acyl-CoA produced is hydrated to form 3-hydroxy acyl-CoA, the reaction being catalysed by enoyl hydratase or crotonase, and this product then oxidized, with a concomitant reduction of NAD+ , to form a 3-keto acyl-CoA by the action of hydroacyl-CoA dehydrogenase. In the final reaction the 3-keto acyl-CoA is cleaved by the enzyme thiolase into acetyl-CoA and a new fatty acyl-CoA. The fatty acyl-CoA re-enters the reaction sequence and successive acetyl-CoA units are generated.
Fatty acids with an even number of carbons yield only acetyl-CoA units but those with an odd number of carbons result in the formation of acetyl-CoA and propionyl-CoA. In plant tissues propionyl-CoA is degraded by a modified b -oxidation sequence to yield acetyl-CoA and carbon dioxide.
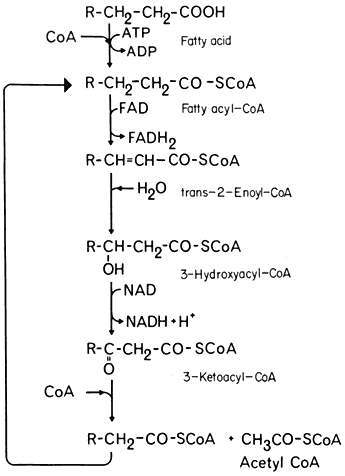
Figure 6.3
The b -oxidation pathway.
The b -oxidation pathway was unequivocally demonstrated to occur in plant tissues by Stumpf and Barber (1956) who showed that long chain aliphatic acids were oxidized to carbon dioxide by mitochondrial preparations from germinating peanut cotyledons, when these preparations were supplemented with a number of cofactors including ATP, CoA, and NAD+ . The rate of release of 14 CO2 from specifiically labelled butyric and palmitic acids was consistent with their degradation by b -oxidation and subsequent oxidation by the TCA cycle.
6.4.2—
The Glyoxyllate Cycle
The acetyl-CoA derived from fatty acid breakdown in germinating seeds could be consumed by the TCA cycle, as indicated by the experiments of Stumpf and Barber (1956). However this would not result in the net formation of a glucose precursor since for each molecule of acetyl-CoA consumed two molecules of carbon dioxide would be produced. Furthermore, it was known at the time that little of the fatty acid was oxidized to CO2 but instead contributed to a net synthesis of sugars.
The problem of how acetyl-CoA was converted to a glucose precursor was solved by the discovery of the glyoxyllate cycle, by Kornberg and Krebs in 1957. This cycle represents a modification of the tricarboxylic acid cycle in which two molecules of acetyl-CoA are consumed and a molecule of succinic acid is formed (Fig. 6.4). Five enzymes are involved in this process three of which,
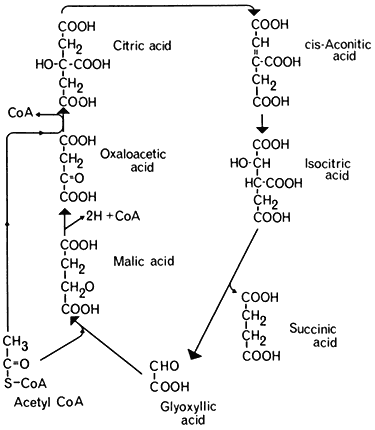
Figure 6.4
The glyoxyllate cycle.
citrate synthetase, aconitase, and malic dehydrogenase are components of the TCA cycle. The remaining two enzymes, the key enzymes of the glyoxyllate cycle, are isocitric lyase (isocitratase) and malate synthetase. The first reaction of the glyoxyllate cycle, catalyzed by citrate synthetase, is the condensation of oxaloacetate and acetyl-CoA to form citrate, which is then converted to isocitrate by the action of aconitase. The next reaction, unique to this cycle, is the cleavage of isocitrate into succinate and glyoxyllate catalyzed by isocitrate lyase. One of the products of this reaction, glyoxyllate, is then condensed with a second molecule of acetyl-CoA under the catalytic action of malate synthetase, to produce one molecule of malate. Malate is then oxidized by malate dehydrogenase to oxaloacetate with the concomitant reduction of NAD+ . The overall equation for the cycle is therefore:
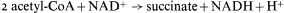
Succinate produced by the glyoxyllate cycle can then be converted to hexose by conversion to oxaloacetate, by the action of succinic dehydrogenase and fumarase. The oxaloacetate is converted to phosphoenolpyruvate, a reaction catalised by phosphoenolpyruvate carboxykinase,
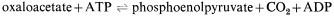
and the phosphoenolpyruvate converted to glucose by a reversal of the reactions of the Embden-Meyerhof-Parnas pathway. Thus four molecules of acetyl-CoA will give rise to two molecules of succinate and this in turn will result in the
formation of one molecule of glucose and the loss of two molecules of carbon dioxide (Fig. 6.5).
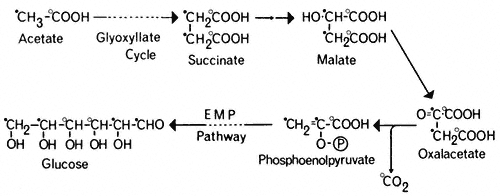
Figure 6.5
The pathway of incorporation of [14 C] from [1-14 C]-acetate (O) or
[2-14 C]-acetate (

The glyoxyllate cycle was first demonstrated in the bacterium Pseudomonas (Kornberg & Krebs, 1957) grown on two-carbon compounds, and has since been shown to operate in many micro-organisms and plant tissues. The evidence for the cycle is based on the presence of the two key enzymes, malate synthetase and isocitrate lyase, and on the distribution of 14 C in organic acids, sugars and carbon dioxide when the tissue is supplied with specifically labelled [14 C] acetate. Malate synthetase and isocitrate lyase are found in a wide variety of plant tissues (Carpenter & Beevers, 1958), particularly in fatty seedlings where they increase in activity during germination. Similarly the activities of aconitase and citrate synthetase increase in these tissues during germination. Incubation of castor bean endosperm tissue with [I-14 C] acetate or [2–14 C] acetate results initially in the formation of [14 C] malate, and subsequently the radioactivity from [I-14 C] acetate results in about an equal labelling of CO2 and sucrose. In the cotyledons of germinating peanut and sunflower seedlings (Bradbeer & Stumpf, 1959) and castor bean endosperm (Canvin & Beevers, 1961), [1-14 C] acetate was converted to carboxyl-labelled malate and to sucrose in which the glucose moiety was labeled in the 3 and 4 carbons, while [2-14 ]C acetate gave rise to malate labelled in the methylene carbons and to sucrose where the glucose moiety was labelled in the 1, 2, 5 and 6 carbons. These patterns or labelling of the products of acetate metabolism are consistent with the operation of a glyoxyllate cycle in these tissues (Fig. 6.5).
6.4.3—
Metabolic Functions of the Glyoxysome
The reactions of the glyoxyllate cycle and the pathway of b -oxidation were generally thought to be associated with the mitochondria since the enzymes of these pathways were usually present in the mitochondrial fraction isolated from cell homogenates. Elegant studies by Beevers' group at Purdue University however showed that sucrose density centrifugation of crude particulate fractions of castor bean endosperm resulted in the separation of three distinct bands of
particles which were identified as proplastids, mitochondria and a new particle sedimenting at a high density. Enzymes of the glyoxyllate cycle, isocitrate lyase and malate synthetase were found exclusively in this dense particle together with catalase and a large proportion of the glycollate oxidase of the gradient. On the other hand citrate synthetase and malate dehydrogenase were associated with both the mitochondrial band and the band containing the new particle, while succinic dehydrogenase, fumarase and NADH oxidase were located exclusively in the mitochondrial band with cytochrome oxidase (Fig. 6.6).
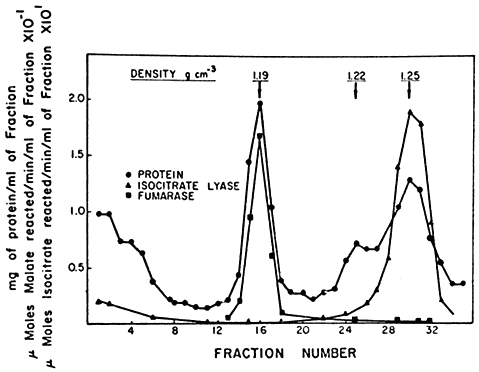
Figure 6.6
The distribution of protein, fumarase and isocitric lyase
after sucrose density gradient separation of the components of
a crude particulate fraction of the endosperm of germinating castor bean.
(Reproduced with permission from Breidenbach & Beevers, 1967.)
The enzyme distribution indicated that the TCA cycle enzymes were located in the mitochondria while the enzymes of the glyoxyllate cycle were compartmentalized in the denser particle. These particles were therefore called glyoxysomes (Breidenbach & Beevers, 1967; Breidenbach et al., 1968). The isolated glyoxysomes were found to be organelles with a single unit membrane bounding a finely granular matrix. Similar structures were recognized in electron micrographs of intact castor bean endosperm tissue indicating that the isolated organelles were not artefacts of the isolation and centrifugation processes. Since the finding of glyoxysomes in endosperm tissue, they have been reported to be the site of the glyoxyllate cycle in the cotyledons of a number of fatstoring seeds including those of watermelon, sunflower, peanut and cucumber. Microbodies containing catalase and enzymes of the glyoxyllate cycle have also been found in yeast (Szabo & Avers, 1969) and although these have been called peroxisomes they clearly have the enzyme complements of glyoxysomes.
In addition to glyoxyllate cycle activity, the glyoxysomes were shown to be the site of b -oxidation in castor bean endosperm (Cooper & Beevers, 1969b; Hutton & Stumpf, 1969). Glyoxysomes isolated from this tissue oxidized palmitoyl-CoA to acetyl-CoA with a concomitant reduction of NAD+ and uptake of oxygen. Addition of [14 C] oxaloacetate during this reaction resulted in the formation of [14 C] citrate and [14 C] malate from palmityl-CoA indicating that the acetyl-CoA produced by the b -oxidation process was consumed in the glyoxyllate cycle also located in the organelle (Cooper & Beevers, 1969b). Similarly, ricinoleate, linoleate and palmitate were oxidized by glyoxysomes of castor bean with the formation of acetyl-CoA, and three enzymes of the b -oxidation complex, crotonase, b -ketothiolase, and b -hydroxyacyl dehydrogenase were found to be located specifically in the organelle (Hutton & Stumpf, 1969). The activity of the b -oxidation complex in this tissue during the germination of the seed was found to parallel the increase in activity of the glyoxyllate cycle enzymes indicating that an integrated system for lipid utilization develops together with the formation of the glyoxysome.
The principal pathways of gluconeogenesis are therefore compartmentalized in a single organelle, the glyoxysome. The complete pathway of gluconeogenesis however involves at least three organelles, the spherosome, the glyoxysome and the mitochondrion. The reactions are initiated by a hydrolysis of lipids in the spherosome by the action of lipase, and the glycerol and fatty acids produced diffuse out of the organelle. Glycerol is utilized directly by the EMP pathway in the cytosol and contributes to sucrose synthesis (Beevers, 1956) while the fatty acids diffuse into glyoxysomes which are located near the spherosome. In the glyoxysome the fatty acid is oxidized in the b -oxidation pathway and the acetyl-CoA released is converted to succinate by the action of the glyoxyllate cycle enzymes located in the organelle. Although the b -oxidation pathway is reversible, the equilibrium is presumably maintained in a catabolic direction by the removal of peroxide, produced in the oxidation step of the pathway, by the catalase present in the glyoxysome. Reduced NAD+ produced in these reactions is probably oxidized in the mitochondria. Succinate produced in the glyoxysome is further metabolized to oxalacetate and finally to phosphoenolpyruvate in the mitochondria since succinic dehydrogenase and fumarate are not component enzymes of the glyoxyllate cycle and are absent from glyoxysomes.
This compartmentalization of the b -oxidation complex and the glyoxyllate cycle together in an organelle discrete from the mitochondrion is probably the reason why acetyl-CoA is utilized in gluconeogenesis in plant tissues rather than being oxidized to carbon dioxide and water as in animal tissues. Free acetyl-CoA-is presumably not released from the glyoxysome and made available for oxidation in the mitochondrion, while consumption of succinate, the final product of the glyoxyllate cycle, by the mitochondrion, would not stimulate the rate of oxidation in the TCA cycle since this can only occur by increasing the supply of acetyl-CoA. Some measure of control of competing metabolic pathways is therefore achieved by separation of these reactions within different organelles.
6.5—
Peroxisomes
Glycollic acid is produced in large amounts in the chloroplast, as a by-product of the reactions of carbon dioxide fixation. The formation of glycollate has been proposed to occur by the oxidation of ribulose-1,5-bisphosphate (RBP) by molecular oxygen, a reaction which would produce a two carbon fragment of phosphoglycollic acid and a three carbon fragment of phospholgyceric acid (PGA) instead of two molecules of PGA resulting from the normal carboxylation of RBP by CO2 . It has been found that RBP carboxylase acts as an oxygenase in the presence of molecular oxygen and that phosphoglycollate and PGA are produced in this reaction in vitro (Andrews et al., 1973). Thus the enzyme RBP carboxylase can act as an oxygenase or as a carboxylase and the formation of phosphoglycollic acid is favoured by high partial pressures of oxygen. Glycollate is produced from phosphoglycollate by the action of phosphoglycollate phosphatase, a chloroplast enzyme. Glycollate is also produced, in vitro, from fructose-6-phosphate by the action of the chloroplast enzyme, transketolase, which may be due to the oxidation, by hydrogen peroxide, of the glycolaldehyde-thiamine pyrophosphate, an intermediate complex in this enzyme reaction (Bradbeer & Racker, 1961). Glycollate is released from the chloroplast into the cytosol, where it is further metabolized in a specific metabolic pathway, the initial reactions of this pathway being located in the peroxisome.
6.5.1—
The Glycollate Pathway
The pathway of glycollate metabolism in leaves has been elucidated by infiltrating excised leaves or leaf discs with 14 C-labelled glycollate or other intermediates of the pathway. Glycollate is converted rapidly to glyoxyllate which may be oxidized non-enzymatically to carbon dioxide and formic acid, in the presence of hydrogen peroxide. In this reaction the carbon dioxide is derived from the carboxyl carbon of glycollate and the formate from the methyl carbon (Tolbert & Burris, 1950). However the presence of catalase, which breaks down peroxide, is thought to preclude such a total degradation of glyoxyllate in the peroxisome and the supply of 14 C-labelled glycollate or glycoxyllate to leaf tissue in the light has been found to give rise initially to labelled glycine and serine and subsequently to labelled glyceric acid hexoses and sucrose (Tolbert, 1963; see Fig. 6.7).
Infiltration of [2-14 C] glycollate into leaves in the light was found to give [2-14 C] glycine but serine was found to be labelled in the 2 and 3 carbons (Tolbert & Cohan, 1953; see Fig. 6.7). From this evidence it was concluded that two molecules of glycine give rise to one molecule of serine with a loss of one molecule of carbon dioxide; the 1 and 2 carbons of serine are derived from carbons 1 and 2 of glycine respectively, while the 3 carbon of serine is derived from the 2 carbon of glycine and carbon dioxide arises from carbon 1 of glycine. In wheat leaves in light [3-14 C] serine was converted to [3-14 C] glycerate
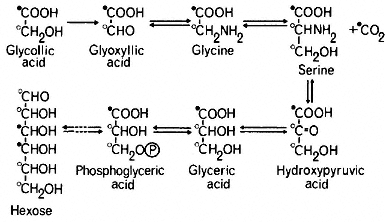
Figure 6.7
The glycollate pathway. The labelling pattern of intermediates of the
pathway ane hexose is indicated for when they are derived from
[1-'4 C]-glycollate (

and this was incorporated into hexose presumably by the reactions of the Embden-Meyerhof-Parras pathway (Rabson et al., 1962). This pathway of hexose formation from glycollate has been confirmed by the finding that [2-14 C] glycollate gives rise to glycose labelled in the 1, 2, 5 and 6 carbons and [3-14 C] serine to glucose labelled in the 1 and 6 carbons (Jiminez et al., 1.962).
The glucollate pathway is therefore gluconeogenic in light. The conversion of glyceric acid derived from glycollate, to glucose and sucrose is inhibited by DCMU an inhibitor of Photosystem II (Miflin et al., 1966). In the dark, supplied [14 C]-glycollate is converted into TCA cycle acids rather than sugars which may be due to an oxidation of pyruvate derived from glyceric acid, or may result from a direct conversion of glyoxyllate to malate.
Glycollate produced during photosynthesis in the presence of 14 CO2 is usually found to be uniformly labelled i.e. both carbons have the same specific activity. This distribution of radioactivity would be expected if the glycollate was derived from carbons 1 and 2 of ribulose-1,5-bisphosphate. Consequently all the compounds arising from glycollate are uniformly labelled. Serine, in particular, has been found to be uniformly labelled while phosphoglyceric acid was predominately carboxyl labelled, indicating that serine was produced from glycollate rather than directly from phosphoglyceric acid formed in CO2 -fixation (Rabson et al., 1962). The uniformly labelled glyceric acid formed in this pathway in turn produces uniformly labelled hexoses instead of the 3,4-14 C-hexoses resulting from incorporation of 14 C from the photosynthetic carbon cycle.
The operation of the glycollate pathway in leaves has been shown by tracer experiments but it has been confirmed by the detection of enzymes in leaves which are necessary for some reactions of the pathway. The initial reaction, the oxidation of glycollate to glyoxyllate is catalysed by glycollate oxidase, an enzyme first isolated by Zelitch and Ochoa (1953). It has FMN as the prosthetic group and utilizes molecular oxygen as the electron acceptor. The enzyme is competitively inhibited by bisulphite addition compounds of aldehydes, a -hydroxy-sulphonates, which have the general structure R-CHOH-SO3 H and
are therefore structural analogues of glycollate. The most commonly used a -hydroxysulphonate is a -hydroxypyridylmethane sulphonate (HPMS) and treatment of leaf discs or infiltration of excised leaves with this compound results in the accumulation of glycollic acid while having no effect upon the rate of CO2 -fixation. Experiments of this type have shown that 40 to 70: of the carbon fixed in photosynthesis will accumulate as glycollate in tissues treated with HPMS and these results have been interpreted as indicating that a large fraction of the carbon fixed in photosynthesis is metabolized by this pathway.
Transaminases are present in leaves which catalyse two steps of the pathway: a glutamate-glyoxyllate transaminase catalysing the formation of glycine is widespread in plant tissues, and a serine-pyruvate aminotransferase catalysing the formation of serine to hydroxypyruvate is found in leaves. The conversion of glycine to serine is catalysed by serine hydroxymethyltransferase which has been found in pea and wheat leaves (Cossins & Sinha, 1966). This reaction is inhibited by isonicotinyl hydrazide, and treatment of leaf tissue with this compound during photosynthesis in 14 CO2 causes an accumulation of [14 C] glycine and [14 C] glycollate and a decrease in the incorporation of 14 C into glucose (Miflin et al., 1966). Use of this inhibitor thus provides additional evidence for the operation of the pathway.
Two types of glyoxyllate reductase have been found to occur in leaves. One, an NADP-linked enzyme, is thought to be located in the chloroplast, while a second NAD-linked enzyme is present in the cytoplasm and is referred to as hydroxypyruvate reductase since the enzyme isolated from some sources has a higher activity to hydroxypyruvate than to glyoxyllate. The enzyme appears to catalyse the reduction of hydroxypyruvate to glycerate in the glycollate pathway.
6.5.2—
Metabolic Reactions of the Peroxisome
It was generally accepted that the enzymes of the glycollate pathway were soluble proteins of the cytosol and the failure of several attempts to localize these enzymes, particularly glycollate oxidase, in discrete organelles confirmed this idea. The first successful localization of enzymes of glycollate metabolism in a discrete organelle was achieved by Tolbert and coworkers at Michigan State University (Tolbert et al., 1968). They demonstrated that sucrose density gradient centrifugation of spinach homogenates separated three bands of particles: broken chloroplasts, mitochondria, and small bodies distinctly separated from, and denser than the other organelles. Electron microscopic examination of these bodies showed them to be organelles bounded by a single unit membrane and since they closely resembled peroxisomes from animal cells in size and morphology, they were referred to as leaf peroxisomes. The most important finding was that these peroxisomes contained the bulk of the activity of the glycollate oxidase, catalase and hydroxypyruvate reductase (NAD-glyoxyllate reductase) of the gradient, while cytochrome c oxidase was specifically located in the mitochondrial fraction. Peroxisomes were later
isolated from the leaves of nine other plant species including tobacco, maize, and sugarcane, and all contained the same enzyme complement (Tolbert et al., 1969). These studies therefore demonstrated that processes of glycollate oxidation and peroxide breakdown were localized together in an organelle discrete from the chloroplast. Electron microscope studies, particularly by Newcomb, have since revealed the presence of peroxisomes in leaves of many plant species (e.g. Frederick & Newcomb, 1969).
Further studies in Tolbert's laboratory have shown that other enzymes of the glycollate pathway are also localized in the peroxisome. Two aminotransferases, glutamate-glyoxyllate aminotransferase catalysing the conversion of glyoxyllate to glycine, and serine-pyruvate aminotransferase catalysing the conversion of serine to hydroxypyruvate, were found in peroxisomes isolated from leaves of various species. Serine hydroxymethyltransferase is the only enzyme of the glycollate pathway not found in the peroxisome, and is probably located in the mitochondrion. Supply of [14 C] glycollate and [14 C] glyoxyllate to isolated peroxisomes gave rise only to [14 C] glycine and while oxygen uptake occurred, no 14 CO2 release was detected (Kisaki & Tolbert, 1969).
The glycollate pathway appears to require enzymic steps located in three subcellular organelles (Fig. 6.8). Glycollate, formed in the chloroplast is
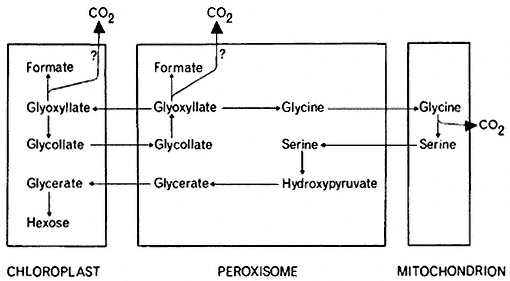
Figure 6.8
The distribution of the reactions of the glycollate
pathway among organelles of the leaf cell.
oxidized to glyoxyllate in the peroxisome. The glyoxyllate may then be exported to the chloroplast where it could be reduced to glycollate by the action of NADP-dependent glyoxyllate reductase which is located specifically in the chloroplast (Tolbert et al., 1970). Such a coupling of alternate oxidation and reduction reactions. a 'glycollate-glyoxyllate shuttle', has been proposed as a mechanism for controlling the levels of reduced NADP in the chloroplast, but no unequivocal evidence for such a reaction in vivo has been found. Glycine is formed from glyoxyllate in the peroxisome and then transferred to the
mitochondrion where it is converted to serine, with a concomitant loss of carbon dioxide. The conversion of serine to glycerate can then be accomplished by enzymes localized in the peroxisome. Further metabolism of glycerate to hexose appears to be confined to the chloroplast since these reactions would be initiated by the formation of phosphoglyceric acid, a reaction catalysed by phosphoglycerate phosphatase which is located in the chloroplast.
The metabolism of glycollate by leaf tissue results in the release of carbon dioxide, but the exact site of this CO2 release in the cell is still a controversial question. It has been proposed that CO2 is evolved by the mitochondria during the conversion of glycine to serine, since [14 C] glycine is as good a precursor as [14 C] glycollate for 14 CO2 evolution in leaves and the 14 CO2 evolved is derived from the carboxyl groups of these compounds. The presence of catalase in the peroxisomes is thought to minimize the non-enzymatic oxidation of glyoxyllate by H2 O2 to formate and carbon dioxide and no evolution of CO2 by isolated peroxisomes from glycollate or glyoxyllate could be detected by some workers (Kisaki & Tolbert, 1970). This loss of CO2 from glycine, which amounts to only 25% of the total carbon passing through the glycollate pathway, would not account for the large losses of CO2 which occur as a result of the photorespiration of glycollate in leaves. However the amount of catalase present in the peroxisome may not preclude the non-enzymatic oxidation of glyoxyllate and it has been shown that both [14 C] glycollate and [1-14 C] glyoxyllate can be decarboxylated by peroxisomal fractions at pH 8.0 (Halliwell & Butt, 1974). An enzyme is also present in chloroplasts which catalyses the decarboxylation of glyoxyllate to formic acid and CO2 (Zelitch, 1972). Present evidence indicates therefore that three subcellular organelles have the capacity to decarboxylate components of the glycollate pathway and each may contribute to the production of the carbon dioxide in photorespiration (Fig. 6.8).
6.5.3—
Photorespiration
The oxidation of glycollate in the peroxisome is accompanied by a consumption of oxygen and ultimately results in the release of carbon dioxide. The net result is a respiratory gas exchange where the substrate of respiration is glycollate rather than glucose. Since glycollate is only formed in light this respiration is light-dependent and is called photorespiration.
The direct measurement of oxygen uptake during a net photosynthetic release of oxygen, or CO2 release during net photosynthetic CO2 fixation is impossible. However, indirect methods have demonstrated that photorespiration occurs in plants and that the rate of CO2 loss in light is higher than that in the dark. Measurements of photorespiration rates vary considerably with the assay method used and can only be considered as approximations of the magnitude of the process. Nevertheless, it is becoming clear that the process of photorespiration has great importance in decreasing the rate of net photosynthesis in many plants.
Photorespiration can be detected by measuring the flux, that is the simultaneous uptake and release, of oxygen or carbon dioxide of photosynthesizing tissue by using isotopic tracer methods. The uptake of 18 O2 by leaves during photosynthesis has been detected but experiments using 18 O2 have generally given equivocal results which have been difficult to interpret.
The measurement of carbon dioxide flux during photosynthesis is much more convenient and depends upon the accurate measurement of carbon dioxide concentration in the atmosphere using an infra-red gas analyser and a simultaneous measurement of the total activity of supplied 14 CO2 with an ion counter or Geiger-Müller counter. When a plant is placed in a closed system in light, there is a rapid uptake of carbon dioxide and the CO2 concentration of the atmosphere around the plant decreases to a concentration at which the uptake of CO2 exactly balances the output of CO2 by the plant (Fig. 6.9). This concentration of carbon dioxide is called the CO2compensation point of the plant and is usually measured in parts per million (ppm) of CO2 in air. Plants such as tobacco, sunflower and wheat have compensation points, ranging from 35 to 100 ppm indicating a marked loss of CO2 (i.e. photorespiration) during photosynthesis, but others such as maize and sugarcane have compensation points of 3 to 10 ppm indicating a low loss of CO2 or a low photorespiration rate. If photosynthetic carbon fixation is similarly measured in a closed-system but in an atmosphere containing 14 CO2 , the CO2 arising from, the plant by photorespiration will, over short time periods, be 12 CO2 . Thus, while a decrease in the CO2concentration of the atmosphere around the plant will occur, the radioactivity of 14 CO2 in the atmosphere will appear to decrease at a faster rate because of the efflux of unlabelled CO2 from the plant, and continues to decrease even after the compensation point is reached (Fig. 6.9a). In a plant which
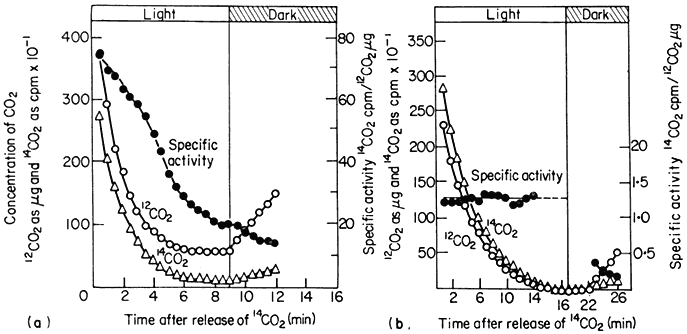
Figure 6.9
The concentration of CO2 and 14 CO2 and the specific activity of 14 CO2 around
a detached sunflower leaf (a) and a detached maize leaf (b) during illumination
and in subsequent darkness in an atmosphere of 21% oxygen and at 21°.
(Reproduced with permission from Hew et al., 1969.)
is photorespiring, therefore, a decrease in the specific radioactivity of 14 CO2 will occur, while in a plant with no photorespiration the uptake of 14 CO2 will not be accompanied by an efflux of 12 CO2 and the specific radioactivity of the 14 CO2 in the atmosphere will remain constant (Fig. 6.9b). In the dark the specific radioactivity of the 14 CO2 decreases with the efflux of 12 CO2 and the rate of decrease of this activity is a measure of dark respiration. The rate of photorespiration as measured by these methods has been shown to be 1.5 to 2.5 times that of dark respiration, while in plants with low photorespiration, such as maize, loss of CO2 in light is only a fraction of the rate of CO2 loss in the dark. This method clearly indicates that photorespiration occurs in some species while it is absent in others.
Another method which has been used to detect differences in the rate of photorespiration and dark respiration was devised by Zelitch (1968). In this method leaf discs are allowed to fix 14 CO2 for a period of 45 to 60 minutes. The remaining 14 CO2 is then quickly flushed out of the closed system, and the release of 14 CO2 from the tissue into CO2 -free air is measured over short periods in the light and the dark. The method is based on the assumptions that the rate of 14 CO2 loss is a measure of total CO2 loss i.e. that the specific radioactivity of the CO2 evolved remains constant over short time periods and that low CO2 tensions have no effect on the loss of CO2 . These assumptions may not be valid for all photosynthetic tissues but within these limitations the method is a very rapid and sensitive means for detecting photorespiration. Photorespiratory loss of CO2 in tobacco has been shown by this method to be 2 to 5 times higher than in the dark while CO2 loss from maize is not detectable (Fig. 6.10a). This method has been similarly used to detect photorespiration in other species.
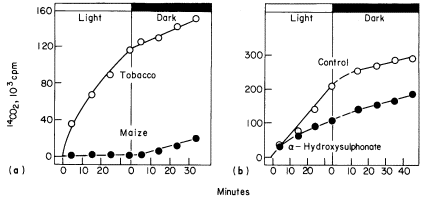
Figure 6.10a
A comparison between the release of 14 CO2 by tobacco and maize discs
in the light and the dark after previously being supplied 14 CO2 in light.
(b). The effect of a -hydroxysulponate on the release of
14 CO2 from tobacco leaf discs in the light and dark.
(Reproduced with permission from Zelitch, 1968.)
Photorespiration is not simply a stimulation of dark respiration since the two process of photorespiration and dark respiration respond differently to changes in oxygen concentration. Dark respiration of leaves has an optimal rate at about 2% oxygen in air and any increase in the O2 concentration up to 100% does not increase the rate (Fig. 6.11). Net photosynthesis however is inhibited
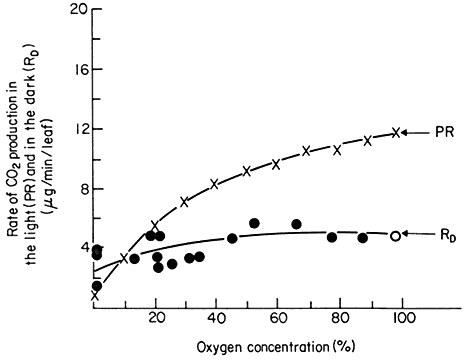
Figure 6.11
The effect of oxygen concentration on the rate of photorespiration
(PR) and dark respiration (RD ) of detached soybean leaves.
(Reproduced with permission from Forrester et al., 1966.)
by oxygen, a phenomenon called the Warburg effect, and this is attributable to an increase in the rate of photorespiration rather than to an inhibition of photosynthesis per se . A reduction in the oxygen concentration in the atmosphere around a leaf from the ambient 21% O2 of air has been shown to lower the compensation point and increase the rate of net photosynthesis, while an increase in concentration raises the compensation point and lowers net photosynthesis (Forrester et al., 1966). Plants with photorespiration have been found to evolve CO2 at a high rate when they are transferred to darkness after a period of photosynthesis. This post-illumination CO2 burst lasts for a few minutes before a steady dark respiration is established and the magnitude of the burst has been found to depend on the light intensity and oxygen concentration during the previous period of photosynthesis: the CO2 -burst increases with an increase in light intensity and decreases with a decrease in O2 concentration (Tregunna et al., 1961). This burst has been explained as being due to the continued slow oxidation, in the dark, of a product produced in photosynthesis after the uptake of CO2 by photosynthesis has stopped. Photorespiration therefore appears to have different characteristics than dark respiration and to have a different substrate.
The substrate for photorespiration is thought to be glycollic acid. Glycollic
acid production in leaves and in chloroplasts is stimulated by an increase in oxygen concentration as is photorespiration, while the inhibition of glycollate oxidation by HPMS has been found to lower the rate of photorespiration in leaf discs to that of dark respiration (Fig. 6.10b). Photorespiratory loss of CO2 is also stimulated by the infiltration of leaves with glycollic acid while acetate has no effect on this rate. Thus the oxidation of glycollate, mediated by the leaf peroxisomes results in a photorespiratory loss of carbon dioxide by the leaf.
Plants with low compensation points are said to lack photorespiration. These plants are C4 plants, that is, the primary carboxylation step is catalysed by phosphoenolpyruvate carboxylase rather than by RBP carboxylase. This efficient fixation of CO2 has been postulated to preclude the formation of glycollate, but maize for example, has been shown to possess a glycollate pathway (Osmond, 1969) and peroxisomes occur in the mesophyll cells of the leaf. Presumably in such plants glycollate may be formed and oxidized, but the efficiency of refixation of the CO2 resulting from glycollate oxidation is such that no CO2 is released from the plant and hence no photorespiration is detected.
Estimates from various plant species suggest that from 15 to 40% of the carbon fixed in photosynthesis is lost by photorespiration. There is no unequivocal evidence to show that energy in the form of ATP is recovered during the oxidation of glycollate and the process appears to be a wasteful one in terms of energy conservation. An important question therefore arises: does photorespiration serve a useful function or is it simply an inevitable consequence of photosynthesis being carried on in an atmosphere of 21% oxygen? It has been suggested that photorespiration might act as a 'safety valve' for the plant, in that, under conditions of high light intensity and low carbon dioxide concentration, the oxidation of glycollate would consume both excess reduced NADP+ and excess oxygen, which would serve to protect the chloroplast from photo-oxidative damage. If the process of photorespiration imposes limitations on the growth of plants, it would be expected that there would have been some selective pressure to eliminate it by natural selection during the evolution of the higher plants. The occurrence of C4 plants, which have low photorespiration, may represent such an evolutionary step to correct for the presence of photorespiration by the development of an efficient CO2 -fixation mechanism. Since variations in the rate of photorespiration occur within a single species (Zelitch, 1971) it may be possible to select artificially for low photorespiration in crop plants and consequently increase net photosynthetic productivity and crop yield.
6.6—
Ontogeny and Turnover of Microbodies
The development of microbodies in plant cells is a difficult process to follow since they usually occur in small numbers and are difficult to characterize
enzymatically. Small, membrane-bound organelles occur in close proximity to the endoplasmic reticulum but these do not always give a positive reaction to DAB, the usual criterion for the presence of catalase. Nevertheless catalase is invariably the first enzyme to be detected in microbodies and it appears to be the most active enzyme in the developing organelle. Studies of microbody development have been carried out principally on tissues having large populations of microbodies such as the endosperm and cotyledons of fatty seedlings.
In the endosperm of fat-storing seeds, particularly of the castor bean, the process of germination results in the development of large numbers of microbodies which reach numbers per cell twice that of mitochondria (Vigil, 1970). This increase in number is correlated with increases in the activities of isocitrate lyase and malate synthetase in the whole tissue. It has been suggested by several investigators that microbodies develop from the endoplasmic reticulum by a budding process. In castor bean the formation of microbodies has been linked directly to the endoplasmic reticulum and connections between the cisternae of the rough endoplasmic reticulum and developing microbodies have been clearly demonstrated (Vigil, 1970). This connection does not appear to persist during the whole course of organelle development although they increase seven-fold in size and increase in density suggesting a continuous addition of newly synthesized protein. The depletion of lipid in the endosperm or cotyledons of fatty seedlings is accompanied by a decrease in glyoxysomal enzymes which is attributable to a decrease in the number of glyoxysomes as measured by enzyme activity and total protein (Gerhardt & Beevers, 1970). It is not clear how these microbodies are destroyed but some at least become enclosed in autophagic vacuoles and are digested.
Increases in peroxisomal enzymes have been detected in germinating seedlings and etiolated seedlings on exposure to light and some of these changes have been correlated with the size and structure of microbodies. The development of microbodies in, leaves of wheat seedlings in light involves distinct changes in the biochemical and physical characteristics of the organelle. During the first few days of germination an increase occurred in the density of catalasecontaining particles from 1.17 to 1.24 g cm–3 on separation on sucrose gradients, which was paralleled by an increase in catalase, hydroxypyruvate reductase and glycollate oxidase (Fierabend & Beevers, 1972).
In etiolated leaves small microbodies occur which are rich in catalase but low in other peroxisomal enzymes. These bodies have not been unequivocally characterized as peroxisomes because of the difficulty of isolation. Upon exposure to light there is an activation of glycollate oxidase and hydroxypyruvate reductase, but these changes in enzyme activity have not been correlated with an increase in size and number of peroxisomes. The activity of glycollate oxidase is stimulated by exposure of etiolated tissue to red light and this activation is reversed by -far-red light. These effects probably reflect a stimulation of leaf development rather than a specific phytochrome involvement in peroxisome development. However an increase in glycollate oxidase occurs under
continuous far-red light in mustard seedlings where chloroplast development is inhibited, suggesting that substrate activation is not involved in this increase of enzyme activity. Similar increases in the activities of glycollate oxidase, and hydroxypyruvate reductase have been found in cotyledons of germinating seedlings on exposure to light (Trelease et al., 1971; Kagawa et al., 1973).
In germinating seeds and in greening leaves the microbodies have easily identifiable functions, those of glyoxysomes or peroxisomes, and only one type of microbody occurs in each tissue. However an interesting situation occurs in some seeds of the Cucurbitacea. In cucumber and watermelon for example, lipid is the main storage material in the seed and during germination there is an increase in activity of glyoxysomal enzymes in the cotyledon. During normal development, the cotyledons become exposed to light and turn green. The glyoxysomal activity of the microbodies, as measured by the activity of malate synthetase and isocitrate lyase, decreases while the peroxisomal activity, as measured by glycollate oxidase activity, increases. In one organ therefore in response to environmental stimulus there is a transition from lipid breakdown and gluconeogenesis, to glycollate metabolism, which is reflected in a change in the microbody population from a glyoxysomal to a peroxisomal function. Since the two microbodies are morphologically indistinguishable the question arises as to whether the structure of the glyoxysome is retained and there is a selective replacement of enzymes to give it a peroxisomal function or whether there is an autolysis of the glyoxysome and a replacement of it by a newly synthesized peroxisome.
The changes in microbody enzyme complements which occur during seedling development have been correlated with changes in the fine structure of the microbodies. It has been reported that in cucumber seedlings the decrease in glyoxysomal enzyme activity is not accompanied by a decrease in the number of microbodies as determined by electron microscopy. The subsequent exposure of the cotyledons to light caused a great increase in the activity of peroxisomal enzymes, particularly that of glycollate oxidase, while there was not a concomitant increase in the number of microbodies (Trelease et al., 1971). No evidence was found for the degradation of microbodies during the loss of glyoxysomal enzyme activity as has been reported in castor bean (Vigil, 1970). These studies suggest that there is a continuity of microbody structure in the transition from a glyoxysomal to a peroxisomal function. On the other hand, in developing watermelon seedlings, the decrease in the total activity of glyoxysomal enzymes in isolated microbodies which occurred after the depletion of lipid, was accompanied by a decrease in the total protein of these microbodies. This was interpreted as indicating a destruction of glyoxysomes in the tissue (Gerhardt & Beevers, 1970). Normally the peroxisomal activities of the microbody increase as glyoxysomal activity decreases, but in this tissue the decrease in glyoxysomal activity of the microbodies could be separated from the increase in the peroxisomal activity. Brief exposure of dark-grown cotyledons to light at an early stage of development stimulated the activities of peroxisomal enzymes, while
this did not affect the rate of decline of the activity of glyoxysomal enzymes (Kagawa et al., 1973). These results suggest that the formation of peroxisomes can occur while active glyoxysomes are present and that two populations of microbody can occur in the tissue at the same time. The apparent contradiction in the results from these two tissues has still to be resolved, and it is still not clear whether the transition from a glyoxysomal function to a peroxisomal function in the microbodies of these tissues is due to enzyme replacement in a single microbody or to synthesis of a new microbody.
6.7—
Algal Microbodies
The microbodies of photosynthetic higher plants have distinct physiological roles either as peroxisomes or glyoxysomes which are active at different stages of the life cycle of the plant. In the algae however their role is not quite as clear since both functions of the microbody, i.e. glycollate oxidation and gluconeogenesis, may be carried on in a single cell. Microbody-like organelles have been found in a wide range of algae, and they are most numerous in algal cells grown on two-carbon compounds. Catalase has been detected cytochemically by DAB staining in situ but has not been shown to be universally present in algal microbodies.
Algae grown autotrophically, that is, in light on carbon dioxide, produce glycollic acid which is metabolized by a pathway similar to that of higher plant leaves (Bruin et al., 1970; Lord & Merrett, 1970). Glycollate oxidase however is not present in algae, with the possible exception of some members of the Zygnematales, Ulotrichales and Charophyceae (Fredrick et al., 1973) but the oxidation of glycollate is catalysed by glycollate dehydrogenase. This enzyme is not FMN dependent and does not couple to molecular oxygen so that hydrogen peroxide is not formed during glycollate oxidation (Nelson & Tolbert, 1970). This may to some extent account for the low activities of catalase reported in many algae. The natural electron acceptor for glycollate dehydrogenase is as yet unknown.
While the presence of a glycollate pathway has been established in green algae, the extent to which glycollate is metabolized by this pathway during photosynthesis is not known. In contrast to higher plant cells, serine formed during photosynthesis in 14 CO2 in several algae is carboxyl labelled indicating its formation directly from PGA rather than by the glycollate pathway (Bruin et al., 1970). There is also a lower rate of CO2 loss by photorespiration in algae than in higher plants (Cheng & Colman, 1974) suggesting perhaps that little glycollate is formed in algal photosynthesis. Glycollate is excreted by algal cells and this has been interpreted to indicate that the algae have a low rate of glycollate metabolism. This can now be discounted however since little glycollate is excreted during steady-state photosynthesis (Watt & Fogg, 1966; Colman et al., 1974) and all the glycollate formed appears to be metabolized in the algal cell.
Many unicellular algae can be grown in the dark on two-carbon compounds such as acetate or ethanol as sources of both energy and carbon for growth. Chlorella grown in the dark on acetate, incorporates [14 C]-acetate into protein and carbohydrate without prior degradation while simultaneously about half of the acetate taken up is oxidized to CO2 in a manner consistent with the operation of the TCA cycle (Syrrett et al., 1964). These cells have high levels of malate synthetase and isocitrate lyase, thereby allowing the formation, from acetate, of four-carbon compounds which can form precursors of amino acids and hexoses (Syrrett et al., 1963). Autotrophically-grown cells however have little isocitrate lyase activity and [14 C]-acetate supplied to these cells in the dark is oxidized to CO2 . Furthermore the activity of isocitrate lyase increases when autotrophically-grown cells are supplied with acetate in the dark and no cell division takes place during this time. Similar increases in malate synthetase and isocitrate lyase in response to growth on two-carbon compounds have been reported in Euglena gracilis.
The enzymes of both the glycollate pathway and the glyoxyllate cycle have therefore been found in several algae and the question arises as to whether these enzymes are compartmentalized in a microbody. The limited number of studies which have been done suggest, on the basis of their enzyme activities, that microbodies isolated from algae have either a glyoxysomal function or are of the non-specialized type often present in non-green plant tissues (Huang & Beevers, 1971). In Euglena gracilis grown on ethanol the clyoxylate cycle enzymes malate synthetase and isocitrate lyase together with glycollate dehydrogenase and glyoxyllate reductase were detected in a particulate fraction distinct from mitochondria and, while no catalase activity was detected, the enzyme complement indicates a microbody with a glyoxysomal function (Graves et al., 1972). However in another flagellate, Chlorogonium elongatum, grown either on acetate in the dark or autotrophically in the light, a catalase-containing microbody was found which had neither glyoxysomal nor peroxisomal enzymes associated with it. In the acetate-grown cells isocitrate lyase and malate synthetase were found to be located in the cytosol (Stabenau & Beevers, 1974), while in autotrophically-grown cells, glycollate dehydrogenase, hydroxypyruvate reductase and glyoxyllate-glutamate aminotransferase were2 found to be localized in a mitochondrial fraction together with cytochrome oxidase, and malate and isocitrate dehydrogenases (Stabenau, 1974).
It is apparent that algal microbodies do not have a peroxisomal function and that this is correlated with the oxidation of glycollate in these cells by glycollate dehydrogenase, thus eliminating the requirement for the compartmentalization of glycollate oxidation with catalase. However, microbodies have been isolated from only a few algae primarily because vigorous methods are required to break algal cell walls and microbody isolation is therefore difficult to achieve. It remains to be seen whether peroxisomes occur in algae such as Nitella and Spirogyra which have relatively high levels of catalase and also have glycollate oxidase rather than glycollate dehydrogenase.
Further Reading
Beevers H. (1969) Glyoxysomes of castor bean endosperm and their relation to gluconeogenesis. Ann. N.Y. Acad. Sci.168, 313–24.
Gibbs M. (1969) Photorespiration, Warburg effect and glycolate. Ann. N. Y. Acad. Sci.168, 356–68.
Jackson W.A. & Volk R.J. (1970) Photorespiration. Ann. Rev. Plant Physiol.21, 385–432.
Tolbert N.E. (1971) Microbodies—peroxisomes and glyoxysomes. Ann. Rev. Plant Physiol.22, 45–74.
Vigil E.L. (1973) Structure and function of plant microbodies. Sub-Cell. Biochem.2, 237–85.
Zelitch I. (1971) Photosynthesis, Photorespiration and Plant Productivity. pp. 352. Academic Press, New York.