5.6.1.5—
Cytochrome b
The cytochromes are hemo-proteins. Three classes of cytochromes, distinguished by their spectral properties, as well as by the nature of their prosthetic groups are found in mitochondria (see Fig. 5.4). The a -cytochrome contains as its prosthetic group, heme a , while the b- and c -cytochromes contain a heme closely related to protoporphyrin IX. In the c -cytochromes, the heme is covalently linked to the protein via sulphur atoms in a thio-ether linkage. In the reduced state, the cytochromes exhibit strong absorption bands in the visible region of the spectrum which have been useful in their identification and in the analysis of their function. In addition, both the oxidized and reduced forms absorb strongly in the region around 400 nm, which is a characteristic of all heme compounds.
The b -cytochromes are best resolved when their spectra are determined at low temperatures, e.g., 77°K. Three b -cytochromes have been identified and two others are suggested. Their spectral properties are summarized in Table 5.3.
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||
Considerable variability exists in the nomenclature of the b -cytochromes. In conformity to the International Union of Biochemistry, the b -cytochromes are designated according to the a -peak of their reduced spectrum at room temperature (25°C). It should be noted that there is a blue shift of about 3 nm in the spectrum at 77°K relative to the spectrum at room temperature. The use of the a -absorption peak is further complicated by the fact that some authors use the absorption maximum at 77°K to designate the various b -cytochromes. In older nomenclature, mammalian cytochrome b –562, as orginally described by Keilin, was designated cytochrome b. As other b -cytochromes were discovered with an a -absorption peak significantly different from 562 nm, these were designated with subscripts. More recently, cytochrome b– 566 was thought to be directly involved in energy transduction and was designated cytochrome bT , a transducing b -cytochrome, to differentiate it from cytochrome b– 562, or bK .
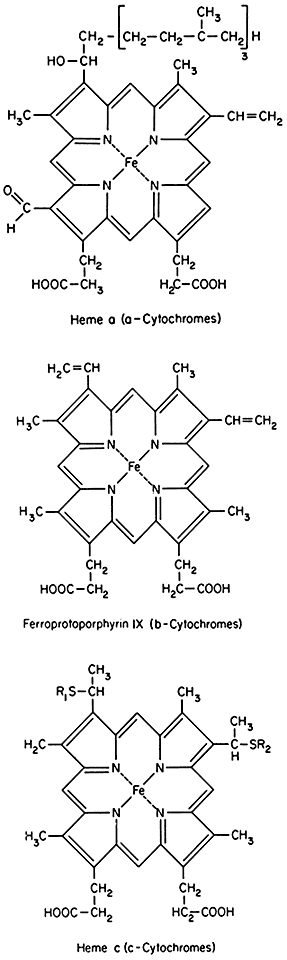
Figure 5.4
Prosthetic groups of the cytochromes.
The multiplicity of b -cytochromes is due to different b -cytochromes in mitochondria rather than a splitting of the absorption bands at low temperature, since the peak heights do not change in synchrony in the presence of reducing agents, inhibitors or uncouplers (Lance & Bonner, 1968). The b -cytochromes are placed in the respiratory chain according to the following sequence (Storey, 1973):
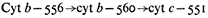
Cytochrome b– 560 (557) was placed on the oxygen side of cytochrome b –556 (553) as a result of the determination of the rates of oxidation of the 556 and 560 components by an oxygen pulse of an anaerobic suspension of mitochondria. The 560 component was oxidized with a half-time of oxidation of 6 to 8 msec while the 556 component was oxidized with a half-time of 150 to 200 msec. The reduction by succinate of these two components in anaerobiosis showed, however, that b– 560 was reduced more slowly than b– 556 which was contrary to the expected rates in view of the rates of oxidation (Storey & Bahr, 1972; Storey, 1973). The slow reduction was ascribed to the more negative redox potential of b –560. The midpoint potentials of b –560 and b –556 would predict that b– 560 would be on the substrate side of b –556. Further resolution of the sequence of the b -cytochromes is necessary.
Cytochrome b– 566 was thought to be analogous to the b– 566 (bT ) of mammalian mitochondria. Cytochrome b– 566 from animal mitochondria was found to undergo a midpoint potential shift as well as an enhanced reduction in anaerobic suspension when the respiratory chain was energized (Chance et al., 1970). This was interpreted as the formation of a high energy intermediate of phosphorylation directly involving cytochrome b– 566. In plant mitochondria, the midpoint potential shift of b– 566 was not observed (Dutton & Storey, 1971; Lambowitz et al., 1974). Although enhanced reduction of b– 566 by ATP or by energization of the respiratory chain could be demonstrated in plant mitochondria, it could be explicable by reverse electron flow through the b -cytochromes (Lambowitz et al., 1974; Lambowitz & Bonner, 1974). Thus the status of a transducing b -cytochrome in plant mitochondria is in question. In fact, cytochrome b –566 was excluded from the main sequence of the respiratory chain, since it remains oxidized in anaerobic suspensions (succinate reduced) while other b -components, pyridine nucleotides and fluorescent flavoproteins are reduced (Storey, 1969, 1974). There was a lack of equilibration between the low redox potential carriers with cytochrome b– 566. The function of cytochrome b –566 is left uncertain.