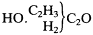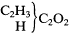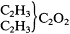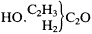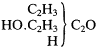8—
Carbonic Acid and Natural Types
While Kekulé was developing his ideas on carbon tetravalence and the use of that concept in formulating molecular skeletons from valence-linked carbon atoms, Kolbe and Frankland were traveling a partially independent path that led to similar ideas. Influenced by electrochemical instincts, these chemists did not develop the second element of structure theory (carbon linking) in the manner that Kekulé did, and so their vision of a general theory of organic "constitutions" looks somewhat different. Both the similarities and the differences between the Kekulé and the Frankland-Kolbe versions of what came to be known as structure theory provide insight into the development of this fundamental set of ideas.
Frankland and the Origins of the Carbonic Acid Theory
Edward Frankland's personal odyssey following the period of his direct association with Kolbe comprised a stay at Giessen, then professorial appointments at Putney College, Owens College (later the University of Manchester), and St. Bartholomew's Hospital, London, before he became Faraday's successor at the Royal Institution in 1863.[1] During this period, his scientific work exhibits a curious blend of old and new, of elements of the reform movement commingled with vestiges of Berzelian electrochemistry.
Frankland's first paper after leaving Marburg and the Kolbe-Bunsen orbit (read to the Chemical Society in November 1849)
announced his discovery of the zinc alkyls, and he placed this work explicitly and repeatedly in the type theory tradition of Dumas, Gerhardt, Wurtz, Paul Thenard, and Hofmann.[2] He was convinced, and rightly so, that he had discovered an invaluable tool by which he could effect substitutions that were in a sense inverse of the traditional ones. Rather than replacing hydrogen atoms with halogen atoms, the intensely electropositive zinc methyl and zinc ethyl might be used to replace halogen or oxygen atoms with methyl or ethyl radicals, in a manner analogous to the way metallic zinc replaced iodine in the formation of zinc alkyls. He laid out this research plan first in an unobtrusive passage in his long paper published in the spring of 1852.[3] As early as 1851, as he later told Kolbe, he sought to realize this plan by trying to react zinc ethyl with phosgene or carbon tetrachloride.[4] But these experiments were not successful. The first chemist self-consciously to succeed in employing metals to alkylate organic compounds was Wurtz (1855), following a route suggested by Hofmann and Brodie (1850), who were themselves influenced by the work of both Williamson and Frankland.[5]
The theoretical conclusion of Frankland's 1852 paper stated a restricted form of the regularity since known as valence. Valence was intrinsically supportive of types and subversive to electrochemical theory since, in Frankland's words, the "combining power" of an atom "is always satisfied by the same number" of other atoms, independent of electrochemical properties. But Frankland also pointed out that the extreme version of type theory, which attempted to account for chemical properties solely on the basis of the arrangement of the atoms in the molecule (that is, by assignment of compounds to immutable types) is likewise untenable. For example, it is impossible to account for the difference in properties between antimony trioxide and antimony trimethyl, or between arsenic acid (AsO5 ) and cacodylic acid (AsO3 Me2 ), without considering the electrochemical properties of the components. Gerhardt came to a similar conclusion the following year: in replacing the hydrogen atoms of ammonia successively by three acetyl groups, a dramatic alteration of properties was apparent.[6]
Due to the difficulties attached to working with the particular substance Frankland had chosen, he continued to have little success in pursuing the promising program outlined in his 1852 paper. A full three years later he reported his subsequent work on zinc ethyl to the Royal Society and to the British Association for the Advancement of Science, but he still indicated only in outline form his future intent to attempt systematic alkylations of organic halides and oxides by means of zinc methyl and zinc ethyl. Kolbe mentioned in a footnote a few years later that Frankland had reported to him in a letter from this period that he
had synthesized two mercaptans by reacting zinc alkyls with carbon disulfide. For some reason, Frankland never published this result; he did, however, later affirm that he had tried to alkylate carbonic acid with zinc alkyls.[7]
In a paper of June 1856 Frankland announced the research program for a fourth time, but once again publicly temporized.[8] He fully recognized the high promise of this plan; if he were successful, it would enable him to "ascend the homologous series of organic bodies," thereby attaining "clearer views of [their] rational constitution."[9] In both his collected papers and his autobiography he said that he knew that it was "an obvious and easy step" to apply valence ideas to carbon and that during this period he continued to try without success to react zinc alkyls with carbonyl chloride and carbonic acid. He noted that James Wanklyn achieved the experimental success, and Kekulé the theoretical success which he had been seeking.[10] In summary, from 1851 on Frankland consistently attempted to develop reactions that could exemplify valence theory as applied to carbon.
By 1855 he had drawn even closer to the thinking of the "typists," which is indicated by his noting a variety of new electrochemical anomalies, citing with approval an 1850 paper by Brodie, and suggesting that zinc ethyl "appears to belong to the so-called water type, and to consist of two volumes of ethyl and one volume of zinc vapour . . ." In 1857 both Kekulé and Baeyer cited this statement as further empirical evidence for "diatomic" elements, in this case zinc. But Frankland continued to formulate zinc ethyl as ZnC4 H5 (i.e., a half-molecule of the compound according to the reformed chemistry), and there is no indication that he had become convinced of Laurent's and Gerhardt's central thesis that formulas should consistently express volume relationships.[11] Frankland, like his friend Kolbe, remained convinced
. . .that, although all the electro-chemical theories hitherto proposed were far from satisfactory, yet, that amongst the factors of chemical action, the electrical character of elements could not be denied a place, without ignoring and leaving unexplained some of the most remarkable of chemical phenomena.[12]
Kolbe remained a more thoroughgoing electrochemist longer than did Frankland. Chapter 6 treated Kolbe's tentative rejection of Frank-land's valence thesis in the first fascicle of his textbook (published in June 1854 concurrently with Frankland's Friday Evening Discourse at the Royal Institution just cited), on the basis of its apparent violation of electrochemical tenets. Frankland and Kolbe later stated, separately and publicly, that through correspondence during the year 1856 Frank-
land succeeded in persuading Kolbe of the truth of the "law of maximum combining capacities."[13] Unfortunately, this correspondence has not survived.
There is evidence that Kolbe was at least partially converted to Frankland's way of thinking as early as the end of 1855. In the article "Radicals" for the Handwörterbuch , completed by the first week of 1856, Kolbe discussed Frankland's law of combining capacities, which he now seemed to accept, as applied to carbon. He formulated acetic acid as "methyl carbonic acid," wherein one methyl radical substitutes for one of the two replaceable oxygens of the "dibasic" carbonic acid:
|
He then offered the "conjecture" that the C2 of carbonic acid might be able to suffer a further substitution of methyl for oxygen, which would result in a compound that "would have the composition of acetone." The article shows signs of persistence of earlier ideas, as the concept of copulas was still strongly represented.[14]
Kolbe developed this approach further in a short sole-authored theoretical article in the Annalen der Chemie , written in December 1856 and published in March 1857, which he ever afterward considered to be the first statement of his "carbonic acid theory." In addition to deriving the homologous fatty acids and acetone schematically by substitution of the two replaceable oxygens of carbonic acid with hydrocarbon radicals, he noted that aldehydes could also be related to the same type by replacing one of the methyl groups of acetone by a hydrogen atom. A final speculation suggests the possibility that substitution of a third oxygen of carbonic acid by alcohol radicals might provide the rational composition of the homologous alcohols and ethers.[15]
When this paper appeared, Frankland thought that Kolbe had violated a private trust by publishing in his name alone. Considering the circumstances just described and the discussion that follows, it is likely that Frankland had suggested the leading idea of this theory in a letter to Kolbe in the summer or autumn of 1856, in the course of mar-shaling all possible arguments to persuade his friend of the truth of the valence law. Given the content of Kolbe's article on radicals written at the beginning of that year, it is clear that his mind must have been prepared for conversion to the general case, but it also appears that he had not yet forsaken copulas.
The story cannot be fully sorted out from existing documents, but several hitherto unpublished letters shed some light on the matter.
After Kolbe's paper appeared, Frankland refused to write to his friend for five years.[16] When Kolbe finally succeeded in prevailing on Frank-land to tell him what had precipitated the break, Kolbe wrote two long letters of exoneration.[17] We have Frankland's response to the first of these. "I am of the opinion," Kolbe wrote to Frankland,
that the foundation of the recent development of chemistry, and especially of our carbonic acid theory, is the truth, first expressed and brought to the attention of chemists by you, that the affinity of elementary radicals is always satisfied by the same number of atoms, regardless of their chemical character. From the instant I correctly understood the sense of your perception it was immediately clear to me that the organic dithionic acids are nothing more than sulfuric acid in which one of the six oxygen atoms is substituted by an alcohol radical, and just as quickly both of us became convinced that fatty acids, acetones, and aldehydes must arise through replacement of oxygen atoms in carbonic acid by alcohol radicals. We immediately agreed to work in collaboration to confirm this hypothesis experimentally by means of your zinc ethyl . . . You undertook to treat carbon [tetra]chloride and carbon [di]sulfide, and I phosgene and carbonic acid, with zinc ethyl.[18]
Kolbe reminded Frankland that after he had come to realize the truth of the law, he and Frankland had successfully worked through all their remaining differences and formulated the carbonic acid theory. Kolbe had written Frankland urging an immediate joint theoretical article outlining the rationale for the experimental program. Frankland had demurred, Kolbe reminded him, saying it was not customary in England to publish purely theoretical papers. But a paper by Piria shortly thereafter (October 1856)[19] made clear that they were close to being forestalled. Thus, in December Kolbe wrote out the eight-page theoretical paper and omitted Frankland's name (he continued), out of respect for Frankland's wishes. He did, however, send the manuscript to his friend, with the hope, he said, that Frankland might still be induced to append his name to it. He quoted directly from Frankland's reply of 18 January 1857—which does not appear to have survived—in which Frankland stated that he "sufficiently agree[d] with what you have said," that their views were "fundamentally the same," and that it was "the highest time, that something should appear from us on the subject."
Frankland appears to have intended these phrases as a clear request for joint authorship,[20] but apparently Kolbe did not interpret them as such. In any case, Kolbe had already made a firm decision in favor of single authorship more than two weeks before he received Frankland's reply (but he did not tell Frankland this!), for on 4 January 1857 we find Kolbe writing Vieweg:
. . . it is a joy to be able to say to you that I have now come to a clear understanding of the important theoretical questions with which I have so long been continuously occupied, and which are so important for the presentation of the subject in my Organic [textbook]. I have just sent off a paper concerning this for publication in the Annalen , and am now proceeding with redoubled pleasure and energy on the continuation of my Organic, which, if I stay healthy, should move rapidly forward.[21]
The first few pages of the published paper use the pronoun "ich" and refer to Frankland by name. Kolbe then changed to "wir," doubtless intending thereby to refer to both of them, but most readers probably inferred the editorial "we" and concluded that inconsistent editing accounted for the change to the plural pronoun.
The matter was of the highest importance to Frankland. Characteristically modest and extremely reticent about priority claims for his whole life, he thought enough about the theory and his co-authorship of it to sever relations with a good friend to whom he owed a great deal:
. . . what I complain of is, that owing to your having in this last memoir [of 1860] ignored my participation in the views of the previous memoir [of 1857] you have practically claimed & obtained the exclusive authorship of the Carbonic Acid & carbonic oxide types. I do not believe that you intended this, but such has been the result, &, as I attach much value to the theory, you will easily conceive that the present position of the matter is unsatisfactory to me & I should wish some way to be devised for setting it right.
Frankland suggested the means of "setting it right" might be a paper by Kolbe alone or by both of them, regarding the historical development of the theory.[22]
Kolbe tried to satisfy Frankland by writing an historical introduction to a volume of his collected papers published three years later. He declared there that the 1857 paper had been joint with Frankland; moreover, "Frankland had a large share, indeed much larger than is generally known, in the formulation of this hypothesis [of deriving organic compounds from carbonic acid]." The germ of the idea lay in Frankland's theory of atomicity of elements. Kolbe wrote
The unclear conception of the mode in which the so-called copulas were chemically combined was a great weakness in [my] hypothesis of copulated radicals. It is Frankland's merit to have been the first to throw light upon this, and thereby to have completely done away with the idea of copulation, by recognizing the fact that the various elements possess definite saturation capacities (atomicities).[23]
Although their correspondence was reestablished after Kolbe's self-exoneration, their relationship was never really warm thereafter, Frankland describing it as "more a scientific than a social friendship."[24] Frankland always considered Kolbe's 1857 paper as co-authored; it appears that way in the edition of his collected papers, Frankland noting editorially that his name had been omitted by "inadvertence."[25] Kolbe's son-in-law Ernst von Meyer agreed in his history of chemistry that this article was "joint" with Frankland.[26]
The affair was no less important to Kolbe, who really did begin thinking of the paper as his own, as is clear from his letter to Vieweg of 4 January 1857 and passages in his papers and textbook after 1857. His monographic paper of 1860 refers to the 1857 paper unambiguously as his alone. This was what was so galling to Frankland and may have led the latter, as we shall see, pointedly to ignore Kolbe in his own review article of 1860. Despite his clear public declaration of 1865, Kolbe was still referring to the paper as his in 1881.[27] Kolbe, however, did recognize Frankland's crucial role, publicly as well as privately, in the creation of the carbonic acid theory. Even if Frankland's name did not appear in the byline, Kolbe gave Frankland extensive credit within the 1857 paper and was absolutely clear in his letters to Frankland concerning his conviction of Frankland's necessary role.
Kolbe was fully cognizant of the importance of the idea sketched in the paper. Within days of its publication, he proclaimed to Vieweg a "small revolution" in organic chemistry, and if anything, he was understating the matter.[28] However, with the theoretical details and research prospectus now public, Kolbe initially had just as little success at obtaining experimental verification as Frankland. Not only were organozinc compounds difficult to work with, but Kolbe became seriously ill shortly after sending his paper off at the beginning of January 1857, and remained a semi-invalid for seventeen months. Wurtz had already forestalled one element of Frankland's program by "ascending the homologous series of organic bodies" using alkyl iodides and a metal (sodium, not zinc).
Early in 1858 news came of an even more direct application of the theory. James Wanklyn, Frankland's assistant at Owens College in 1856 and a fellow Lancastrian, went the next year to Heidelberg on Frankland's recommendation to study with Bunsen, and there attempted to synthesize new organometallic compounds, especially sodium and potassium alkyls. He succeeded in this goal, although he was unable to isolate the compounds in pure form. Even more interesting, he found that the "affinity" of sodium ethyl was so powerful that it attacked carbonic acid (anhydride) itself and transformed it into sodium propionate.[29] Here was a dramatic realization of the carbonic
acid theory, for it appeared to represent a direct substitution of ethyl into carbonic acid. On 8 March 1858, Frankland wrote a gracious letter congratulating his former assistant.
I am delighted to hear of your success with the Propionic Acid experiment. It is to me as you may imagine of peculiar interest & importance on account of its affording such a beautiful & satisfactory proof of the correctness of the views . . . expressed by Dr. Kolbe & myself in Liebig's Annalen more than a year ago.[30]
Kolbe was less happy. He had just succeeded in obtaining propionic acid, unfortunately in only trace amounts, by digesting potassium carbonate with zinc ethyl in ether, but he had not had time to publish when he got the news of Wanklyn's discovery.[31] He immediately wrote to Bunsen, accusing Wanklyn of treachery in poaching on his and Frankland's scientific territory. Bunsen replied with a friendly but strongly worded letter defending Wanklyn's integrity, and he enclosed Frankland's letter to Wanklyn as further evidence.[32] Kolbe let the matter rest, but he was not convinced. In his view, Wanklyn had "skimmed the cream" off of his and Frankland's discovery.[33] Frank-land may also have had later second thoughts, for after another clash in 1867 he consistently opposed Wanklyn's further professional advancement, and with considerable success.[34]
Already aggrieved by Kolbe's action of a year earlier, Wanklyn's paper was a second blow for Frankland (a third, if Wurtz' 1855 research is to be counted). Frankland decided at this time to lay out his own ideas more explicitly. At a Friday Evening Discourse on 28 May 1858, Frankland presented in type formulas the elements of the carbonic acid theory, which he stated to his audience—as he had to Wanklyn—that he had published jointly with Kolbe in the Annalen article of March 1857. One and two methyl radicals substituted for one and two oxyen atoms of carbonic acid (C2 O4 ) yield acetic acid and acetone, respectively, and a methyl and a hydrogen atom yield aldehyde. Furthermore, two methyls substituted for two hydrogens of methyl hydride (marsh gas) produce the new substance methyl-ethyl, and one methyl and one ethyl radical substituted into marsh gas result in the substance he himself had first isolated by chemical means in 1849—"ethyl" gas. He thereby illustrated what he had meant to say in published papers since 1852 by his intention to "ascend the series of homologous organic bodies," although he was still lacking the experiments to demonstrate the idea.[35] He sent Kolbe an offprint of this paper.
Once again, this paper represents an intimate amalgamation of old and new. The type formulas, substitution mechanisms, and hydrocar-
bon formulations are entirely in the spirit of the reformed chemistry, even if Frankland still retained the double carbon atom. He now even seemed to be explicitly conceding that ethyl gas was twice the size of the ethyl radical. However, the carbonic acid formulations could only be understood by reference to Berzelian dualistic formulas, with one and two unspecified water molecules (actually half-molecules, OH, where O = 8) understood to be present for monobasic and dibasic acids, respectively. The schematic clarity of matching four oxygens, rather than two, in carbonic acid, to the four hydrogens of marsh gas must have seemed to be decisive in Frankland's mind, counterbalancing what must have seemed to others a greater inconsistency. His amphibious theoretical position may have been as important as the experimental difficulties in holding Frankland back from rapid exploitation of the new ideas.
Frankland read a long review article "On Organo-Metallic Bodies" to the Chemical Society on 7 June 1860, a section of which further illustrates the application of his 1852 valence law to carbon compounds. For the fifth time, he indicated his intention to substitute alkyl radicals for chlorine in alkyl chlorides; experiments, he said once more, were in progress. But he noted that Wanklyn had already shown experimentally the tenability of the theory as applied to carbonic acid, and the theory could apply as well to aldehydes, alcohols, glycol, and glycerin. He did not even mention Kolbe. He wrote
It would be greatly easy to extend this view of the constitution of organic carbon compounds; but the above examples are sufficient to indicate its general application somewhat more fully than I have previously done [here he cited Kolbe's Annalen paper—as if it were his own—and his 1858 Royal Institution talk], and more than this is not desirable until the hypothesis has been further supported by experimental results.
Curiously, he included neither this nor the 1858 Royal Institution article in his collected papers.[36]
Frankland's formulas suggest that he accepted the molecular magnitudes argument from the Williamson asymmetric syntheses, for he again depicted ethyl as a dimer, water as
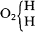
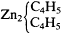
carbon atoms (for the reformers themselves sometimes used these for various reasons), but from the fact that he thought nothing of separating the individual equivalents of oxygen or zinc in certain formulas. For Frankland, these equivalents must have been the same as chemical atoms. Thus, despite his status as one of the discoverers of the law of valence, he had not yet made a fully consistent distinction between the concepts of valence and equivalence, which the reformers were so careful to specify and which was so fundamental to their movement. This failure was a result of his continued adherence to the use, and ontological atomistic interpretation, of conventional equivalents, which tend to mask the phenomenon of valence.
Kolbe's Development of the Theory
In the carbonic acid theory, Kolbe had essentially come to accept Frankland's type-theoretical formulation of organic compounds. His reorientation toward types corresponded to a peaking in his feeling of respect toward Gerhardt. In chapter 6 it was noted that Kolbe regarded Gerhardt's Traité as a very good book in many respects; he used it heavily in writing his own book and was astounded at Gerhardt's rate of production of high-quality manuscript. Shortly before Gerhardt's death, he wrote Vieweg, "I will no longer oppose Gerhardt. I fully recognize his accomplishments; his corrupt theoretical speculations will soon be outlived, and he will be the first to ignore them." And again, in 1858, he stated that he now gave Gerhardt "my full recognition of his services to organic chemistry."[38] Having fully embraced types in the publication of his carbonic acid manifesto, Kolbe set about to treat the theory systematically throughout his textbook.
Consistency was a serious problem. He had already been working on the book for several years, and by the time of his conversion to Frankland's views, he had published five fascicles in three major installments. Kolbe suffered from the circumstance that he was producing a detailed and strongly theory-based survey of the field at a time when the theory was evolving with astonishing speed. Consequently, each installment of the book contained different and sometimes inconsistent theoretical details. That he himself was so theoretically inventive, and honest with his readers about the ideas and hypotheses that he was using, exacerbated the problem of internal consistency. As he wrote Vieweg about the time that the first volume was completed (1859),
In the last four years organic chemistry has found itself in a stage of development in which only just now a crisis has arisen. In composing my textbook, I had three possible paths before me. I could write the text without any particular system as a foundation and without having any firm theoretical viewpoint, which is the situation in which Wöhler and Liebig find themselves; or I could follow Gerhardt's swindle, which even the best minds have done . . . or, finally, I needed to follow my own path, which I must tread all the more cautiously and circumspectly, since everyone is watching me extremely closely in order to find and breach every weak point in my system. . . . Be assured, dear Vieweg, that that which has hitherto failed to recommend my book, that it did not follow the usual accepted patterns and took up an independent position, will be viewed very soon as an advantage, and will transform its current enemies into friends. In this regard I look forward to the near future, confident of victory.[39]
In the summer or autumn of 1857, feeling the need to treat the carbonic acid theory as soon as possible in his text, he wrote a section entitled "Theoretical Views Concerning the Composition of Alcohols, Acetones, Aldehydes and Related Acids," and he fit it into the fourth installment, double fascicle 6/7, somewhat out of place.[40] Production of this manuscript was agonizingly slow, however, due to his recurrent rheumatic fever. Vieweg was losing patience; the book was not selling well for several reasons: its slow appearance, its idiosyncratic and polemical characteristics, and its insecure and shifting theoretical base. "I assure you," Kolbe responded, "that I have for a long time been in the clear regarding all theoretical problems whose solution was earlier so important."
And do not think that in the description of organic compounds I see myself in strict opposition to the various prevailing viewpoints. To be sure, I place my views, which I regard as more correct, in the foreground, and in doing so I believe I bring much that is good and new; but I consider also the viewpoints of others as well. Aside from a few chapters, no one will find anything in my book to be partisan. On the contrary, I am the conservative, who only rejects eccentricities and abnormalities.[41]
The installment was finally published in December 1857.[42] Kolbe was extremely anxious to see it in print. As he wrote Vieweg on 20 December,
The double fascicle appearing now contains a great deal that is new, and I think that several chapters will capture the interest of readers, especially the small section "theoretical views on the constitution of alcohols etc."
Even if many chemists will not yet at present accommodate themselves to these views, I am absolutely convinced that the theory will gain rather general currency in the very near future.[43]
The theoretical section of the fascicle announced some dramatic departures for Kolbe. No longer did he use Otto's buckle symbol to indicate conjugation, nor any symbol at all, although he still employed the term. No longer did he make any meaningful distinction between his two classes of organic radicals—the ether or alcohol radicals in hydrocarbons (and their derivatives) and the conjugate radicals in acids (and their derivatives). He permitted himself the use of "the currently popular expression" of "types" and described both carbonic oxide C2 O2 and carbonic acid (C2 O2 )O2 as "diatomic" (zweiatomig)—a term that had been in use for only eighteen months among type theorists such as Gerhardt, Wurtz, and Kekulé. Alcohols were now formulated in an analogous fashion to aldehydes, acetones, and acids:
|
Hydrogens and methyls simply substitute for oxygens in acetic acid, and ultimately, oxygens in carbonic acid, to produce all of these compounds. Carbonic acid is diatomic because two oxygens are more loosely bound and more readily replaced than the other two, just as one of the two oxygens may be readily replaced in acetic acid (as in the formation of amides or thiacetic acid). Somewhat inconsistently, he also referred to carbonic oxide as diatomic because two atoms (e.g. of oxygen or of chlorine) can add to it.
Kolbe attempted to show that his theory had heuristic power by making several predictions. There is no reason why methyl radicals should not be induced to continue substituting for the two remaining hydrogens of ethyl alcohol, he stated, so the following compounds should be possible:
|
If these latter two substances were produced, they would have to be called "pseudoalcohols," since Kolbe predicted from the theory that they would not share the property common to all other known (aliphatic) alcohols of being oxidizable to aldehydes and acids. The first would be isomeric with propyl alcohol (from propionic acid), and would oxidize only to acetone. The second would be an isomer of butyl alcohol (from butyric acid), and would suffer no oxidation at all (without decomposition). These predictions were derived by implicitly applying the concept of tetratomicity to the carbonyl carbon group (C2 ), although that idea was neither defined nor named here.
Other predictions concerned the recent discovery of glycol, which Wurtz had proclaimed to be a dialcohol. Kolbe objected to Wurtz' categorization since Wurtz had failed to demonstrate the oxidation of glycol to an aldehyde or acid—again, the distinguishing characteristic of alcohols. Kolbe thought that partial oxidation of glycol would not in fact yield an aldehyde; rather, either acetic anhydride or a homolog of glycerin would result:
|
This prediction was based on Kolbe's supposition that glycol was merely a hydrated form of aldehyde, C4 H4 O2 . He conjectured that it ought to be possible to produce glycol directly, by applying hydrating agents to aldehyde. In this section he did not fully justify his preference for considering glycol as an oxide rather than a dialcohol. His real reasons were detailed only in the next installment, published a year later, and are discussed below.
Glycerin is also no alcohol, according to Kolbe, but rather the trihydrate of a tribasic oxide, C6 H5 O3 .3HO. Acrolein is not the aldehyde of glycerin but of allyl alcohol. Oddly, Kolbe continued, the basic oxide of glycerin, C6 H5 O3 , has the same composition as propionic acid. Although, like all organic acids, propionic acid has one oxygen atom bound more loosely than the other two, perhaps glycerin possesses the same hydrocarbon radical but with the three oxygens bound in a different fashion, that is, in a chemically equivalent way. This would explain why propionic acid is monobasic, while glycerin is tribasic.
Before his next installment appeared, he needed to take account of all the new research that had appeared in the eventful years 1857 and 1858, and once more this occasioned some important modifications in his ideas. About the middle of June 1858 he returned from a Wiesbaden cure, full of energy and feeling "newly reborn" after his long
and serious illness.[44] After the summer semester ended, he was able to make rapid progress. Indeed, as early as 11 August 1858, he presented a paper to a local scientific society that contained a precis of the theoretical content of the Lehrbuch installment that he was then only starting to compose.[45] Kolbe persuaded Vieweg to cancel sheet number 42 (the last sheet of fascicle 7, already published), so that changes could be made in it. The double fascicle 8/9, including a revised sheet 42,[46] was complete in manuscript form by the end of 1858 and was published very early in 1859.[47]
Chief among the new published work that he needed to deal with was that of Wurtz, Debus, and Kekulé. Wurtz' further work with glycol derivatives and other polyfunctional alcohols and acids was especially important. Wurtz was then in his prime, publishing during this period important articles at the rate of one every six weeks or so, filled with fascinating new substances and all used to develop the theory of polyatomic radicals. Debus had succeeded in oxidizing alcohol into a double aldehyde (glyoxal), an aldehyde-acid (glyoxylic acid), and a hydroxy-acid (glycolic acid). Wurtz had shown that glycol could be oxidized to glycolic and oxalic acids, which constituted Kolbe's own criterion of alcohol character and thus should have satisfied him that glycol was a dialcohol.[48] Since glycol was derived from ethylene, a reaction route from ethylene to oxalic acid was now apparent; Debus' reactions showed that alcohol, too, could be transformed into oxalic acid. Because all agreed that ethylene and alcohol had two carbon' atoms (four carbon equivalents), it could hardly be doubted any longer that oxalic acid also had two carbon atoms rather than one and therefore was dibasic.
Kolbe now accepted this conclusion as experimentally demonstrated, but he still resisted characterizing glycol as a dialcohol just because it could be oxidized to an acid. Glycol does not share other (unnamed) properties of alcohols; moreover, there are theoretical reasons for formulating glycol as an oxide hydrate rather than as a dialcohol. He had decided that ethylene must be carbonyl (C2 ) united with both a methyl group (C2 H3 ) and a hydrogen atom, since it is derived from the carbonic oxide type and so their constitutions must be analogous. The reason ethylene is analogous to carbonic oxide is that both compounds undergo similar addition reactions, such as with chlorine. Because the two chlorine atoms both add to the same carbonyl radical (carbon atom) in carbonic oxide (there is only one), they must also add to a single carbonyl radical in ethylene. So ethylene must be methy carbonyl hydride. Since glycol is derived chemically from ethylene, it must be ethylene oxide hydrate:
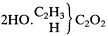
Oxidation of this substance is not analogous to oxidation of ethyl alcohol to acetic acid, Kolbe thought, since it has only one lone hydrogen replaceable by oxygen, while ethyl alcohol must oxidize both its radical hydrogens to form acetic acid. Wurtz' glycol when oxidized should yield a homolog of glycerin (3HO.C4 H5 C2 O3 ), namely, 3HO.(C2 H3 )C2 ,O3 . The true alcohol derived from glycolic acid has not yet been synthesized, Kolbe argued; it would have the composition
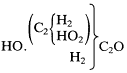
If this hypothetical substance were to be oxidized under mild conditions, it would yield a novel aldehyde isomeric with acetic acid. Kolbe thought that unhydrated ethylene oxide, not yet isolated, must be an isomer of normal aldehyde (and not, as he had suggested earlier, identical to it).[49] The difference in their natures is a consequence of a difference "in the molecular grouping of their components, in other words, in their constitutions."[50]
In effect, Kolbe was suggesting that there were two possible isomers possessing the elemental composition of glycol. One was Wurtz' compound, an oxide; the dialcohol was certainly possible, but it had not yet been synthesized. Of course, Wurtz had succeeded in oxidizing glycol to glycolic acid, but this offered no proof that these two compounds had analogous constitutions. Rather, a rearrangement must take place during the oxidation, in which a hydrogen of the methyl group is replaced by "hydrogen peroxide," HO2 (a modern hydroxyl group), while the carbonyl carbon is further oxidized. Kolbe cited Kekulé's recent paper, in which Kekulé had described hydrolyzing monochloroacetic acid to glycolic acid, to demonstrate that glycolic acid was simply acetic acid with a hydrogen peroxide group substituted for hydrogen, and not the acid derivative of glycol. He thereby summarily excluded the possiblity of Wurtz' point of view, namely, that it was both of these at once.[51]
As problematic as the supposition of all these hypothetical isomeric compounds was, Kolbe seemed untroubled. At least there was a certain consistency here. He asserted that Wurtz' derivation of glycol from ethylene also falsified one of his (Kolbe's) ideas, given emphasis earlier in his textbook and defended since 1850, namely, that ethylene
derivatives are all derived from the vinyl radical, C4 H5 , wherein the four carbon atoms are chemically equivalent. Instead, ethylene is a methyl group united along with a hydrogen atom to the double carbon atom, "carbonyl," whose direct oxidation (addition of two atoms of oxygen to carbonyl) results in aldehyde. Accordingly, there ought to be two distinct monochloroethylenes resulting from substitution of either the single hydrogen atom or one of the three methyl hydrogens.[52]
In this installment of his textbook, Kolbe found a place for a second explicitly theoretical chapter, entitled "Theoretical Considerations Regarding the Saturation Capacity of Simple and Compund Radicals."[53] It began: "Our views regarding the mode of composition of the elements in organic chemistry have received a significant expansion by the perception that, in addition to the customary monatomic radicals, there exist also diatomic, triatomic, and tetratomic radicals." Monatomic radicals include methyl, ethyl, and acetyl; diatomic radicals include methylene, ethylene, and carbonic oxide. Triatomic radicals include formyl (methine) and the hydrocarbon radical of acetic acid, and finally, "carbonyl" itself, C2 , is tetratomic. All of these facts can be derived from the last. Adding methyl to carbonyl reduces its atomicity to three, and three additional oxygens create acetic anhydride. Replacing the oxygen of acetic acid by alkyls or hydrogens creates aldehydes, acetones, and other acids. Carbonic acid, marsh gas, or carbon tetra-chloride represent the satisfaction of atomicities of carbonyl by four atoms of oxygen, hydrogen, or chlorine.[54]
Kolbe announced an important new chemical law in this context: the basicity of an acid, whether organic or inorganic, is always equal to the number of oxygen atoms outside the radical. In the following series, the radicals between the square brackets were regarded as stable groupings whose oxygen content did not affect basicity.
|
What mattered was the number of oxygen atoms outside these radicals, always matched by an equal number of water molecules (or, according to Wurtz' and Kekulé's notation, half-molecules) on the other side. Thus, Kolbe regarded nitric acid as the hydrated oxide of the monatomic radical NO4 , acetic acid as the hydrated methyl derivative of the diatomic carbonic oxide radical C2 O2 , and carbonic acid as the dihydrated dioxide of the same radical. The oxygen within the radical could be replaced without altering the basicity, as in the con-
version of acetic acid to alcohol (which still forms salts with strong bases). If extra-radical oxygen atoms are replaced, however, the basicity is reduced, as in the conversion of acetic acid to neutral amides.[55]
Finally, Kolbe argued that dibasic organic acids are naturally derived from a double carbonic acid type, with succinic acid being
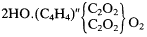
The C4 H4 has a methyl carbonyl hydride composition like ethylene. Organic sulfonic acids, based on the dibasic sulfuric acid type, must have anlogous constitutions, and Kolbe devoted a substantial section of the chapter to these compounds. They also can form dibasic acids upon a double type; for example, Buckton and Hofmann's new "di-sulfometholic acid" was 2HO.(C2 H2 )"(S2 O4 )2 "O2 . It ought to be possible, thought Kolbe, to add the elements of water HO to this acid by heating it in a sealed tube, thereby transforming it into a new tribasic acid, 3HO.(C2 H3 )(S2 O4 )2 "O3 .[56]
From October 1858, when Kolbe was nearing completion of the writing of this installment, until early January 1859 when it was in press and about to appear, he stressed in every letter to Vieweg the novelty, originality, and importance of these ideas. The installment was the "most difficult" of all to write, but its significance would soon be recognized; he had intentionally delayed publishing the material elsewhere to save it for the book. Still, he recognized explicitly that these theoretical advances were only made possible by incorporating the "striking" results of a number of important articles that had appeared in the journal literature during the past half year.[57]
It is hard to escape the conclusion that Kolbe must have been influenced by Kekulé's structure theory articles, which appeared in Liebig's Annalen in November 1857 and May 1858. There is no question that he always read each issue of the Annalen carefully. He cited in this installment another article of Kekulé's (on monochloroacetic acid) appearing in the March issue as supporting his viewpoint, and in 1860 he cited Kekulé's first structure theory paper—but only to criticize Kekulé's excessive use of mixed-type formulas.[58] Moreover, nothing indicates (and much suggests the contrary) that he routinely ignored type theory papers, even if he treated some of them dismissively and did not always understand them fully. By now, in any case, he was regularly using many other type theorists' work—indeed, he had become a type theorist himself. As was the case in Kekulé's structure theory papers, Kolbe laid stress on new research regarding the aliphatic sulfonic acids, behind much of which lay the work of Kolbe's close friends
Hofmann and Strecker and Kolbe's former assistant yon Uslar. A principal theme of Kekulé's two papers had been to destroy the copula theory, and for the first time Kolbe eliminated the word copula from his vocabulary. The development of the idea of atomicity in fascicle 8/9 has much in common with Kekulé's concept, including Kolbe's first explicit statement of the "tetratomic" character of carbon.
Moreover, Kekulé's definition of an organic radical as essentially the hydrocarbon skeleton of a molecule and all heteroatoms fully bound to it finds a parallel in Kolbe's new basicity law. Indeed, it is fascicle 8/9 that first announces Kolbe's conversion to the general concept of polybasic radicals and acids, a cardinal thesis of the reformed chemistry. Williamson had been the first to argue that the basicity of a radical confers an equal basicity on an acid derived from it, such that the dibasic sulfuryl, carbonic oxide, and oxalic radicals all result in dibasic acids. Kekulé had transformed this idea, using Liebig's and Laurent's vocabulary and his own structure theory, into the phrase "inside or outside the radical," which Kolbe followed. And, in a final irony, Kolbe's basicity law was essentially that proposed by Laurent in 1836, a law that Kolbe had particularly singled out for sarcasm and scorn in the first installment of his textbook!
The first volume of Kolbe's textbook required just one more fascicle for completion, along with an addendum updating the work from the journal literature that had appeared during the five years it had taken for the volume to be published. It was finished by the end of July 1859 and finally appeared that autumn. At this point he decided to write a summary of the modified carbonic acid theory for publication in the Annalen , since the number of readers of his textbook was very small compared to the readership of Liebig's journal. Few in the German chemical community were even aware, much less approved of, the details of his theory before 1860, and he lamented this feeling of isolation to his friend Vieweg. Once the paper was shipped off, he wrote with badly mixed metaphors that he was "now looking forward, sitting fast in my saddle, to the coming storms."[59]
The paper was entitled "On the Natural Connection of Organic and Inorganic Compounds, the Scientific Basis of a Natural Classification of Organic Chemical Bodies." To Vieweg he often referred to it as his "confession of faith" (Glaubensbekenntnis). Richard Anschütz referred to it as a kind of counterpart or rejoinder (Gegenstück) to Kekulé's structure theory paper of 1858,[60] and it seems likely that Kolbe thought of it this way as well. Dated 27 September 1859, it appeared in the March 1860 issue of the Annalen . The four types of Gerhardt's theory had been used to try to unify organic and inorganic compounds,
Kolbe related, but the relationships they revealed were unnatural, artificial, and superficial, leading to an "empty game with formulas" and a "dead schematicism"; "nature does not limit herself to variations on four themes." A few formulas from Kekulé's structure theory paper of 1857 were cited as exemplifying this game-playing approach. However, Kolbe's theory—as the title proclaimed—offered a "natural" and "scientific" basis for such a unification. The fundamental axiom was "organic chemical bodies are invariably derivatives of inorganic compounds, and have been formed from the latter, some in a direct fashion, by wonderfully simple substitution processes ," namely, substitution in carbonic acid.[61] Deriving organic compounds from this single progenitor is not only simpler and more general than Gerhardt's approach, it also focuses on the most important substance for plant physiology and biosynthesis. In this way, the chemist's scheme nicely parallels nature's.
The ideas in this article are the same as in fascicle 8/9 of his textbook, but they are treated here with substantially greater cogency and consistency. Kolbe wisely pruned most of the hypotheses and predicted isomers that had appeared in his text, while highlighting the ones that he obviously thought the most of, especially the prediction of isopropyl and tertiary butyl alcohols. The organization is clear and systematic, and the exposition magisterial.
He also discussed two additional important and theoretically troublesome organic diacids. Kolbe suggested that malic and tartaric acids are succinic acids with one and two hydrogen atoms, respectively, of the hydrocarbon radical replaced by hydrogen peroxide, HO2 . In a sense, this substitution is simply an insertion of oxygen O2 into the radical, hence the names Oxybernsteinsäure and Dioxybernsteinsäure for malic and tartaric acids. (Following Kolbe, German organic chemical nomenclature to this day preserves the prefix Oxy- to indicate hydroxy substitution.) They remain dibasic acids because this oxygen is adding inside the radical, and according to Kolbe's basicity law, this has no effect on basicity. If any of these diacids were to decarboxylate (lose carbonic acid C2 O4 ), however, the molecule would lose one of the two dibasic radicals C2 O2 upon which the double type was formed. A third oxygen would then be lost from outside the radical, reducing the basicity to one, and the fourth oxygen to make up C2 O4 would come from one of the two attached water molecules. The hydrogen thus set free would enter the hydrocarbon radical.[62]
The paper just described constitutes Kolbe's virtually final formulation of the theory of organic compounds. After more than a decade of wandering in the theoretical wastes, Kolbe felt he had arrived in the promised land of chemical truth. To the end of his life he regarded the
ideas in this paper as both necessary and sufficient to eventually expose to the light of day all the exotic creatures in the tropical jungle of organic chemistry.
The Response
On the day the article was officially published, Kolbe wrote Vieweg, concerned about the general course of battle in his war with the reformers. The majority of chemists were now following Gerhardt—even his good friend, the otherwise sober and reasonable Strecker—and Kolbe was isolated in his opposition.
In my view this whole movement has already outlived its usefulness. Such mindless ideas as are to be found in Weltzien are its dying echoes. The adherents of Gerhardt's school in Germany as in France have shot their bolt. They are riding to death the poor old nag that carried Gerhardt forward for a time, without producing anything other than variations on the old theme. Not even the entire great Göttingen school has educated anything approaching a capable and intelligent chemist. Limpricht has created a spectacle, but produced not a single piece of work that shows anything like ingenuity or sharpness of intellect. Fully as unthinking a man, but correspondingly foolish a chatterer, is Kekulé. In a word, the whole mindless business, on which I expressed my judgment in the beginning and concluding sentence of the article just now appearing, is bankrupt and has been outlived. What people of this kind are doing is not science, but a game.— But I see I am letting myself go on too long. The future will tell.[63]
Considering these views, it must have been extraordinarily discouraging to Kolbe to see that the initial response of many of his colleagues was to place Kolbe himself in the midst of the "whole mindless business" of newer type theory and polyatomic radicals. Wurtz was the only member of the reformist camp to reply in print. In a review of the article in his Répertoire de chimie pure , he wrote: "Monsieur Kolbe has so fully adopted the fundamental idea of types that not only does he want to multiply them, but even, with Gerhardt, to assume condensed types, as are represented by [his] molecules of carbonic acid." The carbonic acid type, Wurtz affirmed, is nothing more than the water type with diatomic carbonic oxide functioning as the oxygen atom, as Williamson had formulated it in 1851.
Wurtz thought Kolbe's argument that Gerhardt's four types are artificial was not a trivial one; nature should indeed not be limited in this way. But what Kolbe did not see, Wurtz argued, was that a more general principle lurks behind these types, namely, successive degrees
of condensation of matter. Hydrogen, water, ammonia, and marsh gas represent hydrogen once, twice, thrice, and four times condensed, with the oxygen, nitrogen, and carbon atoms representing two, three, and four atoms of hydrogen, respectively, in one unit of action. This is what he had meant to express in his subatomic speculation of 1855. Wurtz thought that the idea of types ought to be replaced by the idea of the atomicity of the elements, which is the foundation of the phenomena that types attempted to express. "Here is a clear, simple, and general principle, which deserves for this reason to be placed at the base of a system of chemistry." Kolbe's approach was fundamentally the same, Wurtz urged, whether he was aware of this or not. "If, then, the ideas this chemist presents are novel, the innovation resides rather in the form than in the essence, and I believe that I have shown that even the form is not fortunate; in truth, he combats Gerhardt's types by counterfeiting them."[64]
Kolbe's friend Hermann Kopp saw the matter similarly, although he phrased his criticism more kindly. Reviewing the 1860 paper in the Jahresbericht , Kopp wrote: "Kolbe still argues against relating organic compounds in general to the hydrogen, water, and ammonia types, as also against the recent assumption of mixed types. But he reveals himself as a de facto adherent of the 'type theory' by conceiving organic compounds as derivatives of inorganic compounds." Kopp also pointed out that Kolbe had used multiple types to formulate diacids, and suggested the influence of Gerhardt. Three years earlier Kopp had also referred to the Gerhardtian and Williamsonian elements in the Kolbe-Frankland carbonic acid paper.[65]
However, Kolbe's paper impressed one very important reader: Justus Liebig. Liebig wrote Kolbe on 3 April 1860 to express his "great satisfaction" with the paper; the schematic derivation of malic and tartaric acids from succinic acid was "the triumph of your theory."[66] Liebig himself never penetrated the sense of newer type and structural formulas. To Wöhler he admitted his disinterest in modern chemical theory and complained about the thoughtless manipulation of formulas that was going on around him. Wöhler's opinion was similar.[67] In the 1860s Kolbe's and Frankland's articles were some of the few that excited Liebig. He told each of them separately that only in their work could he see the guiding influence of "a large scientific idea" directing the experimental program. To Frankland he wrote: "What Wöhler and I saw thirty years ago in dreams, that is, in our imagination, you are now on the road to realizing. . ." To Kolbe he sympathized regarding the "Formelspielerei" of many who were not on the correct path.[68] Ironically, by the time Liebig was writing this letter Frankland had become a full-fledged member of the structuralist school, whose
manipulation of formulas was entirely in their spirit, while Kolbe was making a sharp distinction between his and nearly everyone else's formulas.
Liebig's support fortified Kolbe's spirits at a time when it appeared all the world was converting to the reform. Led by Kekulé, a small cabal including Weltzien, Baeyer, Roscoe, and Wurtz planned an international conference whose ulterior motive was to attempt a world standardization of the Gerhardt-Laurent atomic weights and molecular formulas.[69] The need for such a meeting was by no means uniformly conceded. Some, such as Leopold von Pebal who had been a confirmed Gerhardtian for years, thought such a degree of unity already prevailed that it was not necessary. He expressed this sentiment in a letter to Roscoe, adding "Kolbe will hardly be converted!"[70] Others, including some reformers themselves such as Williamson and Brodie, thought it was unseemly to attempt, or even to give the impression of attempting, to legislate scientific conviction. Several, including Erdmann and Kopp, espoused such sentiments at the meeting itself.[71]
Lothar Meyer, a leader of the reform movement after 1860 and a good friend of Pebal and Roscoe from their Heidelberg period, occupied an ambivalent and interesting position before the meeting. He was still studying with Bunsen in Heidelberg when Kekulé arrived there early in 1856, and Kekulé's eager advocacy for Gerhardt's type theory made a strong impression on Meyer, according to his later reminiscences. "How very well I remember even today," he recalled,
. . . the debates lasting hours and days, in which he won ground step by step. The authority of the accepted dualistic theory and the decided aversion of our honored master to get involved with the new business with formulas [Formelkram] explains why we gradually came over to the other side only after energetic resistance.[72]
But in Meyer's case, it would appear, this did not happen before the Karlsruhe Congress. In July 1860 he wrote Roscoe saying he felt obliged to come to the "idiotic church counsel, to propose the election of an infallible formula-pope," but feared what the edicts of the "Committee of Public Safety" at such a "National Convention" might prove to be.
The good Gerhardt, if he only knew what kind of scandal is being made of his innocent types! . . . These "types" are a dangerous toy for fools; but the expression "type theory " is an insult to science, which recognizes a theory of gravitation, of light, of capillarity, etc., but which can never agree to having a notation called a "theory."[73]
As a matter of form, Weltzien wrote Kolbe to ask for his support. Kolbe replied that since the science was so dominated at the moment by the Gerhardt-Williamson-Kekulé-Wurtz school such a conference would do more harm than good. Any resolutions taken at the meeting would have to be overturned upon the fall of the reform movement, an event Kolbe expected to see "in the very near future."[74] Kolbe had every intention of skipping the meeting, but at the last minute he let himself be persuaded by Fehling to go for one day of the three-day conference. It was, he wrote Vieweg, a real "farce," just as he predicted it would be.
Weltzien let himself be used by Kekulé, to give the latter the opportunity to pass himself off to the assembled chemists as the great chemist of the future. Instead of some kind of result or agreement (the most important questions were never even raised), only a general dissatisfaction and disappointment was achieved. Kekulé gained precisely the opposite of what he wanted; he made a real fool of himself and turned everyone against him.[75]
This was, of course, a jaundiced point of view, but the organizers themselves were likewise somewhat disappointed by the absence of concrete results. However, the conference had long-term effects that were not visible in the fall of 1860. Lothar Meyer, who had been so cynical in July, was profoundly affected, not so much by the events themselves in Karlsruhe, but by having been handed an offprint of Cannizzaro's Sunto di un corso di filosofia chimica . He read it on his journey from Karlsruhe and "repeatedly" after his arrival home. He "was astonished by the clearness that the brochure provided concerning the most important points at issue. It was like blinders being removed from my eyes; doubts disappeared, and a quiet feeling of certainty replaced them."[76] The Russian chemist Mendeleev related a similar story regarding Cannizzaro's influence.[77]
The general historical impression that Cannizzaro was the decisive personality in Karlsruhe and that his brochure helped achieve completion of the reform is probably accurate. The work also appears to have had a strong effect on Frankland. In assessing the relative contributions of various chemists to the formulation of structure theory, he gave Cannizzaro a higher place than Kekulé, for it was Cannizzaro, he said, who established the atomic weights that made structure theory viable—a point made by Meyer as well.[78] In any event, Frankland's publications show that in the months before Karlsruhe he had still not accepted some important elements of the reform, whereas by 1862 he was fully in the modernist camp. Wurtz and Hofmann, who had been reformers for years, only began to use the two-volume atomic weights
in their published papers around the time of the conference, and this may also have had some impact on Frankland's thinking.
But it would be erroneous to conclude that Kolbe was the only holdout after Karlsruhe. The elder spokesmen, especially Liebig, Wöhler, Bunsen, and Dumas, continued to prefer the apparently more empirical conventional equivalents. This was partially because they held themselves aloof from the theoretical trends of the day, as they themselves, as well as their ardent defenders such as Kolbe, often admitted. But exceptions existed even among the younger active chemists. Among the French, Marcellin Berthelot was just as contemptuous of the reform movement as Kolbe was. The empirically minded Friedrich Beilstein, another friend of Kekulé's from his Heidelberg period, accepted the new atomic weights, but was disgusted by the facile manipulation of structural formulas.[79] Even Kekulé complained to Meyer, shortly after the Karlsruhe Conference, of "Constitutions-formel-spieler."[80] Indeed, there were many who were happy multiplying possible formulas far beyond necessity—Kolbe himself was susceptible to this charge. In fact, it was surely to Kolbe that Kekulé was referring in the phrase just cited.
Was Kolbe a Type Theorist?
To comprehend the character of Kolbe's route to his mature theory, it is useful to recapitulate the four stages of his passage in the period from 1856 to 1860. In his article on radicals completed by the beginning of 1856, Kolbe appears to have accepted Frankland's valence regularity, from an application of which he suggested that methyl could substitute once or (perhaps) even twice for oxygen atoms of carbonic acid, to form acetic acid and (perhaps) acetone. By the end of that year, following correspondence with Frankland on the subject, he further generalized the scheme in a theoretical paper for the Annalen , now suggesting, with Frankland, that hydrogen as well as alkyl radicals could replace oxygen in carbonic acid; the experimental demonstration was, however, still lacking. Evidence from correspondence suggests that only then was Kolbe beginning to realize the far-reaching significance of this new idea.
Kolbe entered into the third stage during the summer or fall of 1857. By this time he fully accepted both the vocabulary and the general validity of type theory; he abandoned copulas, conjugate radicals, and buckles, and asserted the "diatomic" nature of both carbonic oxide and carbonic acid. His prediction of the existence and properties of isopropyl and t -butyl alcohol emerged from these considerations. The fourth and final stage was achieved by the summer of 1858, when he
first proclaimed the "tetratomic" nature of the carbonyl radical (C2 ) and for the first time accepted polybasicity as a general phenomenon in acids. Once he had crossed this threshold he was able to devise a general theory that was substantially similar to that of the type theorists. But there were substantial differences as well.
One element in Kolbe's failure to find full accommodation with the reformers was the extremely literal sense in which he interpreted chemical formulas, his as well as those of other chemists, and his inability to see what was intended by others' notation. One obvious instance was his continued retention of conventional equivalents, which created difficulties in communication. During the 1860s, most German chemists began to use the newer weights. As early as 1862 Kolbe was in a distinct minority, and he must have felt the peer pressure in many ways. It was not only his ingrained conservatism that held him back from conversion, but also his discovery of the "basicity law," which states that the basicity of a polybasic acid is equal to the number of oxygen equivalents thought to be located outside the radical (and always matched by an equal number of "molecules" of water, HO, on the other side of the formula).[81]
Kolbe was not the only one to write aggressive critiques in the pages of his textbook. In fascicles of his own textbook published in 1861 and 1864, Kekulé criticized Kolbe's formulas. One problem, Kekulé argued, was Kolbe's inconsistent notation for alcohols. In the formulas for hydroxy acids, Kolbe wrote alcoholic groups as HO2 , whereas alcohols themselves were notated in the same manner as acids, namely, O.OH. More fundamentally, in Kolbe's basicity law an increment of two oxygen equivalents is always necessary when increasing basicity. This itself should have led Kolbe to double his equivalent for oxygen; in Kekulé's view, the law was "explained by the circumstance that two half atoms of oxygen are required to form a whole atom." If the oxygen equivalent were doubled to halve the number of atoms in the formula, then marsh gas rather than carbonic acid must instead be used as the carbonaceous type, "whereby this viewpoint would coincide with that developed in this textbook."[82]
Kolbe, Frankland, and Butlerov all pointed out later that each "O" in formulas written in conventional equivalents could be considered to represent not an atom but an affinity unit, thus remaining consistent with valence theory.[83] Such arguments were later used by Kolbe and Frankland to maintain that they had been the earliest to formulate carbon tetravalence. In an unpublished paper written in 1883, Kekulé rejected this claim. In Kolbe's theory, alkyl radicals or hydrogen schematically and successively substitute for the oxygen of carbonic acid, 2HO.C2 O4 . Kekulé wrote
Probably no one has really understood these formulas. It is not at all clear why the attached water equivalents (i.e., half water molecules) vanish as the oxygen atoms (i.e., half oxygen atoms) are replaced. The whole derivation is only possible if, proceeding from the hypothetical hydrated carbonic acid, hydroxyls or water residues are replaced by hydrogen or radicals. In any case the entire procedure has nothing at all to do with the tetravalence of carbon.
Kekulé also pointed out that even if Kolbe's argument were accepted, his own structure theory articles were published earlier than the installments of Kolbe's book in which the tetravalence of the carbonyl radical C2 was first advanced.[84]
In the most general case, Kolbe interpreted the goal of any chemical formula, once properly formulated, to be the ultimate expression of the chemical properties of the substance. If a new reaction could not be expressed by the formula, Kolbe usually regarded it as a serious anomaly, often rejecting the formula and the idea behind it, even if it was his own. Conversely, he rarely hesitated to predict unknown reactions or to posit unknown isomers as logical consequences of his formulas. This was one reason why the details of his theoretical system kept shifting throughout the 1850s. The emerging structuralist camp, following a perception of Gerhardt, began to see that such conflicts were not so much anomalies as expressions of the incompleteness of most formulas; only a fully resolved structural formula, and one that was adequately understood through a multitude of reactions—such as the formulas for acetic acid or triethylamine—could be judged as ultimate. By "fully resolved" the structuralists meant showing each atom in the molecule and how it was connected by valence bonds to every other atom. This was an essential point, first made in an explicit fashion by Kekulé in 1858.
For example, Kolbe thought that the chemical analogies of ethylene with carbonic oxide (which both undergo addition reactions with chlorine or oxygen) meant that the atoms adding to ethylene had to be assumed to be adding to a single carbon site, as they necessarily did with carbonic oxide. Consequently, both ethylene and its chemical derivative glycol had to have methyl groups. Like the reformist type theorists, Kolbe was convinced (inter alia by Kekulé's monochloroacetic acid hydrolysis) that glycolic acid was a substitution product of acetic acid and so did not have a methyl group in its constitution. Thus, he concluded that glycol and glycolic acid could not have analogous constitutions. This assumption compelled him to propose several yet undiscovered substances: an oxidation product of glycol only isomeric with glycolic acid; a reduction product of glycolic acid only isomeric
with glycol; an intermediate oxidation product of this last compound only isomeric with acetic acid; and an unhydrated ethylene oxide only isomeric with ordinary aldehyde. Kolbe must have considered the hypothesis of all these unknown compounds to be a less serious price to pay than relinquishing the analogy of ethylene and carbonic oxide.
Kolbe shared with Frankland three variances from the Williamson-Kekulé-Wurtz school. One was the belief that valence could vary (e.g., that ethylene and carbonic oxide both possess a di valent carbon). A second was the ontological commitment to conventional equivalents, by Frankland until 1862 and by Kolbe until 1868. A fully resolved formula in which the focus of resolution is equivalents did not and probably could not yield structure theory in its most fruitful form. Even with atomic weights there proved to be enough anomalies to structure theory tenets (such as varying valences in oxygen and sulfur, multiple bonds in olefins, aromaticity, and so on) to require a laborious sorting-out process through the 1860s and 1870s. With conventional equivalents, it was barely possible to perceive valence itself (as exemplified by Kolbe's intellectual path, though Frankland managed it in 1852), much less the most fruitful kind of structure theory. To mention just one example, it was the commitment to equivalents that allowed Kolbe to separate his water "molecules" from the rest of the formulas.
Finally, one can perceive a persistence of instinctive electrochemical dualism, in Frankland until 1860 and in Kolbe to the end of his life. It was this vestigial dualism that prevented Kolbe from accepting the crucial chain-forming ability of carbon. It is difficult enough to understand polyvalence, not to mention chain-forming, under dualistic assumptions. Even after formulating his carbonic acid theory in its final form (by 1860), Kolbe was quite clear that he still regarded coulombic forces as the explanation for chemical affinity. Carbon atoms did not "link" together, and they had no "bonds" at their disposal. Rather, organic molecules were composed of proximate pieces (radicals) that substituted electrochemically for one another, or for certain atoms (such as oxygen in carbonic acid).
To use modern language to describe Kolbe's conception, chemical affinity is always isotropic, like all other (physical) forces. The methyl radical did not have a "bond" or a "hook" or any other form of localized or spatially oriented valence force. It substituted for another atom or radical as a whole , acting isotropically and coulombically. He could not explain these coulombic interactions in detail, and he knew it. No matter. He was absolutely convinced that chain formation was impossible because it seemed to exclude the only correct basis of chemical affinity—electricity. Thus, it happened that he was to develop a theory
that was largely equivalent to structure theory but which denied that critical idea. That was his carbonic acid theory.
One more philosophical underpinning of Kolbe's theory requires mention. After 1859 Kolbe conceded his critics' charge that he, too, was a type theorist. Indeed, he often admitted the early fruitfulness of Gerhardt's theory. The certain indication that adherents of the theory had overreached themselves, Kolbe thought, came when they began to propose multiple and mixed types that had no material connection with the original type. How could carbonic acid, for instance, be formulated on the water type when it is the carbonic oxide moiety, not oxygen as in water, that is the dibasic entity? By deriving most organic compounds directly and materially from carbonic acid, Kolbe was proposing "natural" and "real" types, in contrast to the merely "formal," indeed "fictitious" types of the "modern" type theorists. The merely formal is not real, he felt, therefore the structural chemists were just playing superficial games on paper.[85]
The curious aspect of this line of argument is that it has very much in common with Dumas' original concept of types as both mechanical and material progenitors of their derivatives. As Dumas had done earlier, Kolbe was asserting the ontological significance of the type for its progeny. Or as Wurtz put it before he converted to the Gerhardtian view, the type imprints a "cachet" on all of its derivatives. A consequence of this view was that formulas must be taken very seriously and very literally. The type formula specifies one atom, outside the bracket, that is chemically as well as visually central. A derivative belongs to one type or to another, but not to more than one.
In departing from this point of view, Gerhardt had begun to edge toward a position according to which, at least in principle, all the atoms in a molecule could be accorded the same importance. Different reactions reveal different aspects about the molecule, each of which can be expressed by a different formula and all of which are valid. This establishes a sort of chemical "democracy" among the atoms in a molecule, in contrast to the hierarchical structure of the early type theory—and of Kolbe's types. Multiple and mixed types were anathema to Kolbe precisely because they represented a movement toward this democratic, nonhierarchical view.
It was most natural for Gerhardt to combine this view with a radically empiricist position, a position that Kolbe as an instinctive theorist detested. Here again Kolbe found himself in the same league with Dumas (although Dumas' theoretical and realist orientation was often hedged and even disguised by empiricist protestations). For Williamson, Kekulé, Wurtz, and other theorists in the reformist school, a combination of Gerhardt's relativist view of the atoms in a molecule and
Dumas' realist approach toward "mechanical types" together provided the cognitive route to structure theory. It was also necessary that the emergent structuralists cease to worry excessively about what precisely constituted valence and simply abandon familiar macroscopic physical analogies.
These considerations explain Kolbe's apparently inconsistent position. Gerhardt's radical empiricism clearly was not science to him. Nor was the overly schematic, formalist, conventionalist direction of the newer type theory (otherwise known as structure theory). Schematism is not realism, Kolbe insisted, and conventionalism is a farce; at best, such workers were playing superficial games, at worst they were destroying the science by accepting false speculations as truth. Moreover, abandonment of physical analogy to accept chain formation was in a sense a pragmatic and empirical response to an anomaly. Kolbe could not accept such a move because he was a passionate theoretician and a man of firm principles (although his opponents would have expressed this characteristic as obstinance).
The situation can be expressed somewhat facetiously (and in a way that Kolbe himself would find objectionable, due to his burning hatred for Dumas) as follows. Kolbe, a conservative to the bottom of his soul, retained elements of Berzelian chemistry as long as possible, and dualist vestiges survived in his system until his death. But when, during the period 1853-1860, he was forced to concede many critical points and become a type theorist himself, he remained true to his conservatism by, in effect, choosing the oldest form of the theory (Dumas') and then refusing to modify it for the last quarter century of his life.