Boltzmann's Combinatorics
The basic object of Boltzmann's combinatorics of 1877 is a perfect gas of point molecules, a microstate of which is characterized by the set of molecule velocities and positions. In a first simplifying step, Boltzmann considers only the kinetic energies of the molecules and tries to define the probability of an energy partition (Energieverteilung ), that is, of a distribution of a given total energy E over the molecules. Since energy is a continuous variable, there is no obvious definition of such a probability.
As in his combinatorial considerations of 1868, Boltzmann starts with a "fiction" wherein molecules can take only discrete energy values 0, e , 2e , . . ., i e , . . . . Then, if molecules are labeled by the index (a = 1, 2, . . ., N ), a microstate of the system, or "complexion," is defined by attributing to each molecule a given energy:

where iae is an integral multiple of e . An "energy partition" is given by a sequence of integers N0 , N 1 , . . ., Ni , . . ., where Ni is the number of molecules carrying the energy i e . To a given partition correspond 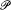 different Komplexionen . Boltzmann calls
different Komplexionen . Boltzmann calls 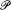 the "permutability," since it is equal to the number of permutations of the N molecules that transfer at least one molecule from one discrete energy value to another:
the "permutability," since it is equal to the number of permutations of the N molecules that transfer at least one molecule from one discrete energy value to another:
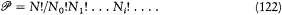
The probability W of a partition (N0 , N 1 , . . ., Ni , . . .) is obtained through division of 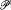 by the normalization factor; Boltzmann gave an explicit formula for this divisor:
by the normalization factor; Boltzmann gave an explicit formula for this divisor:
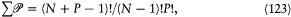
[81] Planck to Graetz, 23 Mar. 1897 (Deutsches Museum), quoted in Kuhn 1978, 265-266.
[82] Boltzmann 1877b.
where the sum is taken over all distributions (N0 , N 1 , . . . Ni , . . .) such that
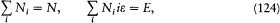
and P is obtained by dividing the total energy E by e . It might be worth mentioning that this combinatorial formula was the one later used by Planck.
If the Ni 's are large enough to allow the Stirling approximation,
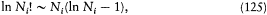
W reaches its maximum value with the constraints (124) on the total number of particles and energy if for any i , and for Lagrange multipliers a and b .
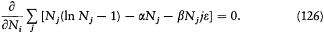
This equation implies that N i must be proportional to e-ibe . Maxwell's distribution, or a discrete imitation of it, appears to be the "most probable" one, in the sense that it has the greatest number of complexions.
So much for the fiction. Boltzmann then turned to the more realistic continuous case. This was readily achieved by supposing that the energy unit e was small enough to consider that molecules whose energies lie between ie and (i + 1)e have the same energy. The numbers Ni now count the molecules in the various energy intervals. Provided that the sums over i can be approximated by integrals, the most probable number of molecules whose energy K lies within an infinitesimal energy interval dK is proportional to e-bKdK .
This is not yet Maxwell's law. To get it Boltzmann had to cut up the velocity space, instead of the energy axis, into uniform cells. Then Maxwell's expression
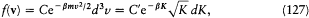
was found to represent the most probable distribution of molecular velocities. Boltzmann also considered the positions r together with the velocities of the molecules. In this case the (r, v)-space has to be cut up into uniform cells, and Ni gives the number of molecules in the cell i . In the continuous limit the most probable distribution f (r, v) is uniform in the r-space and results in Maxwell's distribution in the velocity space. Furthermore, the logarithm of permutability may be calculated in this case to give
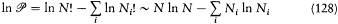
or, in the continuous approximation,
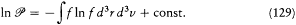
In the case of maximal probability this expression of In 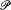 , Boltzmann noted, is identical with the function -H and therefore gives the entropy of a perfect gas up to an additive constant (function of N ).
, Boltzmann noted, is identical with the function -H and therefore gives the entropy of a perfect gas up to an additive constant (function of N ).
These calculations were simple enough, but their point of departure, the expression of the probability of a state distribution, needed further justification, as Boltzmann himself recognized: "I do not think that one is allowed to set this forth [that the equilibrium state is the most probable one] as something obvious, at least not without having first defined very precisely what is meant by the most probable state-distribution." He tried to justify the two main assumptions leading up to his expression for per-mutability, namely, the possibility of cutting up, and the uniformity of, (r, v)-space.[83]
This uniformity, he said, resulted from the invariance of the differential element d3r d3v during a Hamiltonian evolution of a molecule. But the accompanying proof was either incomplete or wrong. In any case it could not fill the conceptual gap later emphasized by Einstein: a proper connection between the evolution in time of a system and the probability of its state was needed to justify not only uniformity in (r, v)-space but also, more generally, the relevance of combinatorial probabilities to thermodynamics.[84]
Boltzmann's combinatorics, if not fully justifiable, had, at least, to be consistent. In this respect the recourse to finite cells could seem problematic. The number of molecules in a given cell had to be large (more precisely, there had to be a negligible number of cells for which Ni is neither zero nor very large) so that the Stirling approximation could be applied. At the same time the size of the cells had to be small enough that the sums over i could be approximated by integrals. Instead of directly investigating the consistency of these assumptions, Boltzmann preferred an analogy with familiar problems of kinetic theory:
Nevertheless, after closer inspection, this assumption must be regarded as obvious. Indeed, any application of differential calculus to gas theory rests on the same assumption. If for instance one wishes to calculate diffusion, viscosity, conductivity, etc., one has to admit in the same way that in every infinitesimal element of volume dx dy dz there is still an infinite number of gas molecules
[83] Ibid., 193.
[84] See Kuhn 1978, 55.
with velocity components lying between the limits u and u + du, v and v + dv, w and w + dw . This assumption means only that one can choose the limits for u, v, w , so that they include a very large number of molecules and that one may nonetheless regard all these molecules as having the same velocity components.[85]
In the content of the 1877 memoir, one can easily check the legitimacy of this hypothesis for the most probable distribution of molecules. For instance, in the simplest case in which the energy axis is cut up into intervals of equal size e , the numbers Ni are given by
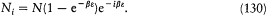
It can easily be seen that the condition for the Stirling approximation to be valid is a large value of the number N0 of molecules in the zero-energy interval:
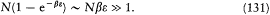
The other condition, that the sums can be replaced with integrals, reads:
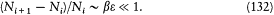
The two conditions are both met if

Consequently, for any value of the available energy (E = N/b ) the size of the cells can be chosen consistently, and it then disappears from the final result for the most probable distribution and the corresponding entropy.
Since in Planck's later combinatorics e is not always a negligible fraction of 1/b ( = kT ) and appears in the final entropy formula, it is important to understand what makes it disappear in Boltzmann's case. The reason is not that e is infinitesimal in the mathematical sense; indeed, it must be larger than 1/Nb . At a purely formal level, the elimination occurs when sums like the one giving the entropy are replaced with integrals. Boltzmann had to take this formal step because the main physical quantity of interest, the distribution of molecules over cells (N0 , N 1 , . . . Ni , . . . ), was expected to be well approximated by a continuous distribution, the most probable of which is Maxwell's law. We will find that neither this circumstance nor its formal corollary occurs in Planck's combinatorics.
To summarize, Boltzmann's memoir of 1877 on entropy and probability was not very explicit about the physical meaning of its main procedural elements, the method of dividing up the space of configurations and the
[85] Boltzmann 1877b, 197-198.
uniformity of this space. It was clear to him, however, that combinatorial methods were relevant insofar as they were able to reproduce the "continuous" entropy formula

which had already been founded on what he considered to be more fundamental bases, that is, on the ergodic hypothesis or on the methods of the H -theorem. On the contrary, Planck, still unfamiliar with the foundations of Boltzmann's theory, would venture to confer physical meaning on the artifact of energy elements.