8—
Water Analysis and the Working Sanitarian
I am not conscious of ever having made a mistake in water analysis.[ 1]
Cornelius B Fox
In May 1884 Alfred Ashby, medical officer of health for Grantham, complained to the Society of Medical Officers of Health of 'the wretched wrangling, during the ten years past, over these rival processes, and . . . the ridiculously exaggerated importance that has attached to them.' This wrangling was the cause of 'the discredit [in]to which water analysis has fallen among engineers and sanitarians.'[2]
It is easy to share Ashby's view that the London chemists had become so preoccupied with deprecating one another's laboratory prowess that they had lost sight of the key question of what water analysis was to be for: the pursuit of precision had come to overshadow questions of sanitary significance. The community of sanitary chemists had become so caught up with whether Frankland's or Wanklyn's process was the better, that the possibility that the best answer might be neither, might instead be something as radical as correlating analytical and epidemiological results, was rarely considered. Frankland and Wanklyn checked the accuracy of their processes on solutions of pure organic compounds of known strength, yet each claimed that the key determination he chose to make also had great significance for distinguishing safe from harmful water. In retrospect their attempts to measure a few parts per million of what was in Wanklyn's case an experimental artefact seem ludicrous. It is not so much that they were using chemistry to detect what turned out to be biological, but that they kept egging on one another for so long without stopping to ask whether they were taking water
analysis in a useful direction. How, we may wonder, could this be tolerated by the larger community, and especially by those actively seeking to safeguard the public from water-borne disease?
When we take into account the reasons analyses were commissioned and the uses they were put to, the rationale is clearer. Chemists of the stature of Frankland, Wanklyn, Letheby, and Tidy were regularly called upon to act as expert witnesses, both in legal actions involving pollution and before Parliament in the assessment of alternative sources for town supply. In neither context was there likely to be a clear-cut right answer; in such cases issues involving water quality were entangled in larger matters of law, policy, finance, and politics. We have already seen that these had become prominent contexts for water analysis beginning in the late '20s, yet we have not yet fully addressed the implications of working in such a context. Expert witnessing called for a set of attributes—in demeanor, authority, willingness to speculate—quite unlike those needed either in pure science or in front-line public health work. In the former uncertainty was admissible, while the latter placed a premium on thoroughgoing analysis of individual cases. The expert witness on the other hand had the job of persuading lay decision-makers that his claims and the perspectives of those he represented were sounder than his opponent's. As matters of water composition became more important and as methods of analysis became more sophisticated and their rationales more complex, the importance of the external symbols of credibility—credentials, assurance, coolness on the stand, the finality with which one could demolish an opponent's argument—all these became more and more important. And one's credibility hung to a great degree on the apparent credibility of one's analytical process. The chemist who worked at an order of magnitude smaller than his rivals and whose process had been more rigorously tested, was more likely to be believed, regardless of whether his measurements contributed anything of import.
This adversary context favoured the manufacture of certainty, however flimsy that certainty might later be shown to be. The expert who could deliver certainty on some matter, no matter how small and useless, was in a stronger position than one who could make informed, prudent, and plausible assessments of the central matters at hand, yet provide no certainty. It was far easier for the former to claim greater territory for his certainty than for the latter to recover from an honest admission of doubt. To the vultures of the parliamentary bar, doubt was vulnerability.[3] One may wonder
how much use all this would be in the pursuit of public health; with a community of analysts bent on showing up one another's incompetence using analytical processes incapable of indicating anything certain about the presence of dangerous microbes, it might seem that decision-makers were in effect operating in ignorance. It is true that the links between an analyst's claims about the safety of a water and any investigation which could reasonably be regarded as warranting such a claim are sometimes tenuous indeed; moreover public health was often not the main agendum either in pollution litigation or in efforts to obtain purer water supplies. Better water was often sought by industrialists for industrial use and litigation undertaken to protect property values. But regardless of the motives of the promoters of better water and regardless of the means their hired scientists used to fight their credibility battles, better water and better health were often the outcome.[4]
To those on the front lines of public health work this sort of performance was much less useful. The health officers responsible for investigating outbreaks of epidemic diseases, both those in central government like John Simon and his staff of medical inspectors at the Privy Council (and later at the Local Government Board), and those serving local governments as medical officers of health, were rarely much concerned with the great questions of water supply policy. What they needed were quick and easy techniques for distinguishing fluctuations in the quality of service on a single street, or perhaps even for a single house. Frequently epidemics of water-borne diseases were the result not of an ongoing contamination, but of some unusual combination of circumstances.[5] The Caterham typhoid of 1879, for example, was started by a workman engaged in digging a new well who had the disease.[6] Sanitary engineers likewise were out of sympathy with the practices of the London chemists: 'the extreme refinements of modern analysis has seemingly led chemists to set up a standard of purity in excess of the necessities of the case,' one complained.[7]
After 1875 the dominance of the London consultants began to wane, as this kind of immediate and on-site approach became increasingly important. The great Public Health Act of 1875 mandated the appointment of a medical officer of health by each sanitary authority. In many cases these positions were for part-time officers only, but it was becoming increasingly common for their holders to have had specialized training in public health. One could earn a diploma in public health medicine at the University of Dublin after
1871, or at Oxford or Cambridge after 1875. In addition, a sort of British Association for sanitary matters, the Sanitary Institute of Great Britain, was founded in 1877. It took it upon itself to certify the competence of local sanitary officials. After 1875 towns were also required to employ a public analyst, an office established to fight food and drug adulteration. Both the medical officers of health and the public analysts founded professional organizations, the Association (later Society) of Medical Officers emerging out of the older Association of Metropolitan Medical Officers in 1875, the Society of Public Analysts being founded in 1877. In 1891 a third organization, the British Institute for Public Health, was founded, its membership restricted to holders of the diploma in public health. Water analysis was officially within the mandate of neither medical officers of health nor public analysts, yet in many cases holders of these offices took an interest in water quality as did their professional organizations.[8]
Relations between these local sanitary professionals and the London elite were not good. Front line sanitarians knew at first hand the inadequacies of the ammonia and combustion processes for distinguishing disease-causing waters. They were also well placed to see how contradictory were the opinions of different chemists on the same source of water. In 1875 the chemist J Carter Bell, later public analyst for Cheshire, wrote to the Chemical News to report such an outrage. A water sample from a newly dug well had been sent to an unnamed chemist for analysis. Finding no putrescent organic matter, this analyst had pronounced it satisfactory. A year later a sample had been sent to a commercial analytical firm, which reported that it contained flocculent organic matter and stated that it was dangerously contaminated. A third analyst, a medical man using the ammonia process, concurred. A fourth, a chemist, judged the water safe on the basis of chlorine and oxidized nitrogen levels. A fifth, relying mainly on the ammonia process, broke the tie by declaring the water safe. Bell found the situation scandalous. Each chemist had used a different set of processes, he noted. He suggested that a jury of the Chemical Society ought to select and enforce a set of standard methods.[9]
Alfred Ashby had a similar story. Having found analytical evidence of 'sewage pollution' in a well water from Newark-on-Trent, he requested the magistrates to order it closed. The well's proprietors countered with an analysis by Charles Graham of University College which showed the water to be good, and faced with conflicting scien-

Figure 8.1
Writers of late nineteenth century public health manuals sometimes
tried to give their readers sample analytical profiles of various qualities
of waters, all the while advising that these were not to be taken seriously
since so many factors had to be taken into account when judging waters
(Parkes' Manual of Hygiene , 6th edn, p 104).
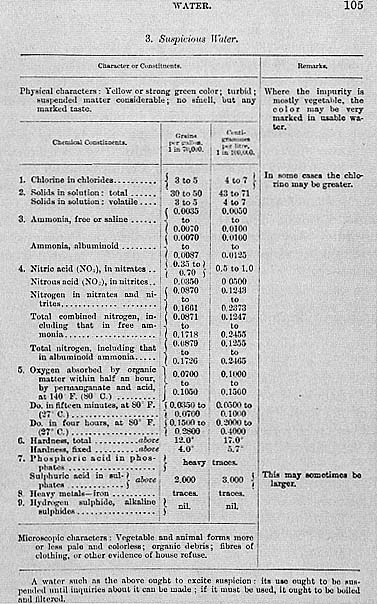
Figure 8.1
(continued)
tific testimony, the magistrates threw out the case. Ashby then had samples analysed by August Dupre and Otto Hehner, prominent members of the Society of Public Analysts. Both condemned the water. Still the magistrates were unconvinced, and sent a sample to
an 'independent' analyst, Frankland. He too pronounced the water unfit—'soakage from drains or cesspools'—and finally they closed the well. Ashby saw all this as unnecessary. In some marginal cases there might be room for disagreement, he admitted, but this had not been such a case. And as local medical officer he had 'unusual facilities for judging' and 'the advantage over any chemist who may have to form an opinion from arbitrary standards alone.'[10]
Ashby's complaint about distant consultants who used their prestige to pronounce on matters about which they really knew little reflects the increasing impatience with centralized water analysts of local sanitarians. In 1876, when Frankland and other senior chemists (including Wanklyn) were attempting to form the Institute of Chemistry as a means of ending this kind of scandal, the Sanitary Record was scornful. Any lack of trust in analytical chemistry was due to contradiction by those at the top, not to the incompetence of those at the bottom.[11]
The Liberation of the Medical Officers
By the late '70s local sanitarians had gone beyond complaining and were actively seeking a foundation for a decentralized water analysis. That search is manifested in three distinct endeavours. The first was the attempt to develop a flexible approach to water quality assessment especially suited to the diverse needs of medical officers of health. It is well represented in two of the main manual/text books for health officers, those by Edmund Parkes and Cornelius Fox. The second was the attempt to revive the sort of microscopy Hassall had done as the mainstay of water assessment. It is exemplified by J D MacDonald's Guide to the Microscopical Examination of Drinking Water . The third is the attempt in the late '70s and early '80s of the Society of Public Analysts to develop standard methods and interpretations for water analysis.
Although Edmund Parkes was the better-known sanitarian, Cornelius Fox was the more outspoken.[12] Fox's Sanitary Examinations of Water, Air, and Food (1878, 2nd edn 1886) was written by a medical officer for his colleagues. Fox put a premium on speed in analysis. He claimed to be able to do a rudimentary water analysis (free and albuminoid ammonia, chlorine, nitrates and nitrites, and total solids) in about 40 minutes, in contrast to the 'two or three months' that might elapse before one received a verdict from Frankland.[13] Fox also emphasized the importance of including all
evidence in forming an opinion. He revived a great range of qualitative tests—the odour of a water when heated, or the appearance of a residue when ignited—that had been discarded by more quantitatively minded chemists. The great weakness of the London chemists was their dogmatic faith in a particular parameter, he asserted, be it albuminoid ammonia or organic nitrogen: relying on the ammonia process alone might lead one to regard rain water as unsafe and fresh urine as safe.[14]
The key question, as Fox recognized, was not the one Frankland and Wanklyn were arguing about, of whose process was more accurate. Much more important was knowing whether the processes would lead the analyst to judge the quality of the water correctly. Leaving aside the particular results obtained, would an analyst using one of the processes reach the same conclusion as one using the other? Fox analysed 93 samples by both processes and in all but one case reached the same verdict; he could conclude only that Frankland and Wanklyn had been leading sanitarians on a quest for ultimate precision that was both futile and needless:
Apart from these very warm controversies . . . such disputes . . . do retard the progress of sanitary science, and lead the public to imagine that the whole question, whether a water is or is not pure, is a 'tossup'; this remark being generally clinched with the further reflection that it is universally acknowledged that doctors differ.[15]
But the main distinction between Fox and the London chemists had to do not with the acceptability of particular processes, but with the question of what analysis was for. Fox wrote that his intention was 'to treat [water matters] as a physician who studies them in connection with health and disease.' His book was to be as free as possible from chemical jargon—'from technicalities and all cloudy and chaotic surroundings,' as he put it. Analytical methods were to be simple enough that medical officers could learn them on their own, and analysis was to be only one among many tools for answering specific questions about water quality. Suspecting that a privy was contaminating a well the health officer was to look for specific signs of that contamination—high chlorine or nitrate levels, a peculiar odour, or a high level of albuminoid ammonia.
Thus, if analyses were to be useful it would be in terms of testing epidemiological hypotheses and the analyst would have to know a great deal about the circumstances of the water in question, including as much as possible about its origins—soil, drainage, what the land was used for. He knew of 'chemists [who] . . . would rather
know nothing about the sample' but held that 'an opinion so formed is worth very little. . . . the medical officer will always do well to obtain every item of information about it [the water] that it is possible to get.' No longer could analytical results be understood as providing ultimate and authoritative answers that would displace other forms of evidence. They were simply one type of information the health officer could use in coming to a diagnosis.[16]
In some ways this perspective resembled Frankland's. Frankland too recognized that analysis might be used to test hypotheses, even if it could not be expected to mirror nature in all particulars. For example, he used analysis to arrive at tentative generalizations about the oxidative capabilities of soils and the seasonal changes in rivers. Yet the kind of hypothesis-testing Fox had in mind, in which a local sanitarian would use certain carefully chosen analytical operations to track down the origin of a particular disease outbreak was not the sort of enterprise in which a distant London consultant, however elite, would be of much use.
Other sanitarians, though they did not go into the detail Fox had in re-orienting water analysis, shared many of his views. They returned to older and simpler processes, such as the ignition test for organic matter and the various approaches for comparing the colour of a water. They did not try to refute the theoretical objections that had been raised to such processes, but maintained that in the right circumstances these methods were good enough, and that they were easier to learn and to use.[17]
The Return of Microscopy
Among the approaches that began to come back into fashion were microscopical examinations. These were undertaken to look for signs of contamination, both life forms characteristic of sewage polluted environments and actual traces of sewage, such as bile-stained meat fibres. In Fox's view such methods held great potential for liberating local sanitarians from the irrelevant precision of the London consultants. With microscopy, local sanitarians could aspire to a similar level of precision. Moreover, it would be a field which they would have to themselves, since it was imperative that microscopic analyses be done on fresh samples.[18]
In the 1850s microscopical examinations of waters had been undertaken in connection with campaigns to end London's reliance on
polluted river water. Like Frankland's monthly announcements of the 'living and moving organisms' in the companies' waters, this use of microscopy had been a way of making the idea of impurity more vivid. By the late 1850s most analysts had abandoned microscopical methods. Hassall, though he continued to rely on microscopy in establishing a reputation as Britain's leading food analyst, acknowledged the superiority of chemistry in water matters. In his journal, Food, Water, and Air (begun 1871), he endorsed Frankland's views on water and recognized that Frankland was doing with chemistry exactly what he had tried to do with microscopy: deliberately setting out 'to alarm the public mind.'[19]
In 1874 Jabez Hogg, a London ophthalmologist and microscopist, revived the old Hassallian programme of trying to force London's water-drinkers to confront the disgusting impurities which the microscope alone could reveal. Hogg even allied himself with Samuel Homersham, the engineer who in 1852 had commissioned microscopical studies by Lankester and Redfern, and who was still advocating an alternative water supply for London. Hogg was a master at the rhetoric of disgust. He was especially fond of the term 'noxious'—in the water there were 'filariae and larvae of the most noxious kinds' and 'small fish, eels, and numerous noxious animals.' The idea that water was a 'breeding ground' for various life forms Hogg also found loathsome; he had a knack for making ordinary biological processes morbid and menacing. Life itself was disgusting; one was not to drink lake water because 'living organisms find a congenial habitat in the mud and water, and rapidly increase and multiply.'[20]
It might be expected that arguments like Hogg's would be taken increasingly seriously as the prospect that zymotic diseases were caused by bacteria or 'germs' became more and more likely. Hogg did find the image of 'contagious cells' to suit his needs well, and he attributed a number of specific diseases to various forms of microscopic life, including bacteria. Yet in 1874 bacteria were still not the primary concern and Hogg's arguments were not taken seriously; he was recognized as an extremist. For Hogg the whole microscopic population was abominable—if a species were not actively harmful he was sure it either predisposed one to disease or facilitated the development of something that was harmful. Indeed, so thoroughgoing was Hogg's condemnation of minute living things that he cut off all opportunity to develop microscopic analysis into a means for making distinctions among waters: if all life forms were as malign as they could be, what was the point of making any distinctions at
all?
The local sanitarians who were beginning to take an interest in microscopical water analysis in the late '70s were unconnected with the Hogg/Hassall polemics. They did, however, run up against the other problem that had plagued microscopists in the '50s, of whether there existed any set of microscopical distinctions that would tell anything one didn't already know. The most sober of the microscopical assays of the '50s had been done by the Bristol microscopists James Brittan and Robert Etheridge in connection with an 1854 cholera outbreak at Sandgate. They had begun their report by listing microscopic invertebrates commonly found in waters, along with the habitat of each. Then they reported the organisms found in the Sandgate water, noting that 'without exception, the organisms found . . . are such as would be detected in waters yielding a considerable amount of decomposing vegetable and animal substances.' On this basis they concluded that the water must have considerable 'organic matter in a state of disintegration and decomposition.' They went so far as to suggest which samples probably had the highest concentrations of organic matter, judging from the 'characters of the organisms found, and their known peculiarities as to habitat, and food, etc.'[21]
The structure of the Brittan/Etheridge report is striking. They were forthright in stating their interpretive standards at the outset (unlike Hassall who sometimes appears to be assigning species to habitats as he goes along). They applied their standards to the water and drew the appropriate conclusions. Yet however rigorous their procedure, the most Brittan and Etheridge could claim to be doing was to be using biological procedures to measure chemical variables. It was, after all, a chemical entity, decomposing organic matter, that sanitarians were interested in and there was no reason to suspect that that entity could not be measured more directly, easily, and accurately through chemical techniques. Had the biologists been able to make qualitative distinctions among types of decomposing matter, their work might have been helpful. But they did not even try to make such distinctions. Brittan and Etheridge had perhaps demonstrated the feasibility of microscopic analysis, but they had shown no reason why it should be adopted.[22]
In the late '70s and early '80s microscopists faced the same problem. The range of conceptions of the nature of the harmful substances in water had shifted over the years—concern with decomposing organic matter had given way to concern with certain sorts of putrefaction and eventually to fears of specific, quasi-living, dis-
ease germs. But the actual disease-causing entities were still for the most part unknown and invisible to the microscopist. The most microscopy could offer was to demonstrate the likelihood of sewage contamination.
The foremost authority on the microscopic analysis of potable waters during the period was J D MacDonald, MD, FRS, professor of Naval Hygiene at the military medical college at Netley, and later Inspector General of Hospitals and Fleets for the Royal Navy. MacDonald's Guide to the Microscopical Examination of Drinking Water (two editions, 1878 and 1883) was the main source for the discussions of water microscopy that Parkes and Fox included in their manuals.
MacDonald's claims for microscopical water analysis were modest. He thought that the 'known habitat of certain organisms detected should enable us, in a general way, to determine whether the water had been taken from a river, stream, lake, pond, or well source. Indeed, if we were more perfectly acquainted with the natural history of the forms occurring in a sample of water even in the absence of more definite information, we would have little difficulty in forming a conclusion as to the source from whence the water was derived.'[23]
Though a water's source was recognized as an important factor in its assessment, the analyst was rarely in the position of having to discover that. But even so limited a goal demanded a much greater knowledge of habitat than was currently available, MacDonald pointed out. In fact, his book was closer to a traditional taxonomic guide, listing species and providing instructions for distinguishing them from one another, than it was to a handbook for microscopical water analysis. MacDonald included information on habitat in some cases, but he did not make a point of doing so in all. This was probably wise, since there were sometimes disagreements among microscopically minded sanitarians as to which organisms indicated which habitats. For example, A Wynter Blyth, following Hassall, insisted in 1874 that paramecia indicated bad water, yet Parkes wrote a few years later than 'subsequent observations have not, however, proved the relation between paramecia and animal matter in the water to be sufficiently constant to allow the former to be used as a test of the latter.' Similarly, ciliated forms indicated sewage to Blyth, while Parkes noted that there were many different kinds of these, and that they showed nothing more than the presence of 'vegetable or animal organic matter.'[24] Reluctantly, he admitted that microscopical analysis was of limited utility: 'So many are the
objects in water that the observer will be often very much at a loss, first to identify them, and secondly, to know what their presence implies.'[25] Fine distinctions were rarely possible. There were exceptions—on occasion a microscopist might discover a clear trace of sewage, one which signified contamination more convincingly (and graphically) than could any purely chemical parameter.[26]
Some British sanitarians, especially those also involved in food analyses where microscopy was central, did make a practice of including microscopical examinations in their water analyses, but it is likely that they did so not so much for what microscopy could show but because it was one means of characterizing water that was available to them. Notably in Germany, France, and the United States the correlation of microscopic aquatic species with aspects of water quality was taken more seriously than it was in Britain. Independent traditions of diagnostic stream ecology developed in each of these countries. It was the German tradition, embodied in the saprobeinsystem of R Kolkwitz and R Marsson, that reignited the interest of British analysts in biological approaches in the first decade of the twentieth century.[27]
The Initiative of the Public Analysts, 1878–84
Among the most persistent advocates of microscopy in water analysis was George W Wigner.[28] Wigner is not one of the better-known analytical chemists, but he had been working in sanitary chemistry at least since the late '60s when he was one of the founders of the ABC Process, the most famous and long-lived of the sewage precipitation processes. In February 1878 Wigner read a paper on water analysis to the newly formed Society of Public Analysts, an organization restricted to those who had an official appointment as a public analyst. The society he addressed was not wholly typical either of learned or of professional societies. It had been founded in 1877 shortly after the office of public analyst had been made mandatory for urban sanitary authorities. While it was concerned with the advancement of knowledge and with the welfare of the nascent profession of the public analyst, the society spent much of its time trying to keep up with the rapidly evolving practices of food-and-drug adulterers and with the legal and technical complications encountered in bringing such villains to justice. In a profession in which consensus on technical matters had profound legal implications, matters of standard methodology were of great import.[29] Wigner shared many
of the views of Fox, Parkes, Ashby, and Charles Cassal, a cadre of sanitarians, many of them young, who were critical of the domination of water analysis by Frankland and Wanklyn. Like Fox, he held that an analyst ought to base an opinion on as wide a range of information as he could; like Ashby and Cassal, he was distressed at the frequency with which water analysts contradicted one another, and the apparent arbitrariness of their opinions of a water.[30] In Wigner's view, the time had come to designate a wide-ranging set of qualitative and quantitative tests to be included in every analysis and to translate the results of each test into a common numerical scale so that at a glance anyone could compare results of two samples simply by comparing the sums of the results of all the tests done on each. The summing of the individual results would automatically ensure that all processes were represented in the conclusion; the common point system would ensure that all chemists would be weighting the results in the same way and thus would arrive at the same conclusion about any given water. It was an ingenious and wonderfully simple idea. The only questions were whether analysts could agree on the weighting and whether they would be willing to substitute a simple formula for the traditional freedom of drawing whatever conclusion one chose on the basis of one's consummate expertise. On both points Wigner was a bit naive.

Figure 8.2
The first version of George Wigner's formula for evaluating waters, presented to the
Society of Public Analysts in 1878. The concept of a uniform scale bothered Wigner's
colleagues, and even after massive revision the scale was not adopted
(The Analyst 2 [1878]: 215).
In the initial version of the scale, Wigner used the quantity of one thousandth of a grain per gallon of albuminoid ammonia as a base—this was to equal one degree of impurity. The rationale was that this albuminoid ammonia was both the 'most injurious factor' discovered in the analysis and the smallest quantity it was useful to consider. All the other determinations and observations were then assigned values in accord with the degree of danger or unsuitability they posed in comparison with this albuminoid ammonia—nitrites were awarded one point per every two thousandths of a grain; free ammonia received a point for every two hundredths of a grain, and so forth. Wigner also awarded various numbers of points for total solids, weight lost on ignition, chlorine, nitrates, oxygen absorbed from potassium permanganate, hardness, and traces of lead and copper. Each determination was essential for a thorough analysis, he argued, hence each ought to figure in the classification of a water.
Wigner realized that while his views on the appropriate weighting of various substances might be questioned, standardization was feasible for quantifiable parameters. He faced a greater problem incorporating physical and microscopical observations for these were qualitative. He stipulated that a good-tasting water was to be given no points, one with a 'slightly saline' taste one point. A taste of 'decayed leaves' or 'flat rain water' warranted two points, a 'decidedly offensive' taste received six as did a water which emitted a urinous odour when heated. Colour was also important, worth as many as nine points in the case of a water which was an 'opaque yellow' in Letheby's two foot tube. The microscopical evidence was valued mainly in terms of its mass, along with the certainty with which it indicated sewage contamination. The presence of bacteria warranted three or four points, depending on their quantity. 'A few living organisms' received six points, 'animal remains' twelve, and traces of urea and partially digested muscular fibres eighteen. Waters with scores under 35 were to be regarded as satisfactory, those scoring between 35 and 55 were 'second class,' those between 55 and 75 were 'suspiciously dangerous,' while any sample exceeding 75 was to be regarded as 'sewage.' To ensure greater unanimity in making the qualitative determinations, Wigner hoped to make solutions of standard colour, taste, and smell, and sets of standard microscope slides.[31]
The expectation underlying the scheme was that a water impure according to one test would be impure according to others; the tests would overlap and amplify one another. Yet as Wigner himself knew,
there were instances in which this was not the case—the danger in a water would show up in only one of the analytical operations done on it or not at all. Wigner admitted that he had examined waters recently contaminated with urine in which the analyst's only clue was the presence of solid urea or urates in the residue. In such cases there would be little albuminoid ammonia and possibly little free ammonia.[32] Hence the summing process, which was intended to ensure that each measurement figured in the conclusion, might actually hide the significance of that single measurement which represented the dangerous impurity. In such cases Wigner considered that 'the water should be condemned on the result of that examination alone.' But such a stance simply re-raised the issue of the utility of a numerical scale. If it remained essential to consider the result of each test independently and then to treat certain danger signs as valid reasons to ignore point totals, why then do the summing at all?
The Wigner scale did not arouse much interest until 1881 when a committee of the Society decided to institute a programme of regular and uniform analyses of the public water supplies of all towns in England. Making up the committee were Wigner, John Muter, August Dupre, A Wynter Blyth, Otto Hehner, Bernard Dyer, and Charles Heisch, all experienced analytical chemists who had managed to avoid the polarization in water analysis issues of the early '70s. Three of the members, Dupre, Hehner, and Heisch, had even at one time or another taken up problems in the methodology of water analysis.
Water analysis was not a part of the public analyst's job and the committee's existence probably reflects members' professional interests more than their specific concerns as public analysts. Public analysts had private practices too, and as an editorial in The Analyst pointed out, water analysis would normally be 'a fairly remunerative part of an analysts's practice.'[33] In announcing its decision to undertake the analyses, the committee made it clear that it was adopting the perspective of Col. Bolton, the metropolitan water examiner, and all who were bewildered by the proliferation of contradictory analyses in incommensurable formats. They envisioned a time 'when the same water sent to every Public Analyst in England will be returned with the same opinion, just like an analysis of milk or butter.' As Heisch recognized, this change would require a different attitude on the part of water analysts; they would have to get used to 'sinking their individual opinions and working loyally on the lines laid down by the majority to secure that which all must
consider of the utmost importance—uniformity of results.'[34]
Ignoring the central issues of the Frankland—Wanklyn dispute, the committee chose processes likely to be the 'most rapid and reliable for such systematic analyses.' These were the oxygen absorbed (permanganate) process and Wanklyn's ammonia process, and several others Wigner had listed: physical and microscopical analysis, measurement of phosphates, nitrates, chlorine, hardness, and total solids. They issued a detailed set of instructions for these processes, the first appearance in Britain of anything resembling a set of 'standard methods' of water analysis. These were sent to all public analysts and given out free to anyone who requested them. So great was the demand that within the first year the Society had commissioned second and third printings.
The January 1881 number of The Analyst carried the first set of analyses of the waters of forty towns done according to this system. Even at the outset there was tension as to what, if any, interpretation of the results the committee should make. Much of the dissatisfaction with water analysis in Britain was with the extreme interpretations made by such men as Frankland and Wanklyn. Initially, the Society elected to offer no opinions whatever on the results it received each month; their value was to lie solely in the comparisons they allowed one to make of the waters of different towns. Since the returns were detailed those 'in the habit of collating such results' would be able to 'form a fair judgment for themselves.'[35] Thus the committee had accepted half of Wigner's proposal—uniformity of method—while ignoring the other half—uniformity in interpretation. It was not a satisfactory compromise: a central concern, for both Wigner and the committee, was that analyses be meaningful to the general public. For this reason they had rejected metric units in favour of the more familiar grains/gallon (1:70,000). Yet in leaving the facts uninterpreted, they were only returning responsibility for interpretation to other experts who alone could make sense of them.
In June 1881 Wigner presented the Society with a revised version of his scale. He assumed his colleagues would agree with him as to the value of uniformity in interpretation and he hoped they would be able to agree on a system itself. Wigner had fiddled with his scale a good deal during the previous three years, leaving out the taste test, for example, and changing the number of points assigned for other constituents. More importantly, he had tried out the system on several sets of analyses to ensure that his point totals actually corresponded to the opinions experienced analysts would be likely
to give. He also tried to verify that the valuing system would clearly signal important changes in water quality, such as the discharge of a sewer into a river.
Despite these revisions and validations (and Wigner's willingness to compromise), when it came time to endorse the scale the membership balked. The discussion never even got to the details, it was the principle of uniformity itself that was called into question. The main objection was that it was erroneous to think that all one needed to know of a water was contained in analytical returns. Several speakers held Frankland's view that the most important factor in interpreting an analysis was knowledge of the water's history. Some northern towns, they pointed out, got moorland water that was heavily laden with organic extracts from peat. Such waters would receive high scores on Wigner's scale, though they were generally judged unobjectionable, being completely free of sewage contamination. Likewise, waters from deep wells frequently contained large amounts of nitrates and even nitrites and ammonia. These too were usually considered excellent waters, yet on Wigner's scale they might yield scores as high as 70 points, far above the fitness range. It seemed unfair to compare cities with such waters with other towns which might have lower scores, yet poorer water.
There was also concern that even though it was a good idea to consider all types of analytical evidence, it was important to be able to condemn water if it proved unsatisfactory in only one category, regardless of its point total. J W Tripe, medical officer and public analyst for some of the east London vestries, was bothered that microscopical evidence, no matter how damning, warranted no more than ten points. Some of the bacteria observed through the microscope might well prove to be the carriers of typhoid fever or some similar disease and yet in Wigner's scheme their presence was still not enough to condemn the water.[36]
The Society postponed action on the proposal but allowed Wigner a trial: The Analyst would publish the scores of the samples analysed as part of the programme of uniform analyses, but the scores were, however, to be printed on a separate page from the analytical results themselves. For nine months, from July 1881 to March 1882, Wigner listed the score of each water for that month, its score for the previous month, and its average score for January—June 1881. Thus anyone, no matter how untrained in chemistry, could at a glance see whether his water was improving or deteriorating and how it compared to the waters of other English towns. There were some
striking vacillations in quality. The water at King's Lynn fell from 110 to 48 in a month and was back to 110 a month later; Darlington's water rose from 39 to 96 in a month.[37]
In February 1882 the Society returned to the question of a common scale and voted 25 to 11 against adopting the Wigner scale. They also decided that Wigner scores would not be published in The Analyst unless they were submitted by the chemists who had done the analyses. In the face of this opposition, Wigner and Muter, who were co-editors of The Analyst , decided to end the experiment.
In April 1883 Dupre and Hehner presented a modification in which standards would be calibrated for each region, and two months later Muter offered another approach to the standards problem,[38] but neither suggestion generated much interest. The fight to establish a chemical formula for good potable water had been lost. The Society's program of uniform analyses ended in November 1882, at the end of two years. Wigner and Muter noted that the analyses had been made 'at considerable cost . . . by over fifty analysts . . . without any payment from the Water Companies or Corporations who have supplied the water, but simply for the purpose of disseminating . . . knowledge.' Nearly a thousand had been done, 'the largest series of uniform analyses of water supplies which have ever been published by any private body of analysts.' But while they insisted that the goals of the project—'to draw public attention to the character of the water' and 'to give facilities which were not then available for judging of the relative qualities'—had been achieved, it is clear that the membership was losing interest. The last of the tables included analyses of only 19 supplies outside London; at the start of the year there had been 39.[39]
It was the unwillingness of chemists to give up the professional's prerogative of independent judgment that made Wigner's initiative ultimately impracticable. The Society had begun its analyses, and Wigner had taken up the cause of uniform interpretation, out of the conviction that some chemists were using the trappings of professionalism as an excuse to justify their arbitrary interpretations and their use of processes not sanctioned by the profession as a whole. Wigner and the SPA had hoped to stop this by insisting that chemists use a standard set of processes and in Wigner's case by insisting on a common set of interpretative rules. Yet even during the two or three years when the attractions of uniformity seemed most compelling, chemists did not give up the conviction that independent judgement was an essential part of responsible professionalism.
The conflict was expressed most clearly by Dupre and Hehner in their defense of local standards. Universal standards had two negative consequences, they pointed out: they 'weaken[ed] the feeling of personal responsibility of the analyst,' and they gave 'a spurious belief in the possession of knowledge to the ignorant.' Good chemists would know when to discard standards, but that only showed that good chemists did not need standards, for they interpreted analyses 'according to the circumstances of every particular case.' There was no escape from the onus of judging, and no expedient way to abolish contradiction:
what we wish to impress on our fellow analysts is this—by all means take into consideration and, on suitable occasions, make use of such general standards as have been laid down by chemists of high ability and large experience; but use these standards cautiously and with discrimination, and judge every case on its own merits. Analysts who lack either the ability or the experience to stand on their own legs, and slavishly adopt standards laid down for them by others, have no business to meddle with water analysis at all, and the sooner they leave such work to their more experienced brethren the better it will be for themselves and for the credit of water analysis.[40]
The Mallet Report
In terms of the distribution of authority among British sanitary chemists the most serious challenge to the dominance of Frankland and Wanklyn in the pre-bacteriological era was that mounted by the Society of Public Analysts between 1878 and 1882. They declared independence from the London chemists and insisted that the average public analyst was not only competent to analyse water, but the right person for such work, since public analysts could better know the circumstances of the analysis and could guarantee results according to a common format.
On the other hand, the most serious scientific challenge to Wanklyn and Frankland was an 1882 report on the accuracy and utility of the main processes for the analysis of organic matter in water, sponsored by the United States National Board of Health and carried out by a team of chemists and medical men under the direction of Professor J W Mallet of the University of Virginia. For nearly fifteen years Frankland and Wanklyn and their partisans had exchanged accusations. They had come up with unanswerable arguments explaining why the other's process could not possibly be
accurate, yet no large-scale, systematic comparison of the two (or three, if we count Letheby's oxygen-absorbed approach) processes had been done to determine whether these arguments were valid. American and continental water scientists found the British scene hard to comprehend. They recognized that water quality was taken more seriously in Britain than elsewhere but were baffled by the vehemence of a controversy about how to measure an unknown and unmeasurable entity. Mallet's report emerged from this background. It was the first systematic attempt to assess exactly what the British chemists had accomplished in the development of analytical processes, the first investigation to provide definitive answers to the controverted questions. There were five of these: first, were the processes replicable; second, did any of them permit one to distinguish between the different sources of contamination waters might be exposed to; third, how accurate were the processes in measuring what they claimed to be measuring; fourth, did any of them have the capacity to allow the distinction of water known to cause disease from water known to be safe; and fifth, were biological or microscopical means of analysis any more satisfactory?
Let us take these in order. Especially in connection with the combustion process, the problem of replicability was one of the most troublesome questions for British water analysts, and the one with the most direct implications for the structure of the profession. Even if the combustion process gave accurate results in Frankland's lab, could it be successfully used by others? And if it could not what were the reasons? Mallet faced the problem directly. Three experienced analytical chemists, W A Noyes of Johns Hopkins, Dr Charles Smart of the U S Army, and Dr J A Tanner of the U S Navy, independently examined portions of a split sample. The three chemists had trained themselves in the use of the processes together to ensure as much uniformity as possible in their operations and they each analysed the sample on the same day to control for possible alteration of the substances in the water. The results were unexpected. The simple permanganate process of Letheby and Tidy gave the best results, but the ammonia process, widely viewed as giving concordant results, turned out to be not much better than the notorious combustion process, which was supposed to be too tricky to yield concordant results.[41]
The second and fourth questions were crucial. Did any process of analysis permit one to distinguish different types of contaminations, including waters which there was strong reason to think had caused
disease outbreaks? Surprisingly, this fundamental issue had rarely been directly addressed by British analysts. Frankland, of course, had admitted as early as 1866 that it was impossible for chemical analysis to distinguish water that caused disease from water that didn't. While most sanitarians probably agreed with Frankland, no one had gone to the trouble of systematically collecting data to discover how well analyses could distinguish bad waters—here too the debate had been conducted through a series of assertions and denials. The results were not encouraging. The quantities of free or albuminoid ammonia or of the oxygen absorbed from permanganate could really tell the chemist very little about what was in the water, Mallet maintained, though the analyst could obtain some useful information from each of these processes by 'watching the progress and rate of the reaction,' rather than paying attention to the quantity finally determined.[42] As for the most important question of whether any of the tests allowed one to distinguish harmful water, none did. 'No one could, with these figures to guide him, refer a water of unknown origin to one or the other of the two classes [waters known to be safe and waters known to cause disease] on the evidence afforded by chemical analysis, using any . . . of the processes in question.' There was one possible exception to this generalization. Waters known or suspected of being harmful did have significantly higher levels of nitrates and nitrites than safe waters. This was not wholly unexpected; we may recall that in 1867 Frankland had made the claim that such compounds, along with ammonia, constituted 'previous sewage contamination' inasmuch as they were the oxidation products of sewage or similar materials. The idea had remained controversial throughout the '70s and Frankland had quietly dropped 'previous sewage contamination' from his analytical returns in the beginning of 1877. Yet by 1883 Ashby and Hehner were reviving it, and maintaining that Frankland had not gone far enough—nitrates, etc. did not reveal previous sewage contamination so much as they demonstrated quite recent sewage contamination.[43]
What was unexpected was that these substances alone should be the peculiar signal of danger. In Mallet's view the link between high nitrates and dangerous pollution was bacteria. Citing the 1877–78 nitrification experiments of the French bacteriologists Theophile Schloesing and Achille Muntz, he speculated that the reason nitrates were associated with pathogenic water was that the microbe responsible for nitrification was probably also pathogenic. This was by no means implausible. Like many British scientists, Mallet had a vague
belief in some sort of germ theory. Yet so little was known about the types of germs, and methods of detecting, isolating, and identifying microbes were still so rudimentary that simply to make a tentative association between disease and microbes was quite reasonable.[44]
It was the third question of which process most accurately measured organic matter (or something similar), that was the focus of so much of the Frankland-Wanklyn conflict. Mallet tried the processes on aqueous solutions of known strength of pure organic compounds and chose substances such as salts of butyric and valerianic acids that were exactly the sorts of things generated in putrefaction.[45] He found that both Frankland and Wanklyn were accurate in their criticisms of the other's process. On average the combustion process measured barely half the organic carbon present, but recorded an average of 118 per cent of the organic nitrogen.
In the case of the ammonia process on the other hand, 24 per cent of the albuminoid and 28 per cent of the free ammonia were generated on average. Contrary to Wanklyn's claim, the process did not accurately indicate the amount of putrescent matter. As for the permanganate process of Letheby and Tidy, the pure substances absorbed only about 13 to 16 per cent of the oxygen needed to oxidize them. Contrary to the claims of these chemists, the test did not discriminate particularly putrescible substances.[46]
All in all the combustion process supplied the closest approximation of organic matter. It was not so much that combustion was accurate, but that its competitors were more inaccurate. Yet Mallet denied that combustion could be regarded as a way of truly 'determining' organic carbon and nitrogen: 'it is a method of approximation, involving sundry errors, and in part a balance of errors.'
To answer the fifth question, Mallet commissioned H Newell Martin and E M Hartwell, both of Johns Hopkins, to classify samples on biological grounds. By itself, this biological method, even when supplemented by bio-assay tests in which waters were injected subcutaneously into rabbits, proved no better than the chemical methods in discriminating safe from deadly waters. Of 19 safe waters Martin marked four as suspicious, Hartwell seven. Of nineteen waters known to have caused disease, Martin marked none as 'dangerous' and two as 'suspicious,' while Hartwell marked none as 'dangerous' and one as 'suspicious.'[47]
Even in this brief summary the thoroughness of Mallet's approach should be clear. In his conclusions Mallet was unsparing. He saw his task not so much as choosing which of several problematic ap-
proaches was the best—the way the issue was framed by British chemists—but of laying out as fully as possible the errors to which each was subject. He had no loyalty to the state of the art; he saw no need to put the best face forward and insist that whatever problems there might be, the processes of water analysis were good enough. Yet however inadequate analytical processes, both biological and chemical, might be, analysts faced the necessity of coming to some decision about water quality on the basis of the methods available to them and Mallet recognized this. He divided his recommendations into three parts: the general problem of how waters should be evaluated, ways analysts could better utilize the existing processes, and substantial improvements to eliminate the more egregious sources of error. But even these improvements would not make the processes any more capable of detecting the deadly forms of pollution.
Most important are Mallet's 'General Conclusions.' In the tradition of Frankland he maintained that measurements of organic matter or its derivatives did not allow one to distinguish safe from unsafe water. Chemical tests had to be integrated with, and even subordinated to, knowledge of a water's history: there could be no general standards for distinguishing water quality of the sort Wigner was trying to formulate. In only two limited contexts would analysing the organic matter in water help. One was to detect unusual cases of gross pollution such as might occur through a leaking sewer. The other was to chart variations in the quality of an urban water supply. In such cases all three processes should be used.
In most respects, then, Mallet's perspective resembled Frankland's. Both were skeptical that water analysis could yield the information most important to the authorities responsible for public health. Yet in a footnote Mallet distanced himself from the Franklandians:
it will not do merely to throw all doubts on the side of the rejection of a water, as has been more or less advocated by writers on water analysis [Frankland], for there are often interests of too serious character involved in such rejection to admit of its being decided on, save upon really convincing evidence of its necessity.[48]
However infelicitous the prose, the meaning is clear. The burden of proof ought not to rest wholly with those who advocated a particular water, where Frankland hoped to place it. To do so would be to put the advocates in an untenable position. Aside from the philosophical impossibility of proving a negative (that the water would do no harm), they would be forced to draw far stronger conclusions
from water analysis than it could conceivably supply. However untrustworthy analyses were, cities needed water, and in Mallet's view just because one could not prove a water to be safe was no reason to think it unsafe. Unless there were strong reasons to think a water was unsafe it could be used.
A short version of the Mallet report was published in Britain in the Chemical News , the weekly organ of the profession, and the report was widely commented on in other journals.[49] To modern readers Mallet's report is apt to seem modern and common-sensical in its continual recourse to experiment to answer questions as they arise. It seems to clear away the mass of heated argument that had characterized British discussions of water analysis since the 1866 cholera. One is tempted to ask why no British chemist or group of chemists had produced anything similar in scope during that period. Two factors are important. The first has to do with the complexity of the sort of study Mallet carried out. It is not clear who in Britain would have sponsored such a study, though it could be argued that the project was not fundamentally different from collaborative researches organized by the Local Government Board or the British Association for the Advancement of Science. Other institutions—the Society of Medical Officers of Health, the Sanitary Institute of Great Britain, the Chemical Society, or the Society of Public Analysts—might also conceivably have undertaken such work. In fact the LGB did sponsor such an investigation in 1881, a short report by R D Cory showing that the ammonia, combustion, and permanganate processes were incapable of distinguishing waters contaminated with the excreta of typhoid sufferers.[50] (Indeed, a sample contaminated with typhoid stool would have shown up as excellent according to Wanklyn's standards.)
The second factor is more important and has to do with the social context of British water analysis during the period and the contrasting context in which Mallet worked. In Britain processes of water analysis had been developed and adopted or rejected in connection with disputes for political control of water supplies—the cases of Frankland and Tidy are exemplary in this respect, where social and political concerns informed the selection of analytical methods and the presentation and interpretation of analytical results. There was no neutral ground on which processes might be compared and evaluated. Men like John Simon and William Farr were hardly in the position to provide an umbrella of unimpeachable credibility for scientific research projects; they were far too ready to exploit science
to rationalize their own social reforms. It was a system with some built-in constraints. In such a context conflict was more important than cooperation, credibility was more important than utility, political achievement was more important than scientific progress.
This context was foreign to Mallet. The National Board of Health, which sponsored the tests, was a new agency. It was controversial, but that had nothing to do with its evaluation of water analysis or similar matters. Simply by virtue of the fact that he was an American chemist Mallet was outside the ongoing conflicts that had produced such disunity among the British chemists. He could look upon the analytical processes as products to be judged, not recognizing the extent to which they reflected Wanklyn's thirst for revenge against Frankland, Frankland's crusade to liberate his countrymen from water-borne disease, the attempts of Letheby, Tidy, and Odling to represent the London water companies by countering Frankland's sensationalism, or the rivalries between the provincial chemists and the London consultants over what kind of institution water analysis was to become and who would be the analysts. In short, the devastating criticisms of Mallet (and Cory) were fine so long as one did not have to practice as a water analyst.[51] All this Mallet could ignore, yet all these factors had produced the products Mallet was evaluating, and would continue to shape water analysis even in the era of bacteriological analysis that was about to begin as Mallet's report was published.