The Wanklyn Affair
Crookes' criticisms were occasional; he had other polemics to occupy him. Such was not the case with James Alfred Wanklyn, for whom revenge on Frankland became a raison d'être . Wanklyn's criticisms of Frankland were the longest sustained, the bitterest, and yet the most substantive. Between 1868 and 1877 the relative merits of Frankland's combustion and Wanklyn's 'ammonia' processes dominated discussions of water analysis in Britain. In Wanklyn's view the dispute was over which process better measured the harmfulness of a water. To Frankland such a quest was futile; analysis was to shed light on a water's history. Nonetheless, the debate was conducted largely on Wanklyn's terms, it being assumed by most chemists that the process that more accurately determined the quantity of putrescible matter gave the better measure of harmfulness. Indeed, quibbling about parts per million of organic nitrogen or 'albuminoid ammonia' completely displaced the more important questions of what, if anything, these entities signified. Still, Wanklyn's
criticisms are important: his attacks on Frankland's character damaged Frankland's credibility; his substantive criticisms of the combustion process were extensively used by others who knew little of chemistry and whose reasons for attacking Frankland lay elsewhere.
In its early stages Wanklyn's career had paralleled that of Frankland, his senior by nearly a decade. A Lancastrian like Frankland, he had been apprenticed to a Manchester doctor and later studied with Frankland at Owens College in the early '50s, where he was Frankland's assistant. Like Frankland he took advanced training at Bunsen's laboratory at Marburg and with Frankland's assistance secured appointment as a demonstrator to Lyon Playfair in 1859. From 1863 to 1870 Wanklyn was a lecturer at the London Institution; he spent the rest of his career as public analyst to a number of authorities, as a lecturer at St George's Hospital, and in private practice as an analyst.[23]
In June 1867 Wanklyn described to the Chemical Society the new 'ammonia' process that he had developed with E T Chapman and Miles H Smith. The process was based on the belief that albumin was the dangerous material in water since albuminoid substances putrefied unusually rapidly. Wanklyn believed that a definite proportion of a water's albumin was converted to ammonia when the water was distilled with a caustic solution of potassium permanganate. The amount of this 'albuminoid ammonia' could be determined by the Nessler colour test, a common and well accepted approach for quantitative determination of ammonia. This quantity was to be the main index of water quality.[24]
Wanklyn's process was thus made public in the middle of the same year that Frankland was developing the combustion approach. Both were working in the Hofmann tradition and were deeply concerned about the types of organic nitrogenous matter that might undergo dangerous decomposition. Like Frankland, Wanklyn rejected existing evaporation techniques. He ridiculed Miller's call for uniformity in analytical procedures and in the statement of analytical results, arguing that to pursue uniformity on Miller's terms would merely be to multiply comparable results of doubtful accuracy.[25]
Initially British chemists were not impressed and Wanklyn and his collaborators quickly found it necessary to moderate their claims.[26] The first Wanklyn–Chapman–Smith paper had reported trials on natural waters. When Wanklyn and associates tried to verify the process by using it on artificial solutions of pure organic compounds they ran into some surprises. In early experiments pure albumen
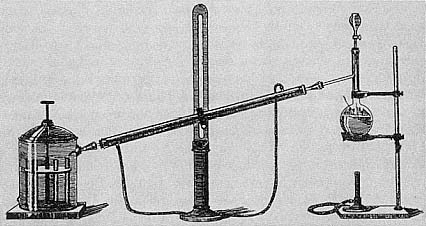
Figure 7.1
Wanklyn's ammonia process was for most British chemists the central technique
for determining the salubrity of potable water during the '70s and '80s
(From T B Stillman, Engineering Chemistry [Easton, PA: Chemical
Publishing Co, 1897], p 75).
(from egg white) gave up two/thirds of its nitrogen as ammonia. But other nitrogenous compounds apparently gave up all, or a half, or a quarter, or even a seventh of their nitrogen as ammonia. When the empirical results were multiplied by the proper factor there was close correspondence between 'theory' and observation, and to Wanklyn this was proof of the soundness of the process: substances were breaking down in an orderly way and yielding a definite proportion of their nitrogen as ammonia. Yet he was able to offer a truly theoretical explanation in only one case. To other chemists the process looked arbitrary. Wanklyn's 'theory' seemed nothing more than selecting the nearest whole number which when multiplied by his empirical result would give a result close to the calculated level of ammonia. Moreover, the question remained of what trials on pure substances implied for natural waters. In Wanklyn's view there was nothing to worry about: natural waters could be relied upon to yield a definite and unchanging proportion (two-thirds) of their nitrogen as ammonia.[27]
It may seem that the rationale for the ammonia process was flimsy. It was. The so-called 'albuminoid ammonia' was not a natural substance but an artefact of the analysis. Nor was it clear whether its quantity bore any relation whatever to harmfulness, potential for harmfulness, or even putrescibility. (In practice sanitari-
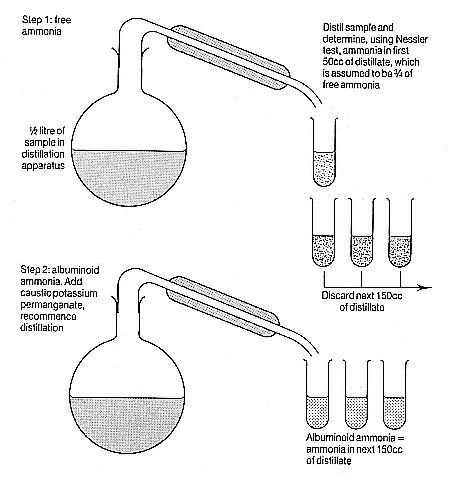
Figure 7.1
(continued)
ans would come to minimize these problems; as Fox put it, the yield of albuminoid ammonia was seen 'to keep pace with the purity or impurity' of waters.[28] )
Indeed, it is not too much to say that the launching of the ammonia process was a disaster and that Wanklyn and his colleagues made fools of themselves in it. Chemical News published a savage review of Wanklyn and Chapman's handbook on water analysis, whose authors suffered from
an excess of scientific fervour, a sort of scientific afflatus , which forms a part of the unconscious poetry of these gentlemen's natures, and which impels them to burst forth in paeans at the Chemical Society whenever any one of them conceives a new idea. We may admire such gushing enthusiasm, but we cannot but regret that the scientific
fame which these gentlemen are acquiring . . . should be sullied by the publication of raw, incomplete, and sometimes inaccurate results.[29]
Even had there been no Frankland and no combustion process to contend with, Wanklyn would have been in for rough treatment from his peers. But for him it was Frankland who came to symbolize the injustice that could be expected from the elite of chemistry.
Their dispute began in June 1867. On the same day that he formally presented the ammonia process to the Chemical Society Wanklyn testified about it to the Royal Commission on Water Supply. He said that he had been in frequent contact with Frankland, who 'had admitted . . . the extreme value of the [ammonia] process' and was testing it. He was familiar with Frankland's attempts to measure organic nitrogen through combustion and claimed that Frankland had now come to recognize that that approach was 'unsatisfactory to the last degree.'[30]
Given Wanklyn's bravado on June 20, the events of ensuing months must have been deeply humiliating. Frankland and Odling did test the ammonia process in their work for the Water Supply Commission, but in a way that was completely unfair in Wanklyn's view. They compared results obtained with the new and unverified ammonia process with those obtained with the new and unverified combustion process. Finding the two processes incompatible they rejected the ammonia process as unreliable. The rejection was not in fact quite so high-handed as it may seem—the combustion process was intended to measure organic nitrogen directly according to a simple and well-accepted principle while the ammonia process depended on the novel and somewhat dubious (and soon discarded) claim that the caustic permanganate would convert all organic nitrogen to ammonia.[31]
Matters came to a head in January 1868 when Frankland publicly introduced the combustion process in a Chemical Society lecture. He made short work of the ammonia process: it had been surpassed. The discussion took up the merits of the rival processes. Campbell, Odling, and Frederick Abel spoke against Wanklyn. Wanklyn maintained that in Frankland's own trials the combustion process had produced errors sometimes larger than the amount of organic nitrogen likely to be in the water. He and Chapman raised what seemed insuperable problems in Frankland's procedure. If combustion were to measure organic nitrogen accurately, it was necessary that all nitrates be destroyed during the initial evaporative step, yet Chapman still found nitrates in the residues of samples evaporated according
to Frankland's instructions. Further, any volatile organic materials would be lost during evaporation. Any attempt to eliminate one of these errors would exacerbate the other. Efforts to ensure total destruction of nitrates such as adding more sulphurous acid would likely increase volatilization.[32]
Wanklyn's points were well taken: 'one of the most formidable pieces of criticism which we have ever met,' noted the British Medical Journal . Throughout the early 1870s others of Frankland's critics reiterated these objections and Frankland himself devoted great attention to resolving these problems.[33] But substantive criticism was not Wanklyn's style. The ad hominem attacks made on him during the early days of the ammonia process, and what he saw as the ruthlessness and arbitrariness of Frankland's rejection of the process, played upon his paranoia. Chemical objections became secondary; he came to believe that he was a victim of a conspiracy: Frankland was the head of a washed-up elite of 'chemists whose activity does not take the direction of . . . original research.'[34] In 1871 he wrote a 'History of the Ammonia Process,' partly to warn 'younger chemists . . . [of] the sort of reception which awaits them at the hands of their chemical brethren in this country, should they be so unfortunate as to make any notable advance in chemical methods.'[35]
Ironically, as Wanklyn became increasingly bitter and increasingly isolated, the ammonia process was becoming more widely used. In 1876 Frankland himself acknowledged that the ammonia process was 'now almost [as] generally used by analytical chemists as were formerly the incineration and permanganate processes.'[36] Using the process did not necessarily mean sympathy with Wanklyn, however; many who used it had little concern with the feud or even with the validity of the two processes. The ammonia process was adopted because it was easy to learn, easy and cheap to use, and because however weak its rationale, chemists saw it as a reliable way to distinguish good water from bad. So long as one did not attach too much significance to absolute quantities of free and albuminoid ammonia, but used the results to suggest further investigations or discover changes in the condition of a particular water supply, the ammonia test was useful. Thus, ironically, the test came to have exactly the significance Frankland thought water analysis ought to have—its results were to complement other information, not to dictate whether water was safe.[37]
By the early 1870s Wanklyn had attacked most aspects of Frankland's water science and had systematically taken positions opposing
Frankland's. He had allied himself with the London water companies, defending them from Frankland's criticisms, and reversing his own earlier position.[38] In 1872 he ridiculed 'previous sewage contamination' and accused Frankland of using it for political purposes. In 1872 and again in 1879 he began programs of alternative analyses of London's waters. By employing a chemist (Frankland) who defiantly used 'illusory and defective methods,' the government had shirked its duty; Wanklyn would selflessly perform that duty.[39] He repeated the analyses of the Cumberland and Welsh waters examined by the Water Supply Commission and found them no better than the Thames.[40] In 1878 he accused Frankland of having stolen his work in the years during which he had been Frankland's assistant and noted that Frankland's great discoveries had ceased when he had left Frankland's lab.[41]
However much social factors may have kept Wanklyn from fulfilling his potential as a chemist, it is hard to grant credence to the accusations or to sympathize with the accuser: the complaints are inconsistent, incoherent, and hysterical. Even as chemists were adopting the ammonia process as an easy and useful component in a broadly based empirical approach to water analysis, Wanklyn continued to insist that it was the ultimate in water analysis and that albuminoid ammonia should be regarded as the harmful material in the water (no matter what form this was presumed to take). As W H Brock has pointed out, Wanklyn still did not accept the germ theory of disease as late as 1906; it was too much a part of Frankland's system.[42]
In the long run Frankland prevailed. He, not Wanklyn, was internationally acknowledged as the greatest expert on water quality. Many analysts continued to use the ammonia process, but in the early 1880s a few, notably Charles Meymott Tidy (who would replace Wanklyn as Frankland's main adversary during the decade) did adopt combustion, if only to put themselves in a better position to attack Frankland's conclusions about water quality.