Eighteen—
Swim Speed and Dive Function in a Female Northern Elephant Seal
Daniel E. Crocker, Burney J. Le Boeuf, Yasuhiko Naito, Tomohiro Asaga, and Daniel P. Costa
ABSTRACT. The objective of this chapter is to discuss the behavioral and metabolic implications of swim speed data recorded from a northern elephant seal during free-ranging diving. Variation in swim speed elucidates the observed diving pattern and aids in understanding foraging tactics.
The data were obtained from a swim speed-distance meter and a time-depth recorder attached to the shoulders of an 8-year-old female at Año Nuevo, California, in February 1990. Swim speed, distance, depth, and duration of dives were recorded continuously for 29 days.
Average swim speeds ranged from 0.91 to 1.66 m/sec for all dive segments save one. These numbers are similar to those obtained from diving otariids and fall within the predicted cruising speed range for an aquatic animal less than 3 m in length. Horizontal swim speed during transit is similar to indirect measurements of transit velocity made by other investigators. Based on swim speed and distance traveled for each of five dive types apparent in the record, the two-dimensional shape of the various dive types observed, and the temporal patterning of dive types, we hypothesize that the four most common dive types observed serve three general functions: transit, foraging (pelagic and benthic), and internal physiological processes.
The advent of the use of time-depth recorder (TDR) technology has opened up a new level of behavioral and physiological analysis of diving in marine mammals (Kooyman 1965, 1968; Kooyman, Billups, and Farwell 1983; Guppy et al. 1986; Hill et al. 1987; Le Boeuf et al. 1989). Changes in depth, however, represent only one dimension of the behavior exhibited by an animal as it moves through the water column. Crucial to the understanding and expansion of the ideas arising from the TDR data is the development and deployment of an instrument that measures swim velocity. Historically, these data have been hard to acquire. Recent development and deployment of a number of different velocity instruments have allowed researchers to
begin to incorporate this measurement into their analyses of diving behavior and physiology (Ponganis et al. 1990, 1992; Le Boeuf et al. 1992).
Long duration divers like elephant seals must reconcile the conflicting metabolic demands of responses to hypoxia and exercise. The physiological adaptations with which the animal defends against hypoxia must be balanced against the rate of oxygen utilization required for active swimming. Much of what is known about the relationship between nondiving swimming velocity and metabolism comes from exercise studies in harbor seals, Phoca vitulina (Davis, Williams, and Kooyman 1985; Williams, Kooyman, and Croll 1990), gray seals, Halichoerus grypus (Fedak 1986), and California sea lions, Zalophus californianus (Feldkamp 1987; Williams, Kooyman, and Croll 1990). The sea lion study showed that oxygen consumption increased as an exponential function of swimming velocity. In contrast, investigations on freely diving Weddell seals, Leptonychotes weddelli (Kooyman et al. 1973), showed that reductions in metabolic rate together with extremely efficient swimming hydrodynamics allow the seal to swim while diving at only slightly higher than resting energy costs.
Swim velocity measured in a freely diving elephant seal offers an important clue to understanding the balance achieved between the opposing demands of exercise and apnea. The rarity of extended recovery periods in northern elephant seals, Mirounga angustirostris , and its implication of aerobic metabolism further underscores the importance of understanding this balance. Variations in swim velocity can offer significant insights toward understanding foraging tactics as well as other behaviors exhibited during diving.
Through simultaneous deployment of velocity and TDR instruments, the animal's swim velocity can be combined with the vertical component of its movements derived from the TDR data to map dives in two dimensions. This allows calculation of angles of descent and ascent as well as the horizontal component of the animal's movement. The shapes derived from this two-dimensional analysis and the energetic and behavioral implications derived from the velocity data allow us to begin to interpret the functions of the dive types classified as a result of the TDR data.
In this chapter, we discuss the behavioral and metabolic implications of swim speeds and the diving record obtained from an 8-year-old postpartum female northern elephant seal in 1990. We summarize the data from B. J. Le Boeuf et al. (1992) and offer further discussion of the results.
Measuring Swim Speed
The swim velocity instrument or, more accurately, the swim speed-distance meter (SSDM) measured distance traveled by the seal as a function of time
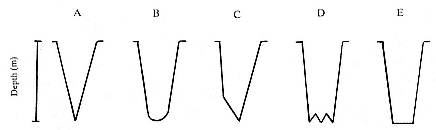
Fig. 18.1
Schematic representations of five dive types that appear in northern elephant seal
diving records. The dive types depicted are idealized and not adjusted for depth
or time. (Adapted from Le Boeuf et al. 1992.)
from which swim speed was calculated. The SSDM and TDR used in this analysis are described in detail in Le Boeuf et al. (1992).
Dive Type Classification
From the TDR data, each dive was classified into one of five categories and a category of unclassifiable dives from shape on a strip chart representation of the time-depth profile (fig. 18.1). Most unclassifiable dives appeared to be composites of two of the five categories. Dive types were classified as follows: Type A dives, characterized by direct descent to a sharp inflection point, followed by direct ascent to the surface; Type B dives, characterized by direct descent ending in a rounded bottom, followed by direct ascent to the surface; Type C dives, characterized by direct descent to depth, at which point descent rate decreases noticeably but proceeds directly to the bottom of the dive, followed by direct ascent to the surface; Type D dives, characterized by direct descent to a depth, followed by 2 to 12 vertical excursions, followed by direct ascent to the surface; and Type E dives, characterized by direct descent to a flat bottom followed by direct ascent to the surface.
Swim Speed and Distance Calculations
Swim speeds were calculated for dive segments and paired with the TDR data to draw two-dimensional vector triangles, the missing dimension being the spatial orientation of the animal at any time. The horizontal distances calculated in this analysis do not equate to linear swimming distance as the animal could have been turning during the course of a dive. These distances calculated are simply the horizontal component of the animal's movement during the course of a dive segment or dive. As the maximum calibrated speed measured by the instrument was exceeded on some dives,
all speeds and distances calculated should be considered minimum estimates. Similarly, angles of ascent and descent calculated are maxima. For a detailed explanation of the methods and potential sources of error, see Le Boeuf et al. (1992).
Swim Speed Measurements
Excluding the second descent segment of Type C dives, mean swim speeds for dive segments ranged from 0.91 to 1.66 m/sec. The mean velocity for all segments of all dives was 1.24 ± 0.21 m/sec.
On average, mean descent speeds were 27% faster than ascent speeds. This difference was statistically significant for all dive types. Descent speeds were similar across all dive types except for the second descent segment of Type C dives, which exhibited a significant reduction in swim speed. The second descent segment of Type C dives averaged 0.59 m/sec. in swim speed. Velocity on descent averaged 1.5 m/sec., while ascent and bottom velocity averaged 1.0 m/sec. No swimming was recorded at the surface above the stall speed of the instrument (0.4 m/sec.). Energetic concerns might preclude swimming above this speed due to increased drag at the surface. No swim speeds below 0.4 m/sec. were recorded during diving.
Comparison of Swim Speeds
The swim speeds recorded by the SSDM fall predominantly within the range of 1 to 2 m/sec., suggested by G. L. Kooyman (1989) as the cruising swimming velocity for animals up to 3 m in length. Recent investigations of swimming velocities in otariids (Ponganis et al. 1990) found a similar range for four species (0.9–1.9 m/sec.). It might seem that the larger elephant seal should swim faster than the smaller otariids since drag increases with the surface area or L2 , while the power increases directly with muscle volume or L3 , where L = length. Y. U. G. Aleyev (1977) suggests that swim speed varies directly with the frequency of the propulsive movement, which decreases with increasing muscle length, but that the drag/power relationship holds true for animals with a length less than 4.5 m. This would include our experimental animal.
The shorter dive duration, trip duration, energetically costly surface swimming, and different foraging strategies seen in the otariids suggest that a different compromise must be reached between the metabolic requirements of exercise and apnea and may explain the similar swim velocities seen in the long duration diving elephant seals. Swimming at MCT velocities allows an animal to cover a given distance with the minimum total oxygen consumption. If, however, the animal's emphasis is on achieving a lower rate of oxygen consumption and thereby a longer duration dive,
swimming at lower than MCT velocities might occur. A nonactive swimming or drift component to diving would contribute to this effect.
This idea is further supported by an energetic and physiological comparison of otariids and phocids. Studies have found that four species of lactating otariids expend energy at sea at five times the predicted basal level (Costa and Gentry 1986; Costa, Croxall, and Duck 1989; Costa, Thorson, and Kretzmann 1989; Costa, Antonellis, and DeLong 1990). Metabolic rates of diving elephant seals were estimated from dive durations and available oxygen stores to be only 1.3 times the predicted basal rate. Phocids have increased blood oxygen storage capacity due to a higher hematocrit (Lenfant, Johansen, and Torrance 1970). This increases blood viscosity and reduces optimal oxygen transport (Hedrick, Duffield, and Cornell 1986). Otariids, in contrast, have lower hematocrits that fall within a more optimal range for oxygen transport (Hedrick and Duffield 1991).
Comparison with Other Elephant Seal Investigations
A number of investigators in this volume present average daily transit velocities that allow comparison with the speeds measured by this instrument. We predict that these transit velocities would be somewhat lower than our recorded swim speeds as they ignore the vertical component of the animal's movement and include surface intervals for which our data indicate a lack of swimming. We can derive a comparable measure by dividing the calculated horizontal distance covered for each presumed transit dive by the dive duration. This yields a mean horizontal transit velocity of 1.00 ± 0.46 m/sec.
This speed is similar to the values reported for adult northern elephant seals, 0.89 to 1.03 m/sec. (Le Boeuf, this volume) and 1.04 to 1.15 m/sec. (Stewart and DeLong, this volume), and southern elephant seal females, 0.55 to 1.03 m/sec. (Fedak et al., this volume). D. J. Slip, M. A. Hindell, and H. R. Burton (this volume) present a range of transit speeds for southern elephant seals from 80 to 240 km/day or 0.9 to 2.8 m/sec. While the lower part of this range is similar to our measurements, a more complete accounting of which individuals exhibited higher transit speeds is required for meaningful comparison.
If we assume similar geometries to those obtained in this study, a transit speed of 2.8 m/sec. seems somewhat high. Using angles of ascent and descent derived from this study and assuming a nonswimming, surface interval of 1.5 minutes and an average dive duration of 20 minutes, a horizontal transit speed of 2.8 m/sec. yields a sustained swimming velocity of 3.4 m/sec. for the transit period. This is clearly beyond the capabilities exhibited by our female.
One possible explanation is that elephant seals might sometimes adopt a different mode of swimming that maximizes transit velocity at the expense of the total dive duration. This could represent flexibility in diving behavior that might be essential in a high predation environment where the depth of dives might be bathymetrically constrained. This alteration in swimming mode would be evident in the relationship between transit velocity and dive duration. A number of new records obtained from southern elephant seals show a dramatic shift from high transit velocity, short duration dives over the continental shelf to the more typical diving pattern as the animals reach deep water (C. Campagna, B. J. Le Boeuf, unpubl. data).
Recent deployment of velocity instruments on translocated yearling elephant seals has measured swim velocities similar to those presented in this study (R. Andrews, pers. comm.; P. Thorson, pers. comm.).
Speed Changes during Dive Segments
Since the sampling regimen of the SSDM was linear with respect to distance, not time, we were unable to assess instantaneous velocity or acceleration. However, numerous instances of what we termed "burst diving," rapid decreases and increases in speed within dive segments, were observed in the data. This level of detail was lost in the average speeds calculated for dive segments. To quantify this aspect of diving behavior, we looked at the variance among speed values within segments of dives. Burst diving occurred predominantly during descent and most often at the bottom of Type D dives. This behavior was significantly less frequent on the ascent segments of all dive types. Changes in swim speed occurred most infrequently during the second descent segment of Type C dives.
Angles of Ascent and Descent and Distance Traveled
Figure 18.2 shows the average two-dimensional plots for each dive type. Angles of descent were on average shallower (30° to 56°) than angles of ascent (52° to 82°). Angles of descent and ascent could not be calculated for Type B dives, due to their rounded bottom. However, the great similarity between Type B and Type A dives in every other aspect of this analysis argued for combining them as Type AB dives for subsequent analysis.
Functions of Dive Types
The data suggest that the different dive types serve three main functions. We hypothesize that Type AB dives may serve as transit dives. The average two-dimensional shape of Type A and B dives is notable for the great amount of horizontal distance covered: the seal averaged 1.2 km per dive.
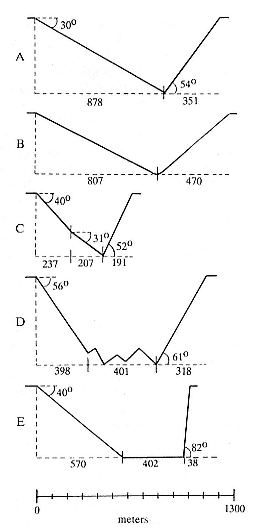
Fig. 18.2
Two-dimensional shapes for each of the five
classified dive types, showing mean angles
of ascent and descent and mean distance
covered for segments of dives.
(Adapted from Le Boeuf et al. 1992.)
Similarly, Type A and B dives had the shallowest angles of ascent and descent. Moreover, there is the strong degree of asymmetry seen in these dives. Horizontal distance covered was approximately three times greater on descent than ascent.
Returning to the central question of oxygen utilization and the balance between swimming effort and responses to hypoxia, we propose that this
asymmetry represents a more energetically efficient way of covering horizontal distance. If the animal is negatively buoyant, it could drift down or swim aided by gravity and then swim actively to the surface. On an average Type AB dive, this would enable the animal to cover 1.2 km of horizontal distance, while actively swimming or swimming unaided by buoyancy for only half of that distance. The energy savings would have to be substantial, as actual distance covered is increased. However as metabolic expenditure is directly related to swimming effort, this is possible.
Type AB dives were interspersed throughout the record but occurred in greatest numbers in long uninterrupted series during the first 19 days at sea. The strong asymmetry in shape, great horizontal distance covered, and temporal patterning of Type AB dives are all consistent with their hypothesized function as transit dives.
The data suggest that Type C dives may be "processing" dives, where behavior and energy utilization are focused on internal processes such as digestion and clearing of metabolites rather than external processes. Type C dives showed the shortest horizontal distance covered. The second descent segment of Type C dives averaged about a third of the speed of other segments of all dive types. This reduction in swim speed tended to occur at the same depth within a series of Type C dives. On average, this slow swimming period accounted for almost half (46.9%) of a Type C dive's duration.
It has been hypothesized that diving mammals are trying to maximize their time underwater while foraging (Kooyman 1981; Fedak, Pullen, and Kanwisher 1988; Kramer 1988). While this idea is not necessarily applicable to other types of dives, we argue that the pattern of C dives exhibited by northern elephant seals suggests its application to Type C dives. These dives occur in distinct bouts, with little variance in the inflection depth or second segment descent rate. It is unreasonable that the seal would return to the surface with remaining oxygen reserves and then spend potential foraging time and energy to return to depth and repeat the same behavior. If this is true—and whatever happens metabolically during diving is a constant across dive types—a dive type with a significantly lower swim speed would be expected to have a significantly longer duration. However, the mean duration of Type C dives (21.4 min.) is not significantly different from that of Type AB dives (21.6 min.), where the animal swims faster and covers on average twice the horizontal distance. Analysis of TDR records from 23 elephant seals including yearlings, adult females, and adult males show that Type C dive durations are not significantly different from Type A dive durations (Paired t-test, t = –0.05, p > .05; B. Le Boeuf, P. Morris, and D. Crocker, unpubl. data). From this, we infer that something different is happening metabolically during Type C dives.
Oxygen savings from reduced swim speed might be used in the process-
ing of food or metabolites incurred during diving. Oxygen savings could be used in perfusion of organs that incurred restricted blood flow on other types of dives. Type C dives did not appear in the record until day 17. This coincides with a relative decrease in the frequency of Type AB dives and a concomitant increase in the frequency of Type D dives.
Feeding studies on northern elephant seals have shown increases in metabolic rate averaging 65% 4 to 5 hours after feeding (P. Thorson, pers. comm.). If foraging occurs in long bouts, the increase in energy expenditure due to digestion might negatively impact the balance between the increased oxygen utilization required for active foraging and the hypoxia of diving. However, if the metabolic changes associated with food processing could be coupled temporally with decreased oxygen requirements due to decreased swimming effort, this impact could be significantly lessened.
A subsequent paper will test a number of predictions based on this idea in an attempt to demonstrate a more conclusive pattern of evidence supporting this hypothesis. The real test, however, will be to attempt to illicit this dive type in the homing paradigm (see Le Boeuf, this volume) and to directly measure perfusion of the digestive tract and swimming muscles. The metabolic issue raised here, the overall significantly lower swim speed, and the temporal patterning of this dive type in the record, are consistent with Type C dives serving a physiological process.
Type D and Type E dives appear to have characteristics consistent with foraging. The vertical excursions seen at the bottom of Type D dives are consistent with searching, pursuing, and capturing prey. Type D dives occurred in long series and were the most common dive type. Both of these characteristics are consistent with foraging. Type E dives might serve feeding on the bottom, or benthic foraging. These dives also occurred in long series. The depths of the bottom segments within a series of Type E dives were relatively invariant. Though the depth sometimes changed slightly during the course of a bottom segment, the succeeding bottoms in a series appeared to start close to the depth at which the preceding bottom ended. This gives the impression that the animal's movement was following the bottom topography, though geolocation and bottom depths are needed to be conclusive. Moreover, the descent and ascent angles were abrupt in both of these dive types, suggesting that the animal descended directly to a depth where prey were found, spent one-third of the dive's duration foraging, then returned directly to the surface. The increased incidence of speed changes seen in both dive types, especially during the bottom segment of Type D dives, is also consistent with foraging.
A recent investigation of sex differences in foraging patterns in northern elephant seals (Le Boeuf et al. 1993) offers strong support for this characterization of Type D and E dives. Diurnal variation in depth for Type D dives was found which is consistent with movements of the deep scattering layer
and was not evident in any other dive type. This dive type might account for most of the diurnal variation in dive depth and duration reported by many investigators.
A strong association of Type E dives with coastal regions was also demonstrated. However, the geolocation of many of these dives might be sufficiently far away from the continental shelf to preclude benthic foraging. Stewart and DeLong (this volume) also argue that the geolocation of their Type E dives precludes benthic foraging. However, a recent shipboard validation study of a geolocation TDR revealed a strong, consistent, southernly bias to geolocation estimates in the area in question (S. Blackwell and B. J. Le Boeuf, unpubl. data). This bias might preclude the use of light-level geolocation to obtain precise correlations between diving behavior and bathymetric features.
This analysis represents a first effort at using velocity and TDR instruments together to investigate questions concerning diving behavior. New and better instruments that allow assessment of instantaneous velocity and acceleration will allow a much finer level of analysis than that presented here. Future generations of instruments will also have higher upper ranges so that a more accurate picture will emerge. However, the relationships between the numbers in this study allowed us to formulate hypotheses that will form the basis of future investigations.
References
Aleyev, Y. U. G. 1977. Nekton . The Hague: W. Junk.
Briggs, G. D., R. V. Henrickson, and B. J. Le Boeuf. 1975. Ketamine immobilization of northern elephant seals. Journal of the American Veterinary Medicine Association 167: 546–548.
Costa, D. P., G. P. Antonelis, and R. DeLong. 1990. Effects of El Niño on the foraging energetics of the California sea lion. In Effects of El Niño on Pinnipeds , ed. F. Trillmich and K. Ono, 156–165. Berlin: Springer Verlag.
Costa, D. P., J. P. Croxall, and C. Duck. 1989. Foraging energetics of Antartic fur seals, Arctocephalus gazella , in relation to changes in prey availability. Ecology 70: 596–606.
Costa, D. P., and R. L. Gentry. 1986. Reproductive energetics of the northern fur seal. In Fur Seals: Maternal Strategies on Land and at Sea , ed. R. L. Gentry and G. L. Kooyman, 79–101. Princeton: Princeton University Press.
Costa, D. P., P. H. Thorson, and M. Kretzmann. 1989. Diving and foraging energetics of the Australian sea lion, Neophoca cinera. American Zoology 29: 71A.
Davis, R. W., T. M. Williams, and G. L. Kooyman. 1985. Swimming metabolism of yearling and adult harbor seals, Phoca vitulina. Physiological Zoology 58: 590–596.
Fedak, M. A. 1986. Diving and exercise in seals: A benthic perspective. In Diving in Animals and Man , ed. A. Brubakk, J. W. Kanwisher, and G. Sundnes, 11–32. Kongsvold Symposium, Royal Norwegian Society of Sciences and Letters, June 3–7, 1985. Trondheim: Tapir.
Fedak, M. A., M. R. Pullen, and J. Kanwisher. 1988. Circulatory responses of seals to periodic breathing: Heart rate and breathing during exercise and diving in the laboratory and open sea. Canadian Journal of Zoology 66: 63–69.
Feldkamp, S. D. 1987. Swimming in the California sea lion: Morphometrics, drag, and energetics. Journal of Experimental Biology 131: 117–136.
Guppy, M., R. D. Hill, R. C. Schneider, J. Qvist, G. C. Liggins, W. M. Zapol, and P. W. Hochachka. 1986. Micro-computer-assisted metabolic studies of voluntary diving of Weddell seals. American Journal of Physiology 250: R175–R187.
Hedrick, M. S., and D. A. Duffield. 1991. Haematological and rheological characteristics of blood in seven marine mammal species: Physiological implications for diving behavior. Journal of Zoology, London 225: 273–283.
Hedrick, M. S., D. A. Duffield, and L. H. Cornell. 1986. Blood viscosity and optimal hematocrit in a deep-diving mammal, the northern elephant seal (Mirounga angustirostris ). Canadian Journal of Zoology 64: 2081–2085.
Hill, R. D., R. C. Schneider, G. C. Liggins, A. H. Schuette, R. L. Elliot, M. Guppy, P. W. Hochachka, J. Qvist, K. J. Falke, and W. Zapol. 1987. Heart rate and body temperature during free diving of Weddell seals. American Journal of Physiology 253: R344–R351.
Kooyman, G. L. 1965. Techniques used in measuring diving capacities of Weddell seals. Polar Record 12: 391–394.
———. 1968. An analysis of some behavioral and physiological characteristics related to diving in the Weddell seal. In Antarctic Research Series , vol. 11, Biology of the Antarctic Seas III , ed. W. L. Schmitt and G. A. Llano, 227–261. Washington, D.C.: American Geophysical Union.
———. 1981. Weddell Seal: Consummate Diver . Cambridge: Cambridge University Press.
———. 1989. Diverse Divers: Physiology and Behavior . Berlin: Springer Verlag.
Kooyman, G. L., J. O. Billups, and W. D. Farwell. 1983. Two recently developed recorders for monitoring diving activity of marine birds and mammals. In Experimental Biology at Sea , ed. A. G. Macdonald and I. G. Priede, 197–214. London: Academic Press.
Kooyman, G. L., R. L. Gentry, and D. L. Urquhart. 1976. Northern fur seal diving behavior: A new approach to its study. Science 193: 411–412.
Kooyman, G. L., D. H. Kerem, W. B. Campbell, and J. J. Wright. 1973. Pulmonary gas exchange in freely diving Weddell seals (Leptonychotes weddelli ). Respiration Physiology 17: 283–290.
Kramer, D. L. 1988. The behavioral ecology of air breathing by aquatic animals. Canadian Journal of Zoology 66: 89–94.
Le Boeuf, B. J., D. E. Crocker, S. A. Blackwell, P. A. Morris, and P. Thorson. 1993. Sex differences in foraging in northern elephant seals. In Marine Mammals: Advances in Behavioural and Population Biology , ed. I. L. Boyd, 149–178. Symposia of the Zoological Society of London no. 66. London: Oxford University Press.
Le Boeuf, B. J., Y. Naito, T. Asaga, D. E. Crocker, and D. P. Costa. 1992. Swim speed in a female elephant seal: Metabolic and foraging implications. Canadian Journal of Zoology 70: 786–795.
Le Boeuf, B. J., Y. Naito, A. C. Huntley, and T. Asaga. 1989. Prolonged, con-
tinuous, deep diving in female northern elephant seals, Mirounga angustirostris. Canadian Journal of Zoology 67: 2514–2519.
Lenfant, C., K. Johansen, and J. D. Torrance. 1970. Gas transport and oxygen storage capacity in some pinnipeds and the sea otter. Respiration Physiology 9: 277–286.
Naito, Y., B. J. Le Boeuf, T. Asaga, and A. C. Huntley. 1989. Long-term diving records of an adult female northern elephant seal. Antarctic Record 33: 1–9.
Ponganis, P. J., R. L. Gentry, E. P. Ponganis, and K. V. Ponganis. 1992. Analysis of swim velocities during deep and shallow dives of two northern fur seals, Callorhinus ursinus. Marine Mammal Science 8: 69–75.
Ponganis, P. J., E. P. Ponganis, K. V. Ponganis, G. L. Kooyman, R. L. Gentry, and F. Trillmich. 1990. Swimming velocities in otariids. Canadian Journal of Zoology 68: 2105–2112.
Williams, T. M., G. L. Kooyman, and D. A. Croll. 1990. The effect of submergence on heart rate and oxygen consumption of swimming seals and sea lions. Comparative Physiology 160B: 637–644.