4—
Gerhardt and Wurtz
Liebig and Dumas
As Hermann Kolbe's relationships with the principal French chemists during his early career were influential on his ideas and actions, both in these years and later, we must spend some time with them.[1] A summary of the early history of the theories of substitution, nuclei, and types has been provided at the end of chapter 2. The leading personalities in this story were Liebig and Dumas, who first met as young men in Paris in 1823 and who spent the next two decades jousting with each other in developing overarching concepts in organic chemistry.
It was perhaps inevitable that these men would become rivals. Both were demon workers with extraordinarily creative minds, cultivating a field that had too many mysteries and too few facts. Both had occasion to accuse the other, sometimes justly, of experimental work that was "geschwind abet schlecht." Both had occasion to accuse the other, probably also sometimes justly, of poaching results. As violent as their disputes at times became, by 1840 they found themselves not very far apart—though neither man was then willing to admit this to the other.
In his worst moments, Liebig thought of Dumas as a true charlatan or "Schwindler" who was not above using questionable tactics or sleight of hand to achieve renown and whose greatest concern was pursuit of effect, flourish, and the rhetorical turn of phrase, all for the sake of personal ambition. For his part, Dumas often viewed Liebig as a heavy-handed and hotheaded chemical empire builder. After a brief alliance at the end of 1837 and the beginning of 1838, Liebig became
dissatisfied with the pact he had made with Dumas. In 1840 he made a "total break" from the Frenchman, the quarrel resulting from substitution theories and based upon some real issues along with some pure misunderstandings. Dumas was a "tightrope dancer," a "Jesuit," a "highwayman," and a "thief," like "nearly all Frenchmen."[2] To Berzelius, Liebig complained,
These Frenchmen truly have no feeling of true honor, no sense of justice and fairness, they have for many years been occupying themselves with theoretical speculations that are useless for science, and solely to satisfy their own vanity and arrogance; they have discovered that the word Radical must be banned and must be substituted by the word Type. This is the greatest of their discoveries. Unfortunately when I step forward there is in Germany only envy and weakness, so I stand completely alone, no one who has enough power to stand up to them supports me. In short, it is a bad time and I am very unhappy, and have turned from these miserable matters to applications of chemistry to physiology, which now interest me tremendously.[3]
Unfortunately, here again Liebig collided with Dumas, as Liebig became convinced in 1842 that Dumas had stolen his original ideas on plant and animal nutrition, and the heat of discord only became more intense.[4]
Even in the midst of some of these disputes, however, Liebig was able to recognize Dumas' merits and to concede when he had been in the wrong, and when the violence of his replies sometimes had done nothing but damage.[5] On 23 April 1850, Liebig wrote his friend C. F. Kuhlmann in Lille, whom he was about to visit to help dedicate a new factory. He was very much looking forward to seeing Dumas there, as he was anxious to renew their old friendship,
. . . since I have always very highly esteemed Herr Dumas as one of the most outstanding and ingenious men among the chemists and scientists of our day. Perhaps more than any other chemist in Europe I found myself in the position of judging and prizing the value of his work, since we very frequently encountered each other in our investigations, and have cultivated the same fields.[6]
Liebig's hopes for the encounter were realized, as he wrote to Wöhler:
We all arrived at the same time, embraced each other, and everything was fine. Dumas was extremely cordial, and looked so young that I hardly recognized him. His wife and daughter were with him, to serve as witnesses to the plans for revenge that he had brewed. On Whitsunday the celebration was splendid and merry, the next evening a banquet, to
which the civil and military leaders of Lille were invited. At the end of the banquet Dumas stood up, gave a long speech, flattered me with various puffery, and finally took a decoration for the legion d'honneur from his pocket, and handed it to me along with the brevet in the name of the President of the French Republic. I was; unprepared and thought I would faint; but I managed a speech and received an accolade. Thus he revenged himself on me. Despite all he has a magnificent nature.
The following year Liebig dedicated a new edition of his Chemische Briefe to Dumas, and the two exchanged a number of warm letters until Liebig's death in 1873.[7]
It will be noted that during the entire period over which we have followed Kolbe's early career in chapters 2 and 3, Dumas and Liebig were feuding intermittently. In fact, the interval between Kolbe's first published paper and his long review article on radicals and types precisely brackets the period when Liebig's and Dumas' relationship was at its lowest ebb. Kolbe certainly imbibed an extremely negative view of Dumas from Liebig, who was one of his idols and models and whose diatribes were often openly published in the scientific literature. Berzelius, and his former student Wöhler, the author of the Schwindler satire, also had opinions of Dumas and other French chemists which were not much more positive than Liebig's. But Dumas had retreated about 1840 from a leading theoretical role, replaced by such chemists as Laurent and Gerhardt and in the 1850s by Wurtz as well. Kolbe's relationships with Gerhardt and Wurtz paralleled Liebig's relationship with Dumas, except for the lack of a final reconciliation. It was with Gerhardt and Wurtz that Kolbe felt the strongest sense of rivalry, enmity, hatred—and occasionally, even affinity, if not regard.
Gerhardt
Charles Gerhardt (1816-1856) was born in Strasbourg to a bourgeois Jewish family with both German and Alsatian roots. With the prospect of helping his father make a success of a white lead factory in which he had a major financial interest, Gerhardt was sent to the Karlsruhe Technische Hochschule in 1831, then two years later to the Leipzig Gewerbeschule. Here he: lodged and studied with O. L. Erdmann and published his first scientific paper in Erdmann's (and later Kolbe's) Journal für praktische Chemie . During winter semester 1837/38, he studied in Liebig's lab in Giessen, thereby acquiring an influential protector. On Liebig's advice, he then traveled to Paris where he was associated with Dumas and H. Sainte-Claire Deville at
the Faculté des Sciences and the Sorbonne and with A. Cahours at the Jardin des Plantes.
Liebig, who just then was becoming disillusioned with polemics over theory, sought to discourage his protégé's predilections in that direction:
You will destroy your future and irritate everyone, like Laurent and Persoz, if you continue to make theories. . . . The Academy [of Sciences in Paris] has always reserved for itself the right of making laws in science, and it considers anyone else doing this as a thief and an assassin. . . . You always think you are on the neutral soil of Germany, but in fact you are standing on ground which contains all sorts of combustible matter.
Liebig urged Gerhardt to tie himself personally to Dumas, who has "a magnificent character" and likes to take promising young chemists under his wing. "But for the love of God, don't write any more theories except for German journals!"[8]
Even as a young man, Gerhardt was headstrong, outspoken, and dogmatic, and his relationship with Dumas was from the beginning somewhat uneven. His ardent republican political convictions did not mesh with Dumas' moderate conservatism, and his ties to Liebig created friction in a period when Dumas' relations with the Giessen school were starting to unravel. Moreover, Liebig was correct in perceiving a fundamentally positivistic flavor among the leaders of Parisian chemistry, which conflicted with Gerhardt's visceral theoretical orientation. In 1841, Gerhardt received a provisional appointment at Montpellier, which was made permanent in 1844. In a letter of gratitude to Liebig, Gerhardt attributed both this career triumph and his general success as a chemist to his teacher's influence. But no sooner had Gerhardt arrived in Montpellier than he began to complain bitterly to Liebig of provincial life and the poorly provisioned laboratory facilities.[9]
In 1842 Gerhardt published one of his greatest papers and simultaneously committed one of his greatest faux pas. He had come to the conclusion that organic chemists needed to unite their formula notation with inorganic conventions by taking all formulas at two volumes instead of the usual four. This would eliminate many dualistic formulas that posited the preexistence of water, ammonia, oxides, and so on in organic acids, bases, and salts, but since the advent of type theories such electrochemical rational formulas were no longer popular in France anyway. In the context of French chemistry of the early 1840s, a very good case could have been made in favor of such ideas. They
were later viewed as constituting the essential step away from a misleading and inconsistent notation to untitled, consistent, and essentially modern formulas. Although Gerhardt did not say so and may not in fact have realized it, his reform proposal was equivalent to accepting the hypotheses of Avogadro.[10]
Unfortunately, Gerhardt committed two serious errors in this paper. One mistake was a certain degree of conceptual and linguistic confusion. Even when a historian reads this paper with full knowledge of both contemporary and subsequent events in mind, it is sometimes difficult to extract Gerhardt's precise meaning. There were a few egregious lapses: one example is Gerhardt's claim that atoms, volumes, and equivalents are exact synonyms. A second paper on the same subject published the following year solved a few of these problems, but not the most serious ones.[11]
Even worse for the future of his career, Gerhardt expressed himself in socially inappropriate terms. When his paper was read at the Acadé-mie on 5 September 1842, some members of the audience were shocked to hear what seemed to be arrogant and imperious language from this young man, and they let him know in no uncertain terms. Dumas had wanted to suppress the theoretical part. V. Regnault was furious at some of his formulas being declared "wrong," and Baron L. J. Thenard said the style would not have been appropriate even for a Lavoisier. Later that month back in Montpellier, Gerhardt recounted the disaster in a letter to his friend Cahours. He said he had asked Thenard timidly later that day why he didn't like the paper. "For a moment I thought he was going to eat me," Gerhardt related. He and others agreed, Thenard had said, that the language was "not French" and "not academic" and that the style was infuriatingly imperious (such-and-such a formula is "false," the notation "must be changed," and so on). Thenard concluded by screaming "Adieu, monsieur!" at least ten times before Gerhardt got the hint and excused himself. Liebig received the same news from Gerhardt and replied, "You have Italian blood, too hot for the Parisians." Liebig noted that Gerhardt's fatal error was not to have heeded his warning about avoiding theory in Paris.[12]
In the fall of 1843 Gerhardt met Laurent, and they quickly became friends and comrades in arms. Gerhardt's judgment of Laurent in his letters to Cahours underwent a reversal from derogation to adulation. Laurent, like Gerhardt, was an uncompromising iconoclast and an ardent republican, cut off from the power structure in Paris and a professor at a provincial faculty, namely, Bordeaux; he may also have been Jewish.[13] Also like Gerhardt, Laurent was collegially closer to the Giessen circle than to Paris. He had visited Giessen for a time dur-
ing the autumn of 1844 and had quite favorably impressed both Liebig and his assistant August Wilhelm Hofmann (it was under Laurent's influence that Hofmann began to study the halogenation of aniline). His angry confrontations with Dumas over priority matters during the late 1830s, which had alienated his former teacher, could not but have stirred sympathy for him in Liebig's circle.
Laurent was soon converted to Gerhardt's atomic weight reform, and in December 1844 Laurent wrote to Liebig and Hofmann to urge them to accept it as well. Unfortunately, Liebig considered the reform to be nothing more than yet another French theory, and declined; Hofmann was presumably too cautious and too professionally insecure to associate himself with the reformers. "Eh! bien, marchons seuls," Laurent wrote Gerhardt in resignation.[14]
A more serious problem arose a year later when Gerhardt infuriated Liebig by criticizing as "completely false" his work on a series of complex nitrogen compounds. Liebig promptly published a blistering diatribe entitled "Herr Gerhardt und die organische Chemie, Erste Artikel," openly accusing Gerhardt of the vilest perfidy and calling him a "highwayman."[15] He also urged Laurent to break off his "monstrous alliance" with Gerhardt. "If you associate yourself with him," Liebig wrote, "it is you who will lose because he has nothing to lose. Read carefully my article, and tell me whether this man has the truth in his soul."[16] But Laurent refused to abandon his friend and wrote him a kind letter of support and consolation. Laurent related that on his recent visit to Giessen, Liebig had apologized to him for his earlier unjustified attacks and had vowed that his next angry polemic would rest six months in his desk first! He assured Gerhardt not only that Liebig would cool off, but that he would eventually realize that the new atomic weights are correct.[17]
Gerhardt had criticized Liebig's formulas largely on the basis of the "even-number rule" developed by Laurent from certain regularities in the numbers of hydrogen atoms in organic formulas noted by Gerhardt in his 1842 article. The rule as generalized by Laurent (1845) states that although there may be any number of carbon and oxygen atoms in organic formulas when expressed in Gerhardt's two-volume convention, the sum of all the hydrogen, halogen, and nitrogen atoms must be an even number.[18] At the time of its formulation, this law was merely an empirical generalization unmotivated by theory, but it seemed to hold for every well-studied organic compound; the only exceptions were cases where there was some reason to suspect analytical inaccuracies, and a handful of contrary instances, such as Liebig's nitrogen compounds. It was little wonder that Liebig was unconvinced by this sort of reasoning.
By this time Laurent had given up his unhappy post in Bordeaux and was living in poverty in Paris. As he and Gerhardt were virtually cut off from the traditional French journals, they had started their own, entitled Comptes rendus des travaux de chimie . This was essentially a review journal modeled on Berzelius' Jahresberichte , as well as an organ for publication of their own papers.
It would seem that at least in one respect Gerhardt had learned his lesson, for after 1842 he pursued a much more openly, indeed often dogmatically, positivist course. Having long denied the preexistence of the dualists' water and oxides in acids and salts, he now denied the real existence of any and all radicals within compounds. In fact, he argued that it was beyond human capabilities to discern any information at all regarding molecular constitutions, and so he began to rely exclusively on empirical molecular formulas. In this he was resisted by his compatriot Laurent, who used constitutional theories habitually and continuously, though professing more a conventionalist than a realist philosophy of science.[19]
Perversely, this new Gerhardtian positivist-empiricist organic chemistry was rich in new doctrines and ideas, among which were the revised atomic weights and Laurent's even-number rule. The former was an early version of the reform established nearly two decades later at the Karlsruhe Congress of 1860; the latter represented the first seed of what would eventually grow into the theory of atomic valence. Gerhardt also began to develop the concept of homologous series, an idea first broached by Dumas, which would prove extremely productive and heuristically important. These ideas were treated systematically in Gerhardt's first book, Précis de chimie organique (Paris, 1844-1845). Although this work was not well received—not even by Laurent, who subjected it to a searching critique—and although Gerhardt himself described it later as a "horrible old book," it contains the basis of many of his contributions to organic chemistry.[20]
The way out of the morass was finally perceived by Laurent. In the summer of 1846 he wrote Gerhardt:
I have carefully pondered equivalents. The words equivalents, atoms and volumes cannot be synonymous. . . . There are atoms, there are proportional numbers, but not equivalents. . . . Indeed, the proportional number is a number chosen arbitrarily; one can take it to agree with the atom or volume and to give the simplest notation. The equivalent is different: it is that quantity of a simple body which must be employed to replace another simple body and play its role.[21]
These thoughts were published that fall in a major theoretical paper that fully clarified Gerhardt's terms arm straightened out the concep-
tual confusion in the 1842 reform proposal. "M. Gerhardt's atom," Laurent stated, "represents the smallest quantity of a simple body which can exist in a compound . My molecule represents the smallest quantity of a simple body which must be employed to produce a combination ; this quantity splits in two in the act of combination." For instance, an oxygen molecule splits into two oxygen atoms to form two molecules of water; a molecule of chlorine splits into two atoms, one of which substitutes for a hydrogen atom of an organic molecule, with the other combining with the hydrogen thus split off to form a molecule of hydrochloric acid.[22]
In hindsight, Laurent's modification of Gerhardt's reform appears to establish at a stroke the basis for modern chemistry. In fact, it did nothing of the kind. In the late 1840s the Laurent-Gerhardt system was only a schematic proposal with considerable esthetic appeal, but based essentially on thoughtful rationalization rather than empirical verification. Moreover, it was in conflict at many points with well-accepted chemical theory and was still troubled by a number of anomalies. Laurent himself recognized all of these weaknesses and urged Gerhardt not to lose courage.[23] The theoretical anarchy of this period is well summarized by Laurent's ironic summary description of a course he had been invited to teach at the Sorbonne in the summer of 1847:
Introduction . The entire science is placed in doubt. Atoms are perhaps divisible; dualism is attacked; nomenclature is insufficient; ditto for classifications; all compass bearings are lost, we need a guiding thread. Embarrassment of the professor who teaches a science in which he does not believe.[24]
Laurent was then suffering both extreme penury and depression. "What misery! What a bitch life is! Not a sous in my pocket!" he cried out to Gerhardt.[25]
The following year Gerhardt converted Laurent's sarcasm into a book entitled Introduction à l'étude de la chimie par le système unitaire , dedicated to Laurent. It is here where Gerhardt most clearly stated his conviction that molecules must be treated as unitary entities and must be described only by empirical formulas. Reactions of synthesis and analysis say nothing about the constitutions of compounds, for every chemical reaction sets up a violent motion among the atoms in the molecule that totally scrambles their arrangement. Organic chemical theories, he said, must therefore be based on taxonomic rather than constitutional ideas, on reactions rather than structures. In this he was representing a well-established French positivist tradition in chemistry that was fundamentally opposed to many German, English, and Berzelian conceptions.[26]
This book, reflecting the theoretical anarchy in chemistry, appeared the same month when political anarchy emerged in Paris, the opening wedge of the long sought republican revolution. Understandably, the fortunes of Laurent and Gerhardt suddenly appeared promising again. In March Laurent was named assayer at the national mint, where he fitted up a small laboratory; Gerhardt immediately abandoned his position in Montpellier to join him there. Arago and Quesneville, republicans, and Cahours, a liberal, greeted the revolution warmly; many who had more conservative or centrist convictions were frightened by it. Dumas appeared to change camps, writing a discourse strongly favoring the republic; the apparent hypocrisy scandalized those on the left.[27]
Gerhardt called on Dumas on 26 March, and described the encounter in a letter to his wife, still residing in Montpellier. Dumas received him coldly and was about to show him the door when Gerhardt suggested that Dumas' conduct "had been unworthy."
At once he became timid as a lamb. So he was afraid. I changed tactics and told him that he had been wrong to abdicate the good position that he had had in science, to turn to politics and administration; that he alone in France was able to understand my ideas (mine and Laurent's) . . . Then Dumas protested of his devotion to science, vigorously denied any hostility to me, and began to weep! Was he sincere at this moment? I have no idea, but I want to believe it. He shook my hand several times and we parted perfectly well. Our conversation had lasted nearly an hour and a half, and surely no one has ever dared to tell Dumas the truth as energetically as I. . . . I am sure now that he will do anything for me to prevent me from denouncing him.[28]
There were several vacancies in Paris, and Gerhardt and others mounted a vigorous attack on the system of cumul that allowed prominent academics such as Gay-Lussac and Dumas to accumulate almost any number of posts simultaneously. Laurent and Gerhardt both sought to influence the provisional regime's minister of culture Hippolyte Carnot (brother of Sadi) to procure appropriate positions, with promising results. Gerhardt reported to his wife that all of his former enemies were treating him very well now. "If there is justice in the world I will succeed this time; now or never."[29]
Unfortunately for Gerhardt, Carnot was gone by July, and by the following year the republican tide was waning; all of his hopes began to evaporate. In the spring of 1849 he described himself to his wife as being in a "veritable state of fever," reading ten or a dozen newspapers every day, but lying low for the sake of his family. During the June riots he wrote: "I must tell you, you are making a coward of me! If the
insurrection is vanquished this time, it will be the death of democracy in all of Europe." Vanquished it was.
Gerhardt's enemies, especially Dumas, emerged stronger than ever before. Even more tragic, from working in his cold and damp cellar laboratory at the mint—described by Gerhardt as a "veritable glacier"—Laurent contracted a severe case of tuberculosis in December 1850; he lingered on over two years before finally succumbing. His last days were bitter in the extreme.[30]
Now essentially unemployed and desperately in need of a protector, Gerhardt wrote Liebig in an attempt to patch over their quarrel:
You know, monsieur, that basically there is no important difference between our theoretical opinions. . . . One could still today define organic chemistry as the chemistry of compound radicals; it is only a question of making very precise the meaning of the word radical, and of removing from it that absolute signification which you yourself have never assumed, and which has been interred with Berzelius.[31]
Liebig's response, although friendly, was noncommittal. To Hofmann, Liebig expressed suspicions concerning Gerhardt's sincerity and motives. In February 1851, using money lent by Gerhardt's Scots mother-in-law, he and the infirm Laurent began to accept pupils in a private "École de Chimie Pratique," and both began to write definitive treatises of chemistry.[32]
This was the low point for the reform movement of Gerhardt and Laurent. Its revival was associated with remarkable breakthrough discoveries by Gerhardt and Alexander Williamson and by the work of Wurtz, to whom we now turn.
Wurtz
(Charles) Adolphe Wurtz was born on 26 November 1817, the eldest son of Jean Jacques Wurtz and the former Sophie Kreiss. The elder Wurtz, the only child of a "simple bourgeois family" of Strasbourg, became a Lutheran pastor in Wolfisheim, a small village three miles west of the city; Sophie's father, also named Jean Jacques (Johann Jakob), was also a Lutheran pastor, at the church of St. Pierre-le-jeune in Strasbourg. It is said that Adolphe's boyhood in Wolfisheim provided him with both a robust constitution and a love of the countryside. He spoke Alsatian with his family, but learned French and Hochdeutsch in his youth (he is said to have spoken both languages without a trace of regional accent).[33]
The parallels here to Hermann Kolbe's family background and up-
bringing are remarkable. Born less than a year apart, both died in 1884 at the age of sixty-six. Despite their different nationalities, both were raised in German-speaking households of the same social class; both were eldest sons of Lutheran country pastors who led congregations in small towns near important university cities. Both remained sincere Protestants and became outspoken nationalists in their later years.
In 1826 Wurtz' father was called to Strasbourg as third pastor at his father-in-law's church, and Adolphe entered the Strasbourg Protestant Gymnasium. His father wanted him to enter the ministry, but Adolphe had conceived a passion for science. As a sort of compromise, Adolphe was allowed to study medicine, entering the Strasbourg Faculty of Medicine in 1835. Between 1839, when he was named the faculty's Chef des travaux de chimie, and his departure for Paris in 1844, he worked mostly in the laboratory of Amedée Cailliot (1805-1884). He also spent five months (summer semester 1842) at Liebig's laboratory in Giessen, and he always considered Liebig as one of his mentors. In the spring of 1844, he went to Paris seeking a chemical career. He started with Balard at the Faculté des Sciences, then after a year was appointed Dumas' préparateur at the Faculté de Médecine. Simultaneously he accepted the post of Chef des travaux chimiques at the École Centrale des Arts et Manufactures, founded by Dumas sixteen years earlier. He soon became Dumas' favorite and ultimately was to inherit the mantle of the master.
Of course, fellow Strasbourger Charles Gerhardt was also a presence in Wurtz' early years. Most biographers note, correctly, that Gerhardt and Wurtz were schoolfellows at the Strasbourg Gymnasium and that Wurtz translated Gerhardt's first book, the Précis (1844-1845), into German. The conclusion has often been drawn that Wurtz came to Paris already in the Gerhardt-Laurent orbit, against the established ideas of such figures as Liebig and Dumas.[34]
The situation was quite the opposite. Although Gerhardt was only fifteen months older than Wurtz, he was two Gymnasium classes in advance and left a year earlier than was normal, when Wurtz was but thirteen years old; they appear not to have become acquainted then.[35] Moreover, Wurtz did not offer to translate Gerhardt's book from a position as disciple. Rather, on a trip home in 1842, Gerhardt inquired of Cailliot regarding an appropriate bilingual Alsatian chemist; Cailliot recommended Wurtz, who was happy to do the work both for the chemical interest and for the money involved.[36] Gerhardt was in Montpellier for the first four years after Wurtz arrived in Paris, and even after Gerhardt returned to Paris in 1848, Wurtz saw little of him. Wurtz scarcely knew Laurent personally.[37] During Gerhardt's lifetime, Wurtz never adopted the new chemical "equivalents" championed in the Pré-
cis . Finally, at the time Gerhardt began work on the Précis and hired Wurtz as translator (1842-1843), he was still on good terms with Liebig. He was rightly seen as a disciple of both Liebig and Dumas and had not yet joined forces with Laurent.
By agreeing to translate Gerhardt's Précis , Wurtz was not declaring his allegiance to Gerhardtian ideas, and those ideas had not yet been labeled as heretical. Rather, he was accepting an assignment that might well lead to professional advancement, as well as providing much needed income. By the time Wurtz finished his translation in 1846, open warfare had broken out between the Gerhardt-Laurent alliance and the uneasy Dumas-Liebig axis, but Wurtz could not have foreseen this situation four years earlier. In short, all the evidence suggests that in the 1840s and early 1850s, Wurtz was a loyal adherent of the established chemical theories of Liebig and especially of his patron Dumas.
Wurtz' early work illustrates many of these ideas. In Liebig's lab he investigated hypophosphorous acid, presenting the results in terms of Liebig's hydracid theory (and also citing Liebig's predecessors Davy and Dulong).[38] Wurtz' first papers after his association with Dumas exhibit a distaste for dualist ideas and a concern for merging organic and inorganic chemistry under unitary assumptions. They also contain references to Dumas' "edifice" analogy for molecular structure (despite conventional empiricist protestations). Wurtz even proposed PCl3 as the "type" of most phosphorus compounds—choosing a word that had been used in 1839 and 1840 by Liebig as well as Dumas.[39]
As we have seen, from about 1840 both Liebig and Dumas simultaneously and apparently independently retreated from the new directions they had been heading. Both began to pursue more practical and purportedly less theoretical subjects such as agricultural and physiological chemistry. Liebig turned away from his unitary hydracid formulations toward more dualistic oxide formulas. In 1844, Liebig shifted from four-volume Berzelian atomic weights, which he had been using for fourteen years, to Gmelin's four-volume conventional equivalents. In approximate modern terms, this was equivalent to moving from doubled molecular formulas to formulas with doubled numbers of even-valence atoms but correct numbers of odd-valence atoms. This shift was connected to the other changes since the new formulations were regarded as perfectly empirical, in contrast to Berzelius' theoretically derived atomic weights.
For several years, Wurtz mirrored the ambivalence of Liebig and Dumas. On the one hand, he continued to advocate substitutable radicals and the unification of organic and inorganic formulas. On the other hand, he followed Liebig's "empiricist" atomic weight shift of
1844 and continued to use many of the formula conventions attacked by Gerhardt and Laurent. For instance, his novel substance "ammoniacal cyanic ether" (modern ethyl isocyanate, C2 H5 NCO) was written C2AzO,C4 H5 O ,AzH3 , a formulation that reflects an implicit dualism as well as incompatibility with Gerhardt's new atomic weights and with his constitutional ideas.[40] This example is not an arbitrary one: alkaline hydrolysis of this substance led to Wurtz' first major discovery, ethyl-amine, in 1849. It was the first primary amine, indeed the first amine of any kind to be recognized, and the progenitor of an immense number of synthetic organic bases.[41]
Hofmann provided an interesting gloss on this event. Hofmann and Wurtz had become good friends during Wurtz' stay in Giessen in 1842, where Hofmann was assistant to Liebig. From 1847 they had the opportunity to see each other regularly, for Wurtz had frequent occasion to visit his sister and her husband in England, and Hofmann, who was professor at the Royal College of Chemistry in London from 1845, found himself often in Paris.[42] Moreover, they had a major chemical interest in common, namely, organic' bases. Hofmann related that Wurtz had missed the discovery of ethylamine for quite some time since he was assuming that hydrolysis of the ethyl isocyanate ought to proceed analogously to the recently published Frankland-Kolbe hydrolysis of ethyl cyanide, that is, to yield propionic acid and ammonia. Thus, reasoning on the basis of the copula theory, Wurtz assumed that the ammoniacal gas released in the reaction was simply ammonia, and he was long mystified at being unable to isolate any sort of oxidized hydrocarbon in solution. It was only when this gas happened to ignite from a fortuitous flame that light shone in Wurtz' mind, as at his laboratory bench: the gas was in fact the chief product of the reaction, an ethyl-substituted ammonia. Wurtz quickly published the result, commenting that ammonia now could be regarded as the "type" for perhaps all organic bases. Wurtz was only repeating the very words of Liebig, who nine years earlier had predicted the existence and even the properties of ethylamine.[43]
Hofmann, who had been working on substituted anilines, felt he had been forestalled; in a letter to his mentor, he remarked on Liebig's clairvoyant prediction. In his reply Liebig also expressed some chagrin, for he had read Wurtz' paper without even remembering his own earlier remarks, and it was only Hofmann's letter that had brought them to mind (a revealing indication of his disinclination toward chemical theory after 1840). "I was so enchanted by Wurtz' work," he added, "that I wrote him and congratulated him on such lovely discoveries. . . ." He went on to say that Hofmann's studies on aniline had been "very interesting . . . every new compound is the first member of
a new series of homologous compounds, and the thought from which the compounds arose is like a seed corn, which bears its fruits in the minds of others doing similar work."[44] Late that year (1849), Hofmann wrote Liebig about his latest syntheses, the details of which were soon to appear in a major paper published in the Philosophical Transactions: secondary and tertiary amines. "These are incredibly remarkable things you have discovered"; Liebig responded, "we are all ecstatic over them. . . . These are very valuable experiments and fully conducive to lead to a definitive view [of the constitution of the organic bases]. . . . This is not a small jump out in front of the French, although I do not mean thereby to include our friend Herr Wurtz."[45] But it was indeed Wurtz' turn to feel forestalled.
The competition between Hofmann and Wurtz in no way affected their friendship. Indeed, in March or early April 1850, Hofmann visited Wurtz in Paris; he not only had a delightful time seeing the sights with his comrade, but described the trip in amazing detail in his biography of Wurtz many years later. During this trip, Baron Thenard hosted a dinner party in Hofmann's honor; his son Paul introduced the company to Hofmann with the words, "Voilà tousles jeunes chimistes de Paris, moins les deux. " Hofmann related this incident in a letter to Liebig, commenting that "this is characteristic of the general mood that has gradually formed in France," that is, against Gerhardt and Laurent.[46]
The relationship between Gerhardt and Wurtz was never more than coolly correct. Gerhardt felt a degree of resentment, bordering on animosity, that the younger Wurtz was so favored by the powerful Dumas: Wurtz was made agrégé (loosely, assistant professor) at the Faculté de Médecine in 1847 and then took over Dumas' lectures on organic chemistry two years later, simultaneously continuing his duties at the École Centrale, all while Gerhardt was without any official position. Particularly galling was Dumas' almost fulsome praise in an official report on Wurtz' papers on amines, when in fact Gerhardt felt that Wurtz had used some of his own work without acknowledgment.[47] "Quelle chance il a, çe garçon," he later exclaimed to a correspondent.[48]
In a major summary of the chemistry of the new aliphatic amines that appeared at the end of 1850, Wurtz tried to apportion proper credit to his predecessors and competitors, especially Hofmann and Gerhardt—which he had certainly not previously attempted. In a theoretical conclusion he argued, as Hofmann had earlier that year, that his and his colleagues' results could most easily be explained by expanding Liebig's amidogen theory to include secondary and tertiary amines, in which alkyl radicals such as methyl, ethyl, propyl, and butyl
substitute for hydrogen atoms in ammonia. He also argued that these results were fully compatible with, indeed provide further support for, Kolbe's idea that the homologous organic acids were alkyl radicals substituted for the nonbasic hydrogen of l he simplest acid (although that series progenitor he took, following Gerhardt, to be formic rather than oxalic acid). He also strongly affirmed, with Kolbe, that the elements that form organic molecules are not thrown "pell-mell, without order, without arrangement, without predisposition" (as Gerhardt had sometimes implied), but rather are arrayed in a definite and ordinarily stable structure. He felt that determining those molecular constitutions was one of the most important goals of chemical theory.[49]
The early 1850s were a busy time for Wurtz. He relinquished his position at the École Centrale to open a private chemical laboratory school and to take a position at the short-lived Institut Agronomique in Versailles. The lab school folded in 1851, and the Institut was closed the next year. In the meantime, Wurtz courted and wed Constance Opperman, the daughter of a wealthy Paris banker. At about this time (the beginning of 1852), Dumas decided to take advantage of Wurtz' linguistic skills and his German and English connections by inviting him to prepare abstracts of selected foreign articles for every monthly issue of the Annales de chimie . Finally, in 1853 Dumas retired from teaching in the Faculté de Médecine; Orfila's death at almost precisely the same time (12 March) gave Wurtz two chairs in the Faculté, along with the financial and career security he had been seeking.
The Breakthrough of Gerhardt's Reform
By this time, the Gerhardt-Laurent reform had begun to make headway in the chemical world. Although most French chemists, including all the influential ones, continued publicly to oppose Gerhardt and Laurent, they did begin to receive some private sympathy. As early as 1846 Thenard was treating Gerhardt kindly again, and Dumas began quietly to support Laurent in 1847. In Great Britain and the United States a few disciples began to appear.[50]
It was Williamson's publications on etherification and the water type in 1850-1851 that marked the critical break for the type theory and for its French partisans. His novel syntheses of asymmetric ethers (such as methyl ethyl ether) provided the first compelling experimental support for Laurent's and Gerhardt's conceptions of the relative constitutions of those theoretically most central substances alcohol, ether, and acetic acid. More indirectly, he argued for the truth of the entire new French system, including the atomic weight reform. "In your formulas," Williamson wrote Gerhardt, "I see the future."[51]
On the heels of this work, and taking advantage of the recent research of Holmann, Wurtz, and others, from 1851 Gerhardt developed his modified type theory. In this theory, four inorganic substances (water, ammonia, hydrogen, and hydrogen chloride) served as models for large series of organic derivatives. Ethers, acids, acid anhydrides, aldehydes, and ketones were all schematically derived from water by replacement of one or both hydrogen atoms; similarly, amines and amides were related to ammonia through substitution of hydrogen by radicals, alkyl derivatives were derived from the "hydrogen type," and chlorides from the "hydrogen chloride type."[52]
In 1852 Gerhardt succeeded in producing asymmetric organic acid anhydrides, providing another compelling argument in favor of the view that organic acids must be seen as analogous to water, but containing no water per se even in their hydrated forms. In brief, the dualists assumed organic acid formulas twice as large as Gerhardt's and Laurent's so that every acid could be formulated to have two replaceable hydrogens, namely, the hydrogens of the associated water molecule. Extract the water molecule, the dualists argued, and you produce the acid anhydride. What Gerhardt succeeded in doing, in a complete analogy to Williamson's asymmetric ethers, was to create structurally asymmetric or mixed acid anhydrides, such as acetic-benzoic anhydride. The dualists could write acceptable formulas representing the new reaction, but such an equation predicted two products, a mixture of acetic anhydride and benzoic anhydride. The actual production of a single asymmetric product could only be accounted for by the smaller, non-dualistic formulas for the acids. Another way to put the matter is that the only way to force monobasic acids such as acetic and benzoic acids to form anhydrides is to join two molecules, since the water that must be removed requires two hydrogen atoms from two molecules; the production of asymmetric anhydrides made it inescapably clear that two different molecules must be involved. Thus, there is no pre-formed water in acids, merely replaceable hydrogen atoms.
Gerhardt knew immediately that he had a winner. He was so excited that his letter announcing the discovery to his student Gustave Chancel is barely legible. In May and June 1852 he read two major papers on acid anhydrides to the Académie.[53] The discovery was received with a great deal more immediate acclaim than Williamson's had been, despite the fact that Williamson had predicted Gerhardt's reaction the previous year and that Gerhardt's theoretical argument was identical to Williamson's (Gerhardt did not mention the Englishman's name). Indeed, Williamson's work seems to have been little known, or at least little noticed, on the European continent until 1853.
Even Gerhardt's most implacable adversaries were impressed. After his second memoir was read to the Académie, he wrote Chancel, "You
could not imagine how everyone has changed toward me: Regnault shook my hand, Dumas nearly hugged me, even Fremy paid me the sweetest compliments . . . What a success!" Dumas wrote a somewhat restrained but strongly favorable report on Gerhardt's paper for the Académie. Thenard became an enthusiastic advocate for Gerhardt's career.[54] The following year Gerhardt published a large review article, carefully crafted to be "academic" in tone, although, he thought, "terribly revolutionary."[55]
For once, others agreed with Gerhardt. Chancel congratulated Gerhardt, predicting a "great sensation" in Germany and England, along with many new converts—perhaps even Liebig himself. Indeed, Liebig soon wrote Gerhardt, praising the discovery as "one of the most brilliant of recent times," the (Williamsonian) theoretical argument being "as simple as it is elegant." "It is very strange," he concluded, "that these two theories, although completely opposed, are now unified into a single one which explains all the phenomena in the two senses."[56] Liebig was but echoing the words in Gerhardt's letter to him two years earlier, urging their reconciliation. In October 1853 Gerhardt visited Giessen and was treated very warmly by Liebig.[57]
As Chancel predicted, during the next two or three years the Gerhardtian system began to attract converts, especially in Germany—a story that is explored in greater detail in chapter 6. A few Russian and a few more English and American disciples began to appear. In France, Malaguti, Quesneville, Cahours, Pelouze, and Gerhardt's students Chancel and Chiozza had adopted elements of the reform even before the acid anhydride work.
Important for this turning of the tide was not only Gerhardt's acid anhydride work, but also his massive and magisterial Traité de chimie organique . It was begun in 1851, and by August of the following year, Gerhardt had written close to 600 printed pages worth of manuscript for the first volume, but with no publisher in prospect. It was reading this manuscript in 1851-1852 and conversing with its author that converted Kekulé to Gerhardt's views. On 28 November 1852, Gerhardt wrote Chancel: "Grreat news! I have sold my organic chemistry to Didot for the trifling sum of ten thousand francs." Gerhardt related that Didot had inquired whether Gerhardt might be willing to complete the last unfinished edition of Berzelius' text. He had replied that the book was already out of date, even though the last volume had only appeared two years earlier, but coincidentally, he had the manuscript of a text himself. They agreed on publishing Gerhardt's manuscript under the title "Traité de chimie organique de Berzelius entièrement refondu et enrichi des descouvertes récentes par C. Gerhardt," with the spine reading "C. Gerhardt: Traité de chimie
organique de Berzelius." Gerhardt added that Laurent had joked that if Berzelius had known this would happen, he would have died a week earlier.[58]
The book was published in fascicles starting in June 1853 and was exceedingly and justly popular. Liebig himself heartily praised the work and told Gerhardt on a visit to Giessen in 1854 that the book would have ten times as many German readers as French.[59] Gerhardt worked on it at a furious pace; by February 1856 he finished the manuscript, and the final fascicle appeared in print by May.[60] In five years, he had written a four-volume work of nearly 4000 printed pages. The book provided a detailed description of the entire science of organic chemistry of his day, along with a defense of his ideas. Ironically, however, all but the last volume utilized the older chemical equivalents, for Gerhardt had for once adopted the pragmatic strategy of presenting the material in the most acceptable possible form.[61]
In his preface, dated June 1853, Gerhardt advertised his avoidance of all molecular speculation as a fruitful methodology that he had been employing for the past ten years (since the Précis ). He justified his return to the older Liebig-Gmelin equivalents as a way of showing the world how irrational they are. He would trust to time, he said, the consummation of the reform that his colleagues had not yet generally adopted. But at the end of the preface, he hinted that the time may now have arrived, following the work of Williamson, Hofmann, Frank-land, Wurtz, and others, to begin building more than just a conventional system of chemical theory.[62]
Finally, Gerhardt had achieved the career success he had been seeking, though unfortunately not in Paris. In 1855, through Thenard's influence, Gerhardt was offered and accepted two professorships at his hometown university, Strasbourg. In April 1856 he was named Corresponding Member of the Académie, a cherished honor. However, in August of the same year, soon after the publication of the final theoretical volume of his Traité and just when it appeared Gerhardt might finally be able to enjoy the acclaim and financial security he had long wanted, he died of a sudden fever.
The Conversion of Wurtz
Meanwhile, Wurtz had reached a decisive turning point in his theoretical commitments. On 18 July 1853, Gerhardt read to the Académie what Wurtz subsequently characterized as "one of his most beautiful papers," demonstrating that not only could alkyl radicals substitute more than once in ammonia, but acetyl and other acyl radicals could do the same, producing secondary amides. What was particularly
remarkable about these compounds 'was that the basic property of the ammonia was not simply suppressed, but rather was eliminated entirely; the new substances were acidic , even though derived from the "type" of ammonia.[63] Whereas Dumas in the older type theory had declared arrangement (or "constitution") to be the principal determinant of chemical properties, Gerhardt was arguing a thesis of dualistic chemical theory, that the kinds of atoms in the molecule determine properties; or, more precisely, Gerhardt had taken the flexible position that both factors were decisive.
Two weeks later, Wurtz provided an elaboration of Gerhardt's idea.[64] He found that his isocyanates react with acetic anhydride to yield a tertiary amide containing one ethyl and two acetyl radicals united by a nitrogen atom, but he expressed the results in terms of the water rather than the ammonia type. He then added, "I note here that the relations that exist between water and substances derived from the water type are expressed in a neater and simpler manner with the aid of the equivalents adopted by M. Gerhardt than by employing the notation ordinarily used."[65] Moreover, he found the opportunity to state that he regarded ether as containing two (rather than one) ethyl radicals substituting the hydrogen of water (rather than adding to oxygen), a hallmark of Laurent's and Gerhardt's new ideas. Hitherto he had been faithful to Liebig's and Dumas' conviction that alcohol was nothing more than a hydrated form of ether. However, although here clearly signaling his new view that major elements of the Laurent-Gerhardt system were superior to the older one, he failed to use the new "equivalents" in this paper.
We possess an eyewitness report of the personal encounters between Wurtz and Gerhardt that summer. A student of Wurtz reminisced:
There [in Wurtz's laboratory] lively, passionate scientific discussions were held between the two young masters. Gerhardt, of an outspoken, brusque, and violent character, and Wurtz, fiery, but glib and subtle, argued over atoms and molecules. At times Gerhardt, whose conviction was profound, became impatient at the resistance of his contradictor, and I have seen him break in his fingers the piece of chalk with which he had just traced the formulas on the blackboard.[66]
Gerhardt responded formally at the Académie to Wurtz' paper. He declared that it made little difference to him whether the new amides were portrayed as derived from the water or the ammonia type, for all of his formulas were only synoptic —general, flexible, and empirical and designed to do no more than summarize the experiments. Wurtz, however (he said), was still under the thrall of Dumas' types. Accord-
ing to that concept, the chemical properties of the derivatives had to be directly related to the properties of the progenitor, so it made no sense to classify acidic derivatives under a basic type. Gerhardt concluded that the issue came down to Wurtz' treatment of formulas as hypothesized before experiments and used as a guide in interpreting them, a procedure that once more revealed Dumas' influence.[67]
Two weeks later (on 29 August 1853), Wurtz responded in turn. Yes, their viewpoints regarding the constitution of the amides were essentially the same since they were assuming the same groupings of radicals. Yes, his attitude toward formulas was different from Gerhardt's. His were constitutional in orientation, possessing a "true molecular signification," indicating, inter alia, "the arrangement of the simple or compound molecules," by which he meant, in modern vocabulary, the arrangement of atoms and radicals within the molecule. But no, he did not regard types as "purely mechanical and inert" with respect to properties; the type imprints "un cachet particulier" on all its derivatives, hence the inappropriateness of deriving acidic secondary amides from the basic ammonia type.[68] In all of this Wurtz was thereby pleading guilty, and proudly so, to Gerhardt's charge that Wurtz was still loyal to Dumas' type theory. It was only in his first systematic theoretical treatise a decade later that Wurtz finally averred, fully in the spirit of Gerhardt, that for the sake of simplicity, compound amides should be regarded as ammonia derivatives even though they do not resemble amines chemically.[69]
It would seem that it was only during 1853 that Wurtz became aware of Williamson's dramatic experimental work on the ethers and his theoretical development of the water type, for his first mention either of Williamson's name or of the water type occurred in his first paper of this series. Williamson had published his three seminal papers on the water type in 1850 and 1851, but only the first had appeared in a French translation and that was not in one of the principal French chemical journals. Wurtz may have first come across Williamson's work in connection with his duties as foreign correspondent for the Annales de chimie (after January 1852), through his visits to England and his friendship with a mainstay of the London chemists, Hofmann, or through his discussions that summer with Gerhardt. Once familiar with the work, however, he quickly assimilated it to his own concerns, as documented in the series of papers just discussed. It must have been in the summer or autumn of 1853 that he wrote Williamson to ask him to prepare a résumé in French of all three articles for publication in the Annales . The sixteen-page résumé appeared in the January 1854 issue accompanied by editorial notes by Wurtz explaining the background and circumstances.[70]
Wurtz' high opinion of Williamson's work is revealed in a letter of 18 April 1854.
I would be delighted, my dear Williamson, to send you occasional summaries of my investigations; I will do this each time I present something to the Institute and even before I have fully completed my researches. I hope thereby to be in the position soon of proving to you my good will!
He then praised Williamson's most recent publication, on sulfonyl chloride, and offered to publish it in the Annales de chimie . He added, "I must tell you that your article on etherification has created a sensation" in Paris.[71]
Clearly it had created a sensation with Wurtz, at least. Williamson had developed a strongly realist and constitutionalist program in connection with the water type; his formulas, he stated carefully, depict "an actual image of what we rationally suppose to be the arrangement of constituent atoms in a compound." In his third paper (1851), he had even suggested a mechanistic interpretation of Wurtz' 1849 isocyanate hydrolysis.[72] Wurtz had always signaled a similar constitutionalist orientation, but until this point he had not found the ideas of Laurent and Gerhardt compelling. Wurtz' reorientation dated from his polemic with Gerhardt in August 1853 and his approximately simultaneous discovery of Williamson—not to mention his promotions. But clearly he much preferred Williamson's realist interpretation of the new chemistry to Gerhardt's positivist one.
Wurtz stated this reorientation unambiguously in subsequent papers. In later historical accounts, he repeatedly accorded Williamson principal authorship for the newer theory of types—even after he became an ardent champion of Gerhardt.[73] In 1855 he published two remarkable papers, both of which show the strong (and explicitly acknowledged) influence of Williamson. Inspired by Williamson's development of the multiple water type, Wurtz showed how a triple water model could well account for the reactions of glycerin, with the glyceryl radical forming a bond ("lien") between the three water molecules.[74] Later that year he revealed what history knows as the "Wurtz reaction," using it to make a theoretical argument —explicitly modeled after Williamson's of 1850—to provide what he regarded as "decisive" and "conclusive proof" that the interpretation of the new school regarding the isolated hydrocarbon "radicals" was superior to Kolbe's and Frankland's explanation. At the end of this paper, he provided a brief but reasonably complete summary of the principal points basic to the new chemistry, unequivocally declaring his adherence to it.[75]
Wurtz' public conversion, we have seen, appears to date from the summer of 1853, the same point when he achieved financial and personal security. Was there a connection? Is it not possible that he was in fact converted much earlier, refraining from declaring himself for fear of alienating Dumas and others during a period when he was still trying to achieve the difficult task of establishing a Parisian career? Although this thesis is attractive, there are difficulties with it. If he were a secret partisan of Laurent and Gerhardt in 1851 and 1852, he would not have gone out of his way during those years to posit rational formulas directly contradicting the new chemistry, such as water = HO, alcohol = C4H5 O,HO, and "sulfobutylate de potasse"[76] =
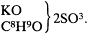
It would have been a simple matter to write formulas that were consistent both with Dumas' and with Gerhardt's and Laurent's ideas. Such empiricist formulas were in fact quite prevalent then and would have excited no comment at all. In such a way he could easily have preserved both his orthodoxy and his flexibility for the future revelations of his present convictions.
If Wurtz had been a closet reformer, he also would not have pursued an oral and written polemic with Gerhardt in the summer of 1853, or at least not with the form and content that we see. Finally, we would not see evidence of a gradual conversion that summer, such as his retention of elements of Dumas' theory. The evidence suggests that he was won to the new views not exclusively or even principally by patronage, peer or career pressures, but by evidence and argument—even though it is true that he may well have felt freer to be convinced by those arguments after the possible career penalties had been removed.
Another sign of the gradual character of his conversion is the curious fact that he continued to refrain from actually using the new "equivalents;" he adopted them in his papers only from the beginning of 1859. Old habits die hard; moreover, he may still have felt a certain loyalty to his patron Dumas that inhibited him, even after he had achieved financial and career security. Another factor may have been what I would characterize as a certain visceral conservatism in Wurtz' character, a trait that can be discerned at many points in the preceding narrative.
Finally, it should be noted that the atomic weight reform of the 1850s and 1860s differed in character from previous seemingly similar shifts. A number of different conventional systems had been proposed and used during the previous decades: Dalton's, Davy's, and Thom-
son's early weights; Wollaston's "equivalents"; the French system initiated by Gay-Lussac and Dumas; Berzelius' atomic weights, represented in two principal modifications (before and after 1826); the proposals of Gerhardt and Laurent, from 1842; and finally, the reform championed by Gmelin and Liebig from about 1845, to which most European chemists pledged fealty. The older chemists had lived through most of these battles and must have been growing tired of the constant changes. Every shift made major portions of familiar pedagogy obsolete, and each time the formulas for most substances had to be recast and committed to memory anew. The latest (Gmelin-Liebig) reform was only a few years old, within memory of even the youngest members of the community, when men such as Williamson and Kekulé began to use the Gerhardt-Laurent two-volume formulas. As noted earlier, even Gerhardt himself had used conventional equivalents throughout most of his Traité , for he was afraid that otherwise the book would suffer poor sales. Indeed, the guiding thought behind the Gmelin-Liebig reform was that the new equivalents were regarded as the most empirical of all possibilities, possessing explicitly conventional status. Since they were fully independent of any theory, they could be used indefinitely: the final and ultimate atomic weight system. In short, resistance by the community to yet another shift was natural and understandable.[77]
Furthermore, advocates of the newest reform were making an important new claim, namely, an ontological one. Despite the positivistic form of Gerhardt's theories, he had no hesitation in declaring the old formulas pure and simply "false" and his the only correct ones. Laurent and Williamson took a much more strongly and explicitly realist stance toward the new weights and formulas, with Williamson providing compelling chemical arguments for their truth. Kekulé made a similar explicitly ontological claim in 1854: "It is not merely a difference in notation, but rather an actual fact" that the formula for water is H2 O.[78] So one may sympathize with Wurtz in pondering his decision whether to sign on to the new reform: such a move would signal not merely an opinion that the new weights and formulas were more convenient, but that they were true . One; would want to be very certain before making such a claim. In 1853 he would only go so far as to say that the new formulas were "simpler and neater," not that they were the only correct ones.
Wurtz appears to have become fully convinced of the actual truth of the Gerhardt-Laurent weights and formulas as a result of the work done during an immensely productive period of his life, 1856 to 1858. During these three years, he published eighteen papers on glycol and its many derivatives and on organic acids, diacids, and hydroxyacids.
He was also influenced by Kekulé's and Couper's 1857-1858 publications on structure theory. Kekulé regarded himself in some measure as a disciple of Wurtz, giving Wurtz significant credit for leading ideas in his major paper of 1858. Couper was working in Wurtz' laboratory when he published a substantially similar paper nearly coincidentally with Kekulé. Wurtz was also a major influence on the third principal founder of structure theory, A. M. Butlerov. In the autumn of 1858, Wurtz finally decided to discard equivalents; in all his papers from 1859, he adopted the new atomic weights and formulas using a notational convention devised by Williamson.[79]
It was just before this final shift by Wurtz that he expressed feelings of isolation and insecurity to his old teacher Liebig. At the last election to membership in the Académie, Fremy had been chosen, and Wurtz conceded that he was not undeserving; but even Berthelot had received more votes that he. Balard had voted for Deville, and Dumas had not supported him. This was an unfortunate omen, indicating that "for my future nothing is secure." He felt more appreciated in Germany than in his homeland.[80] It is curious that this pessimistic letter was written in February 1858, just before Wurtz decided to shift to the new atomic weights and to carry out an extended public campaign for the new chemistry; it also suggests that Wurtz did not yet feel professionally secure. Nothing could have been better calculated to increase his feelings of insecurity and isolation than to begin such a campaign for views that he knew would be unpopular with important people. That he went right ahead with it suggests that he was convinced that the new chemistry was true and would ultimately prevail. He must have been heartened by the obvious successes of the Gerhardt-Laurent reforms in Germany. Until the victory in France was won, however, he was bound to create a professionally uncomfortable life for himself. He could not have known in 1858 that at his death twenty-six years later the reforms would still not have fully succeeded in his native France.