Thirteen—
Variation in the Diving Pattern of Northern Elephant Seals with Age, Mass, Sex, and Reproductive Condition
Burney J. Le Boeuf
ABSTRACT. A principal aim of studies of northern elephant seals from the Año Nuevo rookery in central California has been to obtain complete descriptions of diving behavior for the various stages of life from weaning to adulthood. Studies were conducted during the period 1983–1991 using a variety of methods on known-age animals of both sexes at various stages of development: measurement of the free-ranging diving pattern using attached time-depth recorders, determination of mass before and after trips to sea, and measurement of physiological variables during homing experiments with juveniles.
Some of the principal findings and conclusions are (1) all elephant seals of both sexes and all ages dive deep, long, and continuously for the entire periods that they are at sea; (2) by age 2, the dive pattern is similar to that of adults; (3) dive duration increases slightly with mass in nonpregnant females, but when pregnant females and adult males are included, the relationship weakens, indicating that other variables influence dive duration; (4) dive depth is independent of age and mass in animals older than 2 years of age; (5) adult males migrate farther north and west than females to specific foraging areas along the continental margin, while females disperse more widely in the open ocean and forage en route; (6) pregnant females dive longer and migrate farther away from the rookery than postbreeding females; and (7) yearlings home reliably during the spring molt and fall rest period, revealing a dive pattern like that of free-ranging animals and thus offer the opportunity for short-term studies of diving and the measurement of physiological variables.
In-depth studies of the diving behavior of elephant seals throughout development are filling in gaps in our knowledge of the marine aspect of their natural history and enhancing our understanding of the biology of diving and marine foraging.
Complete dive descriptions for the various stages of life from initial water entry to adulthood do not exist for any diving mammal. This is a serious

Fig. 13.1
Annual distribution of time spent on land and at sea for male and female
northern elephant seals from birth to adulthood. Solid bars represent time
on land, waves represent time at sea, bars and waves represent initial water
experience prior to departure from the rookery. B = birth, W = weaning,
P (for females) = parturition, P (for males) = puberty. Primiparity in females
occurs at 3, 4, or 5 years of age. The numbers in the figure denote
the consecutive trips to sea by each sex.
omission. A thorough description of this kind yields vital information on life history, facilitates the understanding of body functions during diving, sheds light on foraging economics, and puts other dive-related processes in perspective.
One aim of our diving studies at Santa Cruz has been to obtain a description of the diving pattern of northern elephant seals of both sexes dur-
| ||||||||||||||||||||||||||||||||||||||||||||||||
ing all periods at sea throughout development. Except for a single trip to sea during the year preceding puberty, individuals of both sexes spend two periods at sea each year of their lives. The approximate time and duration of these aquatic sojourns for each sex are shown in figure 13.1. This figure makes clear that elephant seals are among the most pelagic of all seals.
I summarize data from 50 dive records obtained from elephant seals at Año Nuevo from 1983 to 1991 (table 13.1), paying special attention to variation in dive pattern with age, mass, reproductive condition, and sex. I also present the main results of a translocation study of yearling elephant seals that shows that it is feasible to study diving in elephant seals in the short run. My aim is to give an overview of certain aspects of the diving research from this laboratory. The treatment will be topical because of space limitations and because each topic will be treated in depth elsewhere.
The general method used to collect all free-ranging dive data consisted of attaching a diving instrument and radio transmitter to a seal shortly before it went to sea and then recovering the instrument when the animal returned to the rookery months later (Le Boeuf et al. 1986, 1988, 1989). Instruments were attached to the pelage above the shoulders with marine epoxy. Nearly all seals were weighed before going to sea and on their return.
Generalizations
The data support several generalizations about diving in elephant seals. All northern elephant seals of both sexes and all age groups, from weanlings on their first pelagic trip to sea to adults, exhibit a diving pattern characterized by (1) continuous diving during all periods at sea; (2) deep diving, relative to other pinnipeds and cetacea; (3) long-duration dives, relative to other diving mammals, interspersed with brief surface intervals (about 1/10 the
mean dive duration) and a few unpredictable surface intervals longer than 10 minutes; and (4) submergence for the majority of the time (83–92%) spent at sea. Data supporting these generalizations are summarized below.
Variation in Dive Pattern
Early Development
First Trip to Sea
On the very first trip to sea when the animals are only 3½ months old, both the depth and duration of dives are great, relative to other diving mammals. Two seals, a male and a female, exhibited mean dive durations of 9.5 and 10.5 minutes with maximum dive durations of 18 and 22 minutes, respectively. The one animal, on whom we measured dive depth over a 30-day period, exhibited a mean dive depth of 206 m and a maximum dive depth to 553 m. These figures are impressive, for they exceed the adult diving performance of most other pinnipeds that have been studied. Further details on the first diving records of these young seals are found in P. H. Thorson and B. J. Le Boeuf (this volume).
Juvenile Diving
Two-year-olds of both sexes dived to the same mean depths, in excess of 400 m, and remained submerged as long as adult males and females, a mean of 18 minutes or more (fig. 13.2). That is, by the end of the fourth trip to sea, when juveniles average 270 ± 26 kg, the adult pattern is essentially set, and 2-year-olds are accomplished divers. This has important implications for conducting studies of diving in the laboratory. It is feasible to transport juveniles but considerably more difficult to transport adult females whose mean nonpregnant weight is 395 ± 19 kg.
Age/Mass Effects
Dive Duration
One expects that dive duration will scale approximately linearly with mass because dive duration is limited by oxygen stores (Scholander 1940; Calder 1984). In seals, oxygen stores are determined mainly by blood volume and hemoglobin and myoglobin concentrations. Blood volume (Vb ) is linearly proportional to body mass (Mb ), Vb = Mb 1.0 , and the relationship between mass and metabolic rate (MR), which determines how quickly the oxygen stores are used, is MR = Mb .75 . In elephant seals, hemoglobin and myoglobin concentrations increase with age (Thorson and Le Boeuf, this volume), and there is a high and positive correlation between age and mass (fig. 13.3; Deutsch et al., this volume; Clinton, this volume). Indeed, one expects dive duration to scale to both age and mass in postbreeding females because of the close relationship between these two variables (fig. 13.3).
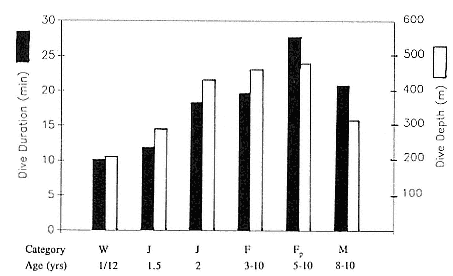
Fig. 13.2
Mean dive duration and mean dive depth of free-ranging northern elephant seals
as a function of age.
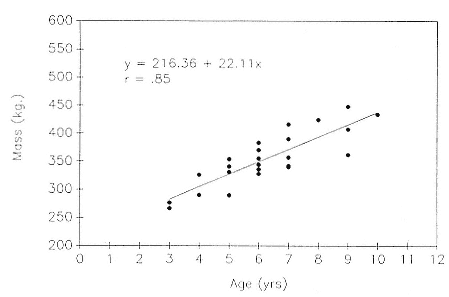
Fig. 13.3
The regression of mass on age in 23 postbreeding, nonpregnant female northern
elephant seals.
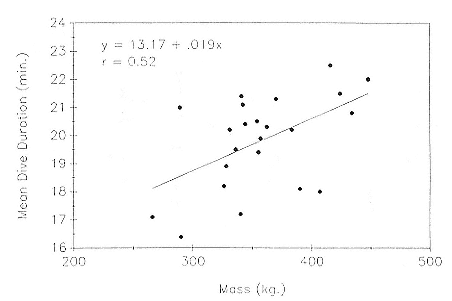
Fig. 13.4
Mean dive duration as a function of mass in 23 postbreeding, nonpregnant female
northern elephant seals.
The relationship between dive duration and mass is positive in postbreeding females, but the association is not strong (fig. 13.4). If one adds weanlings, juveniles, pregnant females, and males to the picture (fig. 13.5), predictability worsens at the upper end. Indeed, it is not clear whether it is males or pregnant females that are outstanding. The long dives of pregnant females contradict the logic that the fetus parasitizes the stores of the mother, causing a shortening of dives. Whatever the explanation, the data indicate that mass predicts dive duration up to a point, but other factors besides mass and oxygen carrying capacity affect the duration of dives. Dive duration does not scale in a simple linear way with mass.
Dive Depth
Mean dive depth does not vary systematically with mass (fig. 13.6), and by association, with age (fig. 13.3), in nonpregnant females. Similarly, there is no relationship between mean dive depth and age across animals from all age categories (fig. 13.7).
Reproductive Condition
Pregnant females, whose diving behavior was recorded during the third trimester of pregnancy, had mean dive durations 39% longer than nonpregnant females (table 13.2). Each one of them had maximum dive durations in excess of 1 hour. Dives lasting more than 1 hour were never observed in
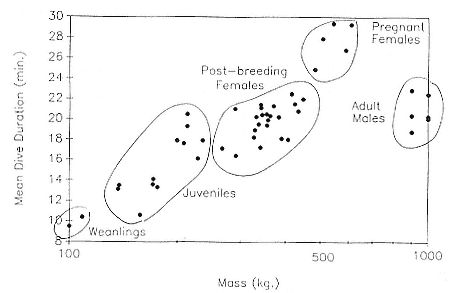
Fig. 13.5
Mean dive duration as a function of mass in northern elephant seals ranging in age
from weanlings to adults of both sexes.
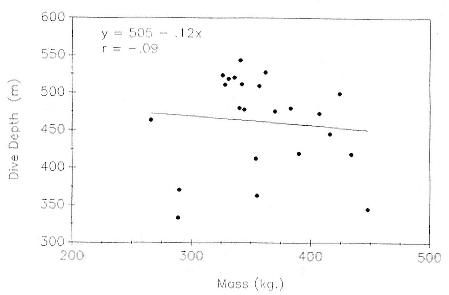
Fig. 13.6
Mean dive depth of 23 postbreeding, nonpregnant female northern elephant seals as
a function of their mass.
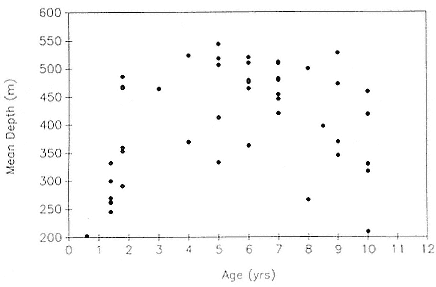
Fig. 13.7
Mean dive depth as a function of age in northern elephant seals.
the 26 nonpregnant females that were recorded. Despite the longer dives, the surface intervals of pregnant females were no longer than those of nonpregnant females. Moreover, pregnant females exhibited slower dive rates and spent less time at the surface than nonpregnant females. These differences were even more prominent in three females that were recorded in both conditions. Differences of similar magnitude have been observed in southern elephant seals, M. leonina (Hindell 1990; Hindell et al. 1992; Slip, Hindell, and Burton, this volume).
Why do pregnant females dive longer than nonpregnant females? One reason might be that it takes pregnant females longer to reach the same depths, the deep waters where prey are located. Pregnant females have a greater lipid composition than nonpregnant females (36% vs. 24%, on average), which makes them more buoyant; increased buoyancy implies greater diving effort and time to reach depths. This is suggested by M. A. Hindell's (1990) observation that pregnant and nonpregnant females do not differ in the duration spent at the bottom of dives; the differences are in descent and ascent duration.
The proximate mechanisms enabling pregnant females to dive longer than nonpregnant females must be either greater oxygen availability or reduced demand for oxygen. Pregnant female mammals are said to have about 40% more blood than nonpregnant females; however, it is not clear how much of this extra blood is involved in nurturing the developing fetus. Alternatively, pregnant females may have lower metabolic rates than non-
| ||||||||||||||||||||||||||||||||||||||||||||||||
pregnant females. A preliminary observation consistent with this statement is that the horizontal swim speed of females during migration is significantly slower during the third trimester of pregnancy than when they are not pregnant (51.5 km vs. 75.0 km per day; t = 2.62, df = 2, p < .05). D. Renouf et al. (1991) report that gray seals, Halichoerus grypus , have lower resting metabolic rates when pregnant than when nonpregnant.
It is also possible that the differences in dive durations of pregnant and nonpregnant females reflect a change in prey or its availability (Lyons 1991). Pregnant females migrate farther away from the rookery than nonpregnant females, but much of the same route is traveled (see below). The diving records under both conditions are similar and offer no obvious evidence for this hypothesis.
Sex
Do the sexes differ with respect to diving pattern and migratory path? The great sexual dimorphism in this species makes this an interesting question: What are the bioenergetic consequences of extreme sexual dimorphism in size, especially with respect to foraging behavior?
Dive Pattern
Males and females have similar dive patterns, except that males dive approximately 180 m less deep than nonpregnant females, on average (table 13.2, fig. 13.2; see also DeLong and Stewart 1991). The maximum depths we have recorded are 1,503 m for a 5-year-old subadult male and 1,273 m for a 4-year-old female.
The shallower dive pattern of males relative to females is due mainly to a single sex difference in diving pattern (Le Boeuf et al. 1993). The most common dive type in the records of females appears to serve pelagic foraging (Le Boeuf et al. 1988, 1992); these dives are characterized by direct descent to depth, with several vertical excursions at the bottom of the dive, followed by direct ascent to the surface. Males exhibit these dives, too, but their most frequent dives are flat bottomed with a mean depth of 331 ± 243 m. Females do not exhibit these dives or do so only rarely. The dive shapes and the location in which they occur suggest that males are pursuing benthic prey on the edge of the continental shelf or on seamounts or guyots. Because males are much larger than females (Deutsch et al., this volume), their daily energy requirements are approximately three times greater than those of females. Benthic prey might provide the additional energy a male requires, or it might be that it is less costly for males to catch these prey. This hypothesis is consistent with reports that males but not females feed on cyclostomes and elasmobranchs (Condit and Le Boeuf 1984; Antonelis et al., this volume). What prevents females from doing the same thing? Two reasons come to mind. They may not be large enough to capture benthic
prey, or it may be more costly for females to migrate to the places where these benthic prey are located.
Migratory Routes
To determine migratory routes and possible foraging areas, we attached geographic location, time-depth recorders (GLTDRs) to 21 animals: 9 postbreeding, nonpregnant females; 5 postmolt, pregnant females (3 of which were also recorded as postbreeding females), 6 males (3 adults of age 10 or over and 3 subadult males, 5–6 years of age), and 1 juvenile female, 1.4 years of age, on its third trip to sea. A photocell in each GLTDR recorded light levels, providing estimates of the time of sunrise and sunset as the animal traveled across latitudes and longitudes; from these data, an algorithm calculated geolocation (DeLong, Stewart, and Hill 1992; Hill, this volume). Geolocation estimates of latitude were adjusted in accordance with surface temperatures recorded by the GLTDR matched to mean sea surface temperature locations compiled semimonthly from satellites by the National Meteorological Center of the National Weather Service (Ashville, N.C.); that is, no geolocation estimate was accepted unless it was consistent with the surface temperature in the area. Recent validation experiments of geolocation from GLTDRs against a ship's Global Positioning System (GPS) off the coast of British Columbia, Canada, in June and July 1992 indicate that light-level estimates have a southerly bias (S. Blackwell and B. Le Boeuf, unpubl. data).
Despite the error associated with light-level estimates of location (Hill, this volume), preliminary data reveal that both adult males and postbreeding females migrated long distances and the migratory paths of the sexes overlapped in the eastern Pacific, especially along the coast up to about 58°N Lat. (fig. 13.8). The most northerly and most westerly migrations were undertaken by males. One adult male traveled as far as the eastern Aleutian Islands in southern Alaska, a round-trip migration of approximately 7,500 km; two others remained near the coast and went no farther north than about 49 degrees, near the state of Washington. Postbreeding females moved in a broad expanse of the eastern Pacific, from near the coastline to as far west as 150°W Long. The longest round-trip migrations of postbreeding females were about 4,866 km. The juvenile female traveled far north into the Gulf of Alaska, exceeding the distances traveled by most postbreeding, adult females.
Pregnant females had minimum round-trip migrations of about 3,900 to 6,800 km. Three of them, recorded when pregnant as well as nonpregnant, took similar routes under both conditions (fig. 13.9). The distance traveled during these biannual migrations was always less during the nonpregnant period.
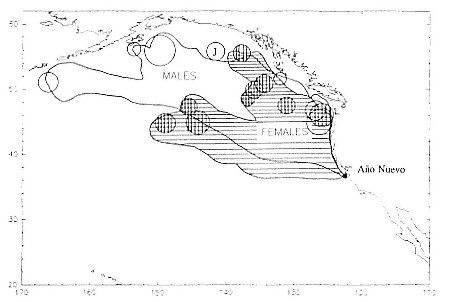
Fig. 13.8
Migratory destination or location at the end of recording of 6 breeding-age males
(open circles), 9 postbreeding females (vertically hatched circles), and 1 juvenile
female (J) that went to sea at Año Nuevo, California. The migratory paths of
males are shown in the unhatched area; those of females are shown by
horizontal hatching. The ultimate locations were derived from tracts plotted
every two days as illustrated in figure 13.9.
The data suggest several points that merit further study:
1. Geolocation by light levels—corrected with sea surface temperature—is not accurate enough to place a seal in a specific location, such as correlating location with bathymetry, but it is sufficient for showing general migration direction and distance.
2. The data presented here, along with more recent findings (Le Boeuf et al. 1993) and reports of other investigators (DeLong and Stewart 1991; DeLong, Stewart, and Hill 1992; Stewart and DeLong, this volume), provide evidence of sex differences in foraging behavior. Adult males migrate to the northern edge of the North Pacific from the state of Washington west to the eastern Aleutians; they move directly to a foraging area, as defined by concentrated diving in this vicinity for up to two months. The migrations of adult females are more removed from the continental margins and are characterized by steady movement in the open ocean in the general range of 44–52°N lat.; females do not forage in narrowly focused areas but forage steadily en route. Differences in the frequency of dive types suggest that
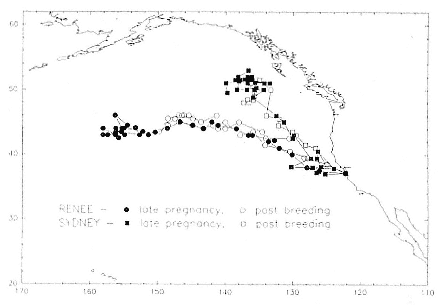
Fig. 13.9
Similarity in the migratory paths of females when pregnant (third trimester) and
not pregnant (postbreeding period at sea). The pregnant period included the
months November, December, and mid-January; the nonpregnant period
included March, April, and mid-May. The records of pregnant females begin at
roughly the furthest point from Año Nuevo; during the postbreeding period,
the records begin at Año Nuevo and end at or near Año Nuevo. A third female,
"B857," followed a similar route during both trips to sea; her record is not
shown because it overlapped with that of the female "Renee."
males are diving in areas where they can reach the bottom and that females are diving in the water column in deeper water (Le Boeuf et al. 1993); however, the geolocation data available are too imprecise to test this hypothesis.
3. Adult seals from Año Nuevo migrate to the same general areas as adult seals from San Miguel Island, located 450 km south of Año Nuevo (DeLong and Stewart 1991; DeLong, Stewart, and Hill 1992; Stewart and DeLong, this volume; Le Boeuf et al. 1993). This suggests that seals from other rookeries in Mexico and California might do the same. This would make for considerable mixing of animals during the foraging periods and would make reassortment on their return to their respective rookeries all the more interesting.
Homing and Translocation
The study of elephant seal diving has revealed a diving pattern that is unusual and difficult to explain with current theory that accounts well for the
diving behavior of the shallower-diving sea lions and fur seals (Le Boeuf et al. 1988). How, for example, can elephant seals spend so little time on the surface following dives lasting over 1 hour? The diving pattern of elephant seals is evidently adapted to spending much of the year at sea and most of time at sea underwater. This regimen, however, is a problem for an investigator who wants to understand the physiology of diving by conducting short-term experiments of the type that have proven so successful with Weddell seals, Leptonychotes weddelli (Kooyman 1981; Guppy et al. 1986; Qvist et al. 1986; Hill et al. 1987). With this practical application in mind, my graduate students and I set out to determine if elephant seals translocated from Año Nuevo would home quickly and reliably.
Recent studies conducted with Guy Oliver, Pat Morris, and Phil Thorson showed that 43 of 45 juveniles, 1½- to 2-year-olds of both sexes, translocated from the Año Nuevo rookery in the spring when molting and in the fall when resting to release sites on a beach or at sea up to 70 km away returned "home" to the rookery. Most of the translocated animals wore radio transmitters that facilitated reidentification and determination of the time of return; the two exceptional animals bore no radios and may have returned but were overlooked. Nearly all returnees were back on the rookery within 1 to 7 days.
Sixteen of the translocated animals bore time-depth recorders. When these animals were released in a place where they had to traverse deep water to return to the rookery, a deep diving pattern indistinguishable from free-ranging diving was observed. We have subsequently used this paradigm to conduct doubly labeled water studies (D. Costa and B. J. Le Boeuf, unpubl. data), measure swim speeds (P. Thorson, G. Kooyman, and B. J. Le Boeuf, unpubl. data). This paradigm should facilitate short-term studies of diving, especially physiological studies of diving requiring completion in a day or two.
Conclusion
The study of free-ranging diving of marine mammals is developing fast. At the marine mammal conference in Chicago in December 1991, there were 24 talks dealing with 18 species that were instrumented with radio transmitters, satellite tags, or time-depth recorders or that were tracked with sonar: 11 pinnipeds, 6 cetacea, and the manatee. Increasingly sophisticated microcomputer time-depth recorders with more channels and larger memories are generating a wealth of detail about the diving behavior of pinnipeds. In the near future, it is anticipated that GPS devices will be attached to seals, permitting precise location of the animals during their migrations. This technology is now being transferred to cetacea.
The incoming data on elephant seals show that variation in dive pattern with age, sex, and reproductive condition as well as individual differences in foraging location can be substantial. Parametric studies taking this variation into consideration will be necessary to obtain a thorough understanding of the marine aspects of an animal's natural history. Satellite monitoring of the migratory path and diving pattern of juveniles during the first trip to sea may elucidate the causes of high at-sea mortality.
We are on the verge of learning much about the free-ranging dive pattern of elephant seals, as well as other diving mammals, and about diving biology in general. Studies of the underlying mechanics of the diving pattern of diving mammals is a fertile field for future investigation.
Acknowledgments
Many collaborators played key roles in the studies summarized here, and they have graciously allowed me to refer to unpublished data. These include Dan Costa, Pat Morris, Phil Thorson, Dan Crocker, Yasuhiko Naito, Tomohiro Asaga, Maria Kretzman, Jeanine Williams, Guy Oliver, and many others. The research reported here was funded by grants from the National Science Foundation, the Minerals Management Service, and the G. MacGowan Trust Fund.
References
Calder, William A., III. 1984. Size, Function, and Life History . Cambridge: Harvard University Press.
Condit, R., and B. J. Le Boeuf. 1984. Feeding habits and feeding grounds of the northern elephant seal. Journal of Mammalogy 65: 281–290.
DeLong, R. L., and B. S. Stewart. 1991. Diving patterns of northern elephant seal bulls. Marine Mammal Science 7: 369–384.
DeLong, R. L., B. S. Stewart, and R. D. Hill. 1992. Documenting migrations of northern elephant seals using day length. Marine Mammal Science 8: 155–159.
Guppy, M., R. D. Hill, R. C. Schneider, J. Qvist, G. G. Liggins, W. M. Zapol, and P. W. Hochachka. 1986. Microcomputer-assisted metabolic studies of voluntary diving of Weddell seals. American Journal of Physiology 250: R175–187.
Hill, R. D., R. C. Schneider, G. C. Liggins, A. H. Schuette, R. L. Elliott, M. Guppy, P. W. Hochachka, J. Qvist, K. J. Falke, and W. M. Zapol. 1987. Heart rate and body temperature during free diving of Weddell seals. American Journal of Physiology 253: R344–351.
Hindell, M. 1990. Population dynamics and diving behaviour of southern elephant seals. Ph.D. dissertation, University of Queensland, Australia.
Hindell, M. A., D. J. Slip, H. R. Burton, and M. M. Bryden. 1992. Physiological implications of continuous, prolonged, and deep dives of the southern elephant seal (Mirounga leonina ). Canadian Journal of Zoology 70: 370–379.
Kooyman, G. L. 1981. Weddell Seal: Consummate Diver . Cambridge: Cambridge University Press.
Le Boeuf, B. J., D. P. Costa, A. C. Huntley, and S. D. Feldkamp. 1988. Continuous, deep diving in female northern elephant seals, Mirounga angustirostris. Canadian Journal of Zoology 66: 446–458.
Le Boeuf, B. J., D. P. Costa, A. C. Huntley, G. L. Kooyman, and R. W. Davis. 1986. Pattern and depth of dives in northern elephant seals, Mirounga angustirostris. Journal of Zoology 208: 1–7.
Le Boeuf, B. J., D. E. Crocker, S. B. Blackwell, P. A. Morris, and P. H. Thorson. 1993. Sex differences in diving and foraging behavior of northern elephant seals. In Marine Mammals: Advances in Behavioural and Population Biology , ed. I. Boyd, 149–178. Symposia of the Zoological Society of London no. 66. London: Oxford University Press.
Le Boeuf, B. J., Y. Naito, T. Asaga, D. Crocker, and D. P. Costa. 1992. Swim speed in a female northern elephant seal: Metabolic and foraging implications. Canadian Journal of Zoology 70: 786–795.
Le Boeuf, B. J., Y. Naito, A. C. Huntley, and T. Asaga. 1989. Prolonged, continuous, deep diving by northern elephant seals. Canadian Journal of Zoology 67: 2514–2519.
Lyons, K. J. 1991. Variation in feeding behavior of female sea otters, Enhydra lutris , between individuals and with reproductive conditions. Ph.D. dissertation, University of California, Santa Cruz.
Qvist, J., R. D. Hill, R. C. Schneider, K. J. Falke, G. C. Liggins, M. Guppy, R. C. Elliot, P. W. Hochachka, and W. M. Zapol. 1986. Hemoglobin concentrations and blood gas tensions of free-diving Weddell seals. Journal of Applied Physiology 61: 1560–1569.
Renouf, D., R. P. Gales, E. Noseworthy, and D. Rosen. 1991. Longitudinal studies of seasonal variations in phocid bioenergetics. Ninth Biennial Conference on the Biology of Marine Mammals, December 5–9, 1991, Chicago, Ill.
Scholander, P. F. 1940. Experimental investigations on the respiratory function in diving mammals and birds. Hvalradets Skrifter Norske Videnskaps-Academie, Oslo 22: 1–131.