The Quantum Condition
Heisenberg's solution to this difficulty is found in a letter to Pauli of 24 June 1925.[332] There he gave a general translation of the Bohr-Sommerfeld condition in terms of quantum amplitudes and frequencies. This step was to provide for the missing "integration constants." In terms of the usual Fourier development (still for one degree of freedom),

the action variable
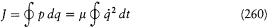
reads
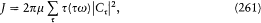
If written under the form "J = nh ," the quantum condition has no clear quantum-theoretical counterpart, since, from previous considerations of formal correspondence, only the translations of tw and Ct were known, not that of t by itself. Heisenberg therefore took the derivative of both members of (261) with respect to J , which gives
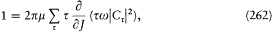
with a straightforward quantum translation dictated by dispersion theory:[333]
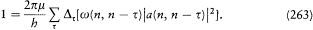
The final quantum condition reads
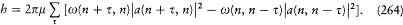
[331] In the same letter to Kronig (5 June 1925), Heisenberg gave a new derivation of Kronig's intensity formulae for the Zeeman effect (see n. 260). This calculation (published in Heisenberg 1925c, 892) was based on a quantum-theoretical translation of the classical rule (236) on p. 237, and of the rule jz = mh (which, in this simple case, could be directly written, without recourse to the general quantum rule later established by Heisenberg).
[332] Heisenberg to Pauli, 24 June 1925, PB , no. 93.
[333] In his letter, Heisenberg assumed the coefficients a (n, n - t ) to be real. As he later realized, this was true only in particular cases (for instance, those treated in the letter to Pauli, fortunately!).
Heisenberg immediately tried it on the harmonic oscillator. In this case the restriction t = ± 1 leads to
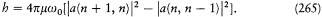
Admitting the existence of a fundamental level n = 0 below which no transition can occur, this equation completely determines the intensities as
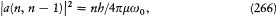
in conformity with Heisenberg's earlier assumption (257).