Chapter 7—
Microtubules
7.1—
Introduction
Microtubules were first described in spermatozoids of the moss, Sphagnum, by Irene Manton in 1957. Since the introduction, six years later, of glutaraldehyde as a fixative for electron microscopy, their presence has been revealed in a very wide variety of plant and animal cells. They have not been observed in prokaryotes.
These organelles are associated with several processes in the repertoire of movements exhibited by the eukaryotic cell, including; movements of individual cells by flagellar motion or axopod extension; movement of components within cells, as in mitosis, axoplasmic flow or transport of certain pigment granules; and morphogenetic movements involving the generation and maintenance of cell shape. In the case of plant cells, in addition to chromosome movements during cell division, the presence of microtubules has been correlated with definition of the plane of cell cleavage, formation of the cell plate and determination of cell wall architecture.
In certain cell types, microtubules are seen only during mitosis and meiosis, suggesting that they were involved originally in these processes alone, and only later did they come to be used for extranuclear functions in advanced eukaryotic organisms. Indeed, it has been suggested (Margulis, 1970) that acquisition of the microtubule was a major factor in permitting the evolution of the eukaryote cell.
7.1.1—
Description
Under the electron microscope, microtubules appear as long, unbranched cylindrical structures with a diameter of 22–25 nm (Fig. 7.1 and Fig. 7.4). In transection, they show an electron-lucent core, ca. 12 nm in diameter, and a wall of, usually, 13 electron-dense subunits. The subunits are 4–5 nm wide and are organized along the long axis of the microtubule into protofilaments. There is an axial displacement of subunits between adjacent protofilaments, resulting in a visible pitch relative to the long axis of the tubule (Fig 7.2).
Microtubules are usually separated from each other or from adjacent cellular components by a clear space of 10–40 nm. Also, in longitudinal view, an electron-lucent zone is often observed along the sides of the microtubule. These observations suggest that each tubule may be surrounded by a specialized region or layer of material. In certain systems, microtubules show 'arms' projecting from the walls of the tubules at regular intervals along their length.
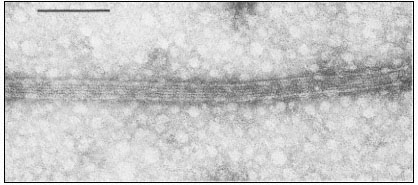
Figure 7.1
Negatively stained microtubules prepared from brain, showing axially aligned
protofilaments and their substructural periodicity. Scale marker 0.1 m m.
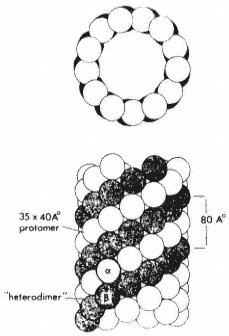
Figure 7.2
Model microtubule, showing in transection the 13 globular
subunits comprising the wall, and in longitudinal view, the
axial displacement of subunits in adjacent protofilaments. From
Bryan (1974) Fedn. Proc. Fedn. Am. Socs. exp. Biol. 33, 152–57,
reproduced by permission of the author and Fedn. Am. Socs. exp. Biol.
Such lateral projections act, in some instances, as cross-bridges between adjacent microtubules. Alternatively, projections can extend from microtubules to adjacent membranes such as the plasmalemma, the nuclear envelope, the endoplasmic reticulum or associated vesicles.
7.1.2—
Background
Eukaryote mitotic spindles are a heterogeneous group of intracellular structures characterized by the presence of anisotropically arranged microtubules. The number of microtubules within the spindle of a given species appears to reflect the mass of the chromosomes, and can vary from a few to several thousand. The highly oriented microtubules are responsible for the weak form-birefringence of the spindle under polarized light and there are natural fluctuations in spindle birefringence during the different phases of mitosis.
Mitotic arrest can be achieved with a variety of physical and chemical agents. Spindle birefringence is also affected by such agents. For example, birefringence in dividing Lilium pollen mother cells is abolished within seconds of exposure to low temperature. Upon return to normal temperatures, spindle birefringence reappears in a few minutes, after which mitosis proceeds normally. Exposure to high hydrostatic pressure causes a similar effect. Chemical agents with antimitotic activity, such as the alkaloids colchicine, podophyllotoxin or vinblastine, also abolish spindle birefringence. The important feature of all these effects is that they are reversible upon removal of the inhibitory agent.
Based on: (a) natural fluctuations in spindle birefringence, (b) reversible effects of inhibitors on both birefringence and mitosis and (c) the findings of many earlier studies that the major components of the spindle are synthesized prior to prophase, Inoué and Sato in 1967 proposed a model in which the spindle is envisaged as a labile structure in dynamic equilibrium with a pool of subunits. Mitosis is thus a process which reflects the sequential assembly and disassembly of different structures to perform different functions. Inhibitory agents may act by disrupting the dynamic equilibrium. This model had a profound effect in engendering the concept of certain microtubules being labile structures capable of being polymerized or depolymerized under cellular control (Tilney, 1971).
However, several lines of evidence suggest that there are different classes of microtubules. Morphological variations in arrangement and in associated components have already been briefly described. Perhaps more importantly, microtubules of cilia or flagella are not depolymerized by treatments which reversibly destroy the birefringence of the spindle. Similarly, certain cytoplasmic microtubules of higher plant cells appear to be resistant to anti-mitotic chemicals. The generalization has been made that microtubules be classified as stable, i.e. those in, for example, cilia and flagella, or labile, i.e. cytoplasmic microtubules in many types of animal cells (Margulis, 1973).
7.2—
Biochemical Studies
7.2.1—
Drug Interaction
The responses of certain types of microtubule to anti-mitotic agents, together with their apparent repeating substructure, thus gave rise to the concept of the
microtubule as an assembled polymer of soluble subunits. Early investigations into the nature of the subunit were hampered by the absence of a method or an assay for the recognition of a soluble microtubule component, even when such enriched sources as flagella or the mitotic apparatus were examined. This problem was overcome in the mid-1960's when it was postulated that the microtubule system was the direct target of the anti-mitotic drug, colchicine, and that the subunit might therefore be recognizable as a drug receptor.
Preliminary experiments with human carcinoma cells in culture established that, at the low levels of colchicine which depolymerized microtubules, there were no effects on other areas of metabolism such as respiration or protein synthesis (Taylor, 1965). Consideration of the kinetics of drug uptake suggested the presence of a single type of binding site. Work in two laboratories confirmed, using radiolabelled drug, that colchicine itself was indeed bound non-covalently to a single, soluble, protein species (Borisy & Taylor, 1967; Wilson & Friedkin, 1967). Since colchicine-binding could be assayed quantitatively, using gel filtration or ion exchange procedures, the reaction assumed great importance in the subsequent isolation and characterization of the subunit (Adelman et al., 1968).
It was quickly established that colchicine is bound in a stoichiometric 1:1 relationship to a dimeric protein with a molecular weight of 120,000 (see Wilson & Bryan, 1974). Denaturation to the protomers results in loss of binding. The binding affinity is high, of the order of 2 × 106 litres mole–1 , and it is important to note that there is no binding at all of the colchicine isomer, lumicolchicine. Although the protein can be stored in the frozen state, an outstanding feature of the binding site is its instability in solution, where it shows first order decay kinetics and a half-life of the order of hours even under optimal conditions of pH, temperature and ionic environment. Inclusion in the medium of the nucleotide, GTP, or the alkaloid, vinblastine, stabilizes and binding activity to some extent.
Evidence that the colchicine binding component is the microtubule subunit was initially circumstantial. High levels of colchicine binding protein were obtained from material rich in microtubules, e.g. dividing cells, cilia and brain cells. The physical characteristics of the binding protein with regard to its molecular weight and dimeric nature were similar to those of the major protein species in flagella and cilia. Evidence of a more direct nature was obtained through ultrastructural observations of the solubilization of the central pair nucleotide, GTP, or the alkaloid, vinblastine, stabilizes the binding activity to binding activity.
The binding site for colchicine seems to be masked when the subunits are assembled into microtubules. This, together with the instability of the site, was responsible for initial lack of success in obtaining colchicine-binding activity from stable outer doublet microtubules of flagella. However, the presence of the colchicine site in such subunits has been demonstrated using appropriate solubilization procedures (Wilson & Meza, 1973). Thus, the colchicine binding
protein has been obtained from a variety of types and sources of microtubule. It is now generally accepted that labile microtubules are depolymerized by the binding of colchicine to subunits in the soluble pool, with consequent disruption of the equilibrium.
The colchicine binding moieties isolated from diverse sources of microtubules display similar physical and chemical properties (see Bryan, 1974). The name 'tubulin' has been applied to this dimeric, functional subunit of microtubules. Tubulin is an acidic, globular protein with a sedimentation coefficient of 6s and an agreed molecular weight of 110–120,000, whose behaviour approximates that of a prolate ellipsoid with an axial ratio of 5:7. The amino acid composition prompted initial comparison with actin, but subsequent comparable peptide maps of both proteins argued against any homology between tubulin and actin (Stephens, 1970). Treatment of tubulin with chaotropic agents such as urea or guanidine. HCl results in a reduction of the apparent molecular weight to 50–60,000. The two protomeric polypeptides can be resolved by appropriate electrophoretic analysis and have been named a and b tubulin, the more mobile electrophoretic component being b tubulin. A number of chemically reactive sites have been distinguished on the tubulin heterodimer. Two sites are known to be involved in drug interaction, a site for colchine and podophyllotoxin, and a separate site for vinblastine. There are also sites which bind endogenous
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
ligands, including a site where GTP (guanosine triphosphate) is tightly and non-exchangeably bound and another site to which GTP is bound in such a way as to allow exchange with exogenously added GTP. Dephosphorylation of the terminal phosphate of GTP and transphosphorylation between the two sites have been suggested to play a role in microtubule assembly. In addition, other sites may be involved in interactions with Ca2+ and Mg2+ ions. The relationship of all these sites to the protein-protein contact sites necessarily involved in the vertical and lateral bonding of the subunits in the assembled tubule is unclear.
Recent examinations of plant preparations for a tubulin-like moiety with characteristic colchicine-binding activity have met with limited success. Such activity has either been undetectable or present only to a low degree (Haber et al., 1972; Hart & Sabnis, 1973; Burns, 1973; Heath, 1975a). The stability and specificity of the binding render it unlikely that all such activity is attributable to a plant tubulin. Table 7.1 shows the levels of colchicine binding activity from a variety of tissues, including plants.
7.2.2—
Polymerization
Since the behaviour of microtubules in vivo obviously involves their strictly regulated assembly-disassembly, a primary aim throughout the biochemical investigations was to develop a system in which polymerization could be studied in vitro (see Borisy et al., 1974). Early studies in this area did indeed demonstrate the presence of ordered structures in tubulin preparations. However, the relevance of such systems to the situation in vivo was considered doubtful, either because the process did not show characteristic kinetic responses to colchicine and temperature, or because the structures did not look like microtubules. It was therefore a notable advanace when authentic microtubules from rat brain, sensitive to colchicine and temperature, were first assembled in vitro, the crucial factors being inclusion in the medium of GTP and the calcium-chelating agent, EGTA (ethylene-glycol-amino ethylether tetracetic acid) (Weisenberg, 1972). It was subsequently reported that polymerization, using pig brain tubulin, also requires nucleation sites which are in the form of discs (29 nm diameter, 17 nm lumen). These successful polymerization studies finally confirmed that the colchicine binding protein is the microtubule subunit. In addition, they have provided a means of demonstrating the probable universality of the tubulin moiety, via co-polymerization of tubulin from such disparate sources as Chlamydomonas flagella and pig brain.
The ability to polymerize microtubules in vitro has initiated several exciting new avenues of research. In the first place, it has provided a method for isolating microtubule protein, rather than tubulin, the drug receptor. Cycles of centrifugation at 37°C and 0°C (i.e. with subunits alternately in the polymerized and depolymerized states) allow microtubules to be isolated with a high degree of purity. Both the ultrastructure and the chemical composition of such preparations have been examined. Electrophoretic analyses of purified microtubules by several
laboratories has confirmed the presence of more than ten other proteins, including some with a molecular weight some with a molecular weight greater than 200,000. The roles of these proteins are the subjects of intense investigation. Some seem to be involved in the regulation of assembly. Others, with a molecular weight close to that of dynein (the ATPase 'arms' of flagellar microtubules) may be involved in the functioning of microtubules.
Secondly, in vitro polymerization has made the assembly process itself amenable to experimental study. The initial test of biological relevance seems settled, with endothermic, reversible, polymerization system responding to many factors known to regulate in vivo assembly of microtubules. The questions of nucleation centres and morphopoeitic proteins have already been mentioned. In addition microtubules have been successfully polymerized in vitro on cellular components such as asters, basal bodies and kinetochores. The pathway of polymerization is being deduced from combined biochemical and ultra-structural examination of experimentally induced intermediates. A commonly observed intermediate is a ring structure of tubulin, varying in dimensions, and complexity with different investigators. Whether the ring structure serves as a nucleation centre for stack-wise growth of microtubules, or whether it uncurls to form protofilaments which then associate laterally, is unclear.
7.3—
Biological Studies
Microtubules appear to be involved in several functional roles within the plant cell (see Newcomb, 1969). For convenience, these can be grouped under five headings: (1) Cytoskeletal role; generation and maintenance of cell shape (2) Cell wall architecture; cell differentiation (3) Intracellular transport (4) Chromosome movements; cell plate formation (5) Cell motility; cilia and flagella. In this section, we shall consider the involvement of microtubules in these phenomena, with emphasis on possible operative mechanisms, and drawing, and drawing, where necessary, on information obtained from studies of animal cells.
7.3.1–
Cytoskeletal Role
Despite the forces of surface tension, animal and some lower plant cells can maintain a non-spherical shape in the absence of a rigid cell wall. Numerous electron microscope studies have revealed that microtubules are present in large numbers and with distinctive orientations in such asymmetric cells or cell extentions. Correlations between cell asymmetry and microtubules have been extensively studied in animal cells, for example in erythrocytes, explanted nerve ganglia or neuroblastoma cells, lens epithelial cells and mesenchyme cells of echinoderm embryos. When these cells are treated with microtubule depolymerizing agents (high hydrostatic pressure, low temperature or colchicine), they assume spherical shapes.
The unicellular alga, Ochromonas, lacks a cell wall or pellicle, yet has a characteristic pear-shape, with a narrow tail or rhizoplast, and a bulbous anterior bearing a small projection, the beak. Two sets of microtubules are involved in maintaining this shape. The plasma membrane of the main cell body is underlain by a series of curved microtubules which extend into the rhizoplast. The other set of microtubules is associated with the beak complex. Disassembly of the microtubules with colchicine or hydrostatic pressure leads to loss of the characteristic cell shape; removal of the depolymerizing agent allows the normal shape to regenerate even in the absence of protein synthesis (Brown & Bouck, 1973).
The two sets of microtubules in Ochromonas are differentially sensitive to colchicine and pressure. At relatively high concentrations of colchicine (10 mg ml–1 ) or at high pressure (8,000 psi), both sets of microtubules are affected but at different rates: first, disassembly of rhizoplast tubules is correlated with disappearance of the posterior tail; this is followed by loss of beak tubules and beak asymmetry. Lower concentrations of drug (5 mg ml–1 ) or lower pressure (6,000 psi) selectively disassemble only the rhizoplast microtubules. These results possibly indicate separate roles for each set of microtubules in determining the shape of this cell. Assuming that different tubulins are not involved, the beak and rhizoplast microtubules may represent two equilibrium systems competing for a common pool of subunits. Alternatively, the differential sensitivity of the two systems, and their sequential reassembly, may reflect characteristics of the sites which initiate microtubule polymerization.
The protozoan, Actinosphaerium, has been extensively used in similar studies on the morphopoietic function of microtubules. The organism is a spherical cell bearing numerous, long, relatively stiff, protoplasmic extensions or axopods, containing a central core, the axoneme (Fig. 7.3a). Each axoneme is composed
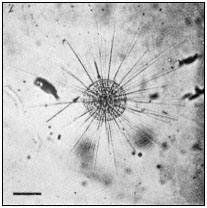
Figure 7.3a
Photomicrograph of Actinosphaerium
showing the slender cell extensions
or axopods radiating from the cell
body. Scale marker 100 µ m.
of several hundred axially aligned microtubules, arranged into two rows that coil about a central axis (Fig. 7.3b). This strict pattern does not seem to be determined by a template of initiating sites, since, during axopod growth, the microtubule array is initially disorganized and becomes progressively more geometrically perfect.

Figure 7.3b
Cross section through an axoneme of Actinosphaerium
near the axopod base. Two spiral rows of microtubules are
coiled about a central axis in a precise geometrical arrangement.
Scale marker 1 µm. From Tilney & Porter (1965) Protoplasma 60,
317–43, reproduced by permission of the authors and Springer-Verlag.
When low temperature, pressure or drugs are employed to depolymerize microtubules of Actinosphaerium, the axopods undergo immediate retraction. In contrast, D2 O, which stabilizes microtubules, prevents the breakdown of axopods by cold or pressure. Retractions and extensions are a normal feature of individual axopods and serve to move the cell across its substratum. Thus, the cell must exert a fine control over the behaviour of microtubules in each axopod separately (see Gunning & Steer, 1975).
In flowering plants, the male sperm is formed from the generative cell which usually has no cell wall and lies within the pollen grain. Growth of the pollen tube is accomplished by the vegetative cell, while the generative cell adopts a spindle-shape before entering and travelling down the pollen tube. Development of the spindle shape is accompanied by the appearance of microtubules aligned parallel to the long axis of the generative cell. Treatment of the generative cell with drugs leads to a loss of microtubules and the cell adopts a spherical form (Sanger & Jackson, 1971).
Thus, microtubules are apparently involved in both generating and maintaining changes in cell shape. The mechanisms regulating orientation and assembly of microtubules in this role are still unknown. Microtubules in the
axonemal cores of Actinosphaerium axopods are connected with their neighbours by cross-bridges. Whether these lateral links function in stabilizing the extended structure or in orienting the microtubules has yet to be determined.
7.3.2—
Cell Wall Architecture
During interphase in higher plant cells, cytoplasmic microtubules lie close to the cell membrane and are arranged circumferentially along the lateral walls of the cells but are randomly disposed underlying the transverse walls (Fig. 7.5a). In the earliest paper reporting the presence of microtubules in higher plant cells, Ledbetter and Porter (1963) noted the parallel alignment of cellulosic microfibrils of the wall and the subjacent cytoplasmic microtubules (Fig. 7.4). Numerous subsequent studies have confirmed this observation, in algal cells and in higher plant cells undergoing both primary and secondary wall formation (see Hepler & Palevitz, 1974).
In the long xylem fibres of Salix, wall microfibrils are deposited in two different orientations simultaneously in the middle and at the extremities of the cell. Even in this situation, microtubule alignment mirrors that of the overlying microfibrils, indicating the ability of the cell to maintain two sets of microtubule-microfibril associations. In the lorica stalk of the alga, Poteriochromonas, the primary wall microfibrils are arranged helically and tend to fasciate into ribbonlike fibrils, 20 nm in width. Every such wall fibril coincides precisely with a microtubule, the plasmalemma separating the two sets of linear structures (Schnepf et al., 1975).
Other examples of the association between microtubules and microfibrils are afforded by cells undergoing irregular or sculptured deposition of secondary wall material. During xylogenesis, microtubules are specifically grouped under the developing wall thickenings in vessels, and, once again, are oriented parallel to the microfibrils. Similar situations prevail during the development of wall thickenings in differentiating guard cells and during the deposition of the nacreous walls of sieve elements.
Attempts have been made to confirm the association between wall microfibrils and microtubules by treating cells with colchicine. However, plant tissues seem relatively resistant to a wide variety of drugs (Heath, 1975b) and, even at high concentrations of colchicine, not all cytoplasmic microtubules in plant cells are depolymerized (Pickett-Heaps, 1967; Wooding, 1969). Also, such levels of colchicine are known to affect other cellular processes, in particular, membrane-related phenomena (see Wilson & Bryan, 1974). Therefore, although wall microfibril orientation is often distorted in the presence of colchicine, such results should be interpreted with caution.
7.3.3—
Intracellular Transport
There is much evidence to indicate that wall precursors are transported to the wall in dictyosome-derived vesicles. On the other hand, there is no evidence to
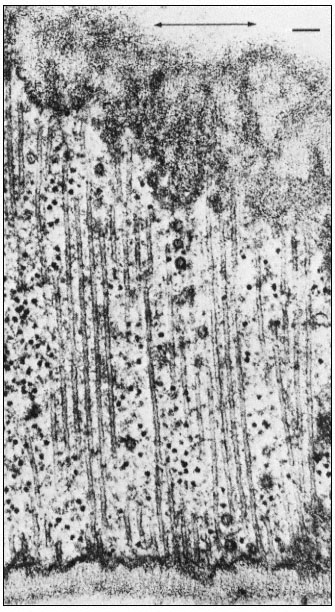
Figure 7.4
Microtubules running parallel to one another immediately beneath
a primary cell wall in root tips of bean ( Phaseolus vulgaris ). The
microfibrils of the wall are oriented parallel to the microtubules. The
axis of cell elongation (indicated by the arrow) is at right angles to the
orientation of the microtubules and microfibrils. Vesicles can be seen
among the microtubules. Scale marker 0.1 µ m. From Newcomb (1969)
A. Rev. Pl. Physiol. 20, 253–88, reproduced by permission
of the author and Annual Reviews, Inc.
suggest that microtubules provide the motive force for such transport. However, it has been proposed that the parallel microtubules girdling the cells of higher plants peripherally provide a framework for the guidance and alignment of these vesicles. In some cases the microtubules lie so close to one another that it has been argued that they could function only by excluding vesicles or elements of the endoplasmic reticulum, preventing their fusion with the plasmalemma at certain sites. In either case, a vectorial role for microtubules is indicated.
Ledbetter and Porter suggested that microtubules may be involved in orienting and even driving cyclosis of cytoplasm, a process which might indirectly be responsible for the alignment of wall microfibrils. However, several lines of evidence have recently implicated cytoplasmic microfilaments in this role (Hepler & Palevitz, 1974). These filaments, 5–8 nm in width, are more consistently located and aligned within the zones of streaming cytoplasm. The microfilaments are morphologically identical to F-actin, one of the major contractile proteins of muscle. Recently, microfilaments in cells of both Nitella and higher plants have been decorated with heavy meromyosin, a characteristic test for actin-like filaments. In primitive organisms, such as the slime mould Physarum, there is overwhelming evidence to indicate that similar micro-filaments provide the motive force for rapid cytoplasmic streaming.
However, around the axonemes of Heliozoan axopods described earlier, cytoplasmic particles stream to and from the cell body. Microfilaments have not been seen in this structure. By contrast, in the giant coenocytic alga, Caulerpa, microtubules are located within the zones of, and parallel to, the numerous sluggish cytoplasmic streams. Mitochondria, plastids and other cytoplasmic components are constantly circulated to and from the growing tips of this large, asymmetric cell. In the chromatophores of animal cells, microtubules have been shown to be essential to the movements of pigment granules. Anti-mitotic agents depolymerize microtubules and simultaneously disrupt the alignment and arrest the movement of the pigment granules (Murphy, 1975). Thus, it remains possible that certain types of cytoplasmic transport require a microtubule framework in both a vectorial and an active role.
7.3.4—
Cell Division
At least two types of microtubule can be distinguished in most spindles, those that traverse the spindle from pole to pole (continuous fibres), and those that connect the kinetochores of the chromosomes to the poles (chromosomal or kinetochore fibres). During separation of chromatids in anaphase, the continuous fibres lengthen, increasing the distance between the poles, while the chromosomal fibres usually—but not always—shorten, further separating the chromatids. After telophase, the continuous spindle may persist and be added to in the interzonal region by numerous additional microtubules, leading to formation of the phragmoplast and eventually, the cell plate.
Considerable changes in microtubule organization (Fig. 7.5, A–F) take
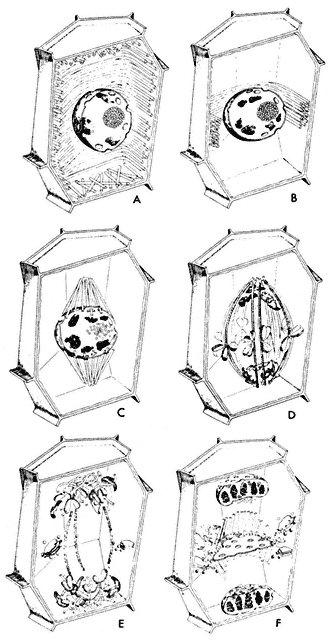
Figure 7.5
Diagram of changes in microtubule orientation and distribution during the phases
of mitosis in a higher plant cell. A—interphase; B—preprophase; C—prophase;
D—metaphase; E—anaphase; F—telophase. From Ledbetter & Porter (1970)
Introduction to the Fine Structure of Plant Cells, p. 44, Springer-Verlag, Berlin,
reproduced by permission of the authors and Springer-Verlag.
place during the events of a typical mitotic cycle (Fuge, 1974). With the onset of division, the peripheral cytoplasmic microtubules of the interphase cell disappear. Concomitantly, a band of microtubules is formed, several layers deep, lying close to the cell wall and girdling the nucleus perpendicularly to the prospective spindle axis. This preprophase band of microtubules appears to predict the plane of the future cell plate (Pickett-Heaps & Northcote, 1966). This has been demonstrated in divisions destined to give daughter cells of different size, for example in the asymmetric divisions of guard mother cells of wheat.
At the beginning of prophase, the number of microtubules in the preprophase band decreases, and new microtubules appear close to the nuclear membrane, this time running parallel to the prospective spindle axis. The microtubules in this so-called 'clear zone' in turn appear to furnish protein material for the spindle. It is possible that a total transformation of these extranuclear microtubules into spindle microtubules takes place.
Prior to spindle formation, the nuclear envelope either disintegrates or becomes perforated at the prospective poles, and cytoplasmic (clear zone) microtubules enter the karyoplasm at these sites. Some of these microtubules appear to establish connections with the chromosomal kinetochores. Since the number of kinetochore microtubules (kMts) increases as prometaphase proceeds, and since clear zone microtubules can be utilized only immediately after breakdown of the nuclear envelope, it is likely that de novo microtubule assembly also occurs at the kinetochores. Thus, it appears that the kinetochore, which varies in appearance in different organisms from amorphous to highly structured, acts as an initiating site or microtubule morganizing centre (MTOC).
In prometaphase, continuous fibres also begin to form and to increase in number. There is some controversy as to whether in higher organisms the continuous microtubules pass from pole to pole. A large proportion appear to overlap in the interzone between the half-spindles (Fig. 7.6), and have been termed non-kinetochore microtubules (nkMts). In lower plants, and all animal cells, with centrioles, the continuous or nkMts originate near the centrioles. These loci are represented by amorphous, electron-dense aggregates of material, and behave as another set of MTOCs. The essential feature of the centriole in mitosis seems to be in determining the spindle axis. In higher plants that lack centrioles, neither the location nor form of polar MTOCs is clear.
During metaphase, engagement of sister chromatids to opposite poles takes place via microtubules, followed by movement of the chromatids to the equatorial plate. At this stage, the maximum number of spindle microtubules is attained, the numbers often trebling between prometaphase and metaphase. Estimates from microtubule counts in serial sections of the metaphase spindle of the sea urchin, Arbacia, indicate a total microtubule length of 50,000 µm. The very large
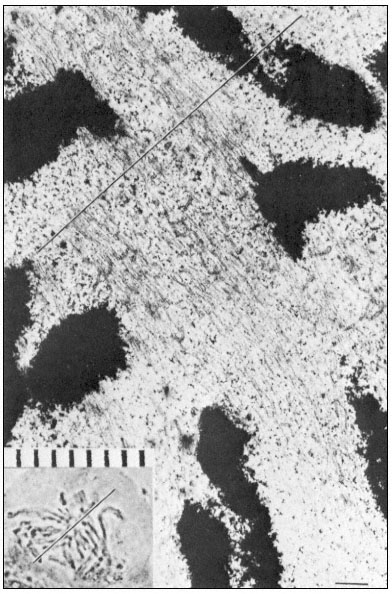
Figure 7.6
Electron and light micrographs of a dividing cell in prometaphase showing
kinetochore and nonkinetochore microtubules. The line indicates the equatorial
plate. Scale marker 1 µm. From Bajer (1968) Symp. Soc. exp. Biol. 22, 285–310,
reproduced by permission of the author and University Press, Cambridge.
spindles of Haemanthus endosperm cells contain considerably more microtubules than those of Arbacia.
Anaphase separation of the chromatids involves direct translation of the kinetochores at almost uniform velocity (0.2–4 µm min–1 ) towards their respective poles. Two experimentally separable processes are involved: first, a movement of the chromatids towards their poles, during which the chromosomal fibres shorten to as little as 20% of their metaphase length; secondly, an elongation of the spindle itself, during which the distance between the poles increases, thereby further separating the two groups of chromatids.
Time-lapse films of dividing Haemanthus endosperm cells show that many other objects, such as vesicles and granules, are commonly transported polewards during prometaphase-anaphase. Conversely, in onion root tip cells, the poleward movement of the chromosomes is accompanied by a reciprocal movement of other materials from the poles towards the mid-plate. Elements of the endoplasmic reticulum accumulate at the poles during metaphase, enter the spindle at anaphase, pass between the chromosomes moving in the opposite direction, and eventually aggregate and coalesce at the mid-plate to give rise to the transverse cell wall at telophase.
Dissolution of kMts and nkMts during telophase is accompanied by the formation of new microtubules in the interzone, leading to the formation of the phragmoplast in higher plant cells (Fig. 7.7). Phragmoplast microtubules appear to be involved in the movement, alignment and fusion of ER- and
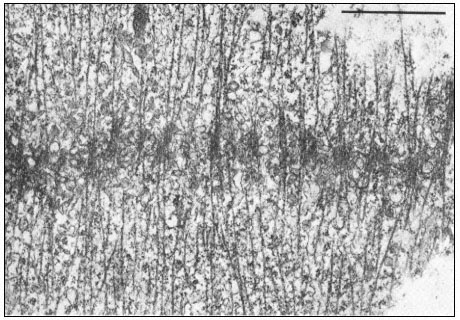
Figure 7.7
Phragmoplast microtubules at the mid-plate during telophase.
Vesicles contributing to the forming cell plate can be seen aligned among the
microtubules. Scale marker 1 µm. From Hepler & Jackson (1974) J. Cell. Biol. 38, 437–46,
reproduced by permission of the authors and The Rockefeller University Press.
dictyosome-derived vesicles to form the cell plate—yet another example of microtubule-associated intracellular transport.
Thus, distinct changes in the polarity and distribution of spindle microtubules take place during the various phases of the mitotic cycle. It should be borne strongly in mind that such changes occur under conditions of very low protein synthesis. Consequently, both assembly and functioning of the mitotic apparatus must involve regulation of microtubule polymerization at a physico-chemical level. Interaction of microtubules with other components of the mitotic apparatus must also be regulated at this level. Several hypotheses have been proposed to explain the mechanism of mitotic movements, although little direct experimental evidence to support the models has yet been obtained.
One prevailing hypothesis to account for spindle elongation (Brinkley & Cartwright, 1971) is that the poles are pushed apart by tip growth of continuous microtubules in the course of which subunits from disassembled kMTs may be utilized. Shortening of the chromosomal fibres is thought to occur by depolymerization at the poles.
Another hypothesis based on the sliding of adjacent spindle microtubules was proposed by Mcintosh et al., (1969). Earlier, several workers had reported the existence of arms or cross-bridges between spindle microtubules. The basis of the sliding tubule hypothesis is that the intertubular bridges in the spindle serve as mechano-chemical elements. Sliding of two microtubules against each other is thought to be generated by the successive breaking and reforming of cross-bridge linkages, a process that is energy-consuming and mediated via ATPase activity of the bridges. It has been estimated that the energy released through the hydrolysis of approximately 20 ATP molecules would suffice to move an average chromosome from the metaphase plate to the pole.
A prerequisite for the McIntosh model is an ordered system of long microtubules, emanating from the poles and reaching far into the opposite half-spindles in metaphase. Microtubules from opposite poles are suggested to have antiparallel polarity with regard to the direction in which their lateral arms could produce force. The kMts in each half-spindle would also show polarity; their lateral arms would only produce force directed towards the spindle equator. If one of the long interpolar microtubules and a kMt of antiparallel polarity came into contact at anaphase, the two tubules, due to the directed activity of their arms, would push themselves in opposite directions by sliding. The model is analogous to the generally accepted model for the interaction of actin and myosin in skeletal muscle. The mechanism could cause chromosome separation, as well as spindle elongation when anti-parallel nkMts interact where they overlap.
A third hypothesis involving actin-microtubule interactions (Forer & Behnke, 1972) has derived from observations of actin-like thin filaments in the mitotic or meiotic spindles of both animal and plant cells. Fluorescein-labelled heavy meromyosin binds to isolated sea urchin spindles. At the ultrastructural level, microfilaments within the spindle have been decorated with heavy
meromyosin, a test for actin-like proteins (Gawadi, 1971; Forer & Behnke, 1972). Several other recent publications have confirmed the presence of actinlike filaments in dividing cells. In kangaroo rat cells, actin was only demonstrable in association with chromosomal fibres (Sanger, 1975), suggesting an actinmyosin type interaction as the force-producing mechanism for chromosome movements, that is, for the autonomous movements of chromosomes in anaphase, as opposed to spindle elongation which may be controlled solely by microtubule assembly-disassembly.
Myosin-like proteins have not yet been located in spindles. Several workers have suggested that microtubules might serve as rigid structures against which the actin filaments could exert a contractile force by the formation and breakage of cross-links. A Ca2+ -dependent ATPase (similar in this respect to both myosin and dynein) is present in the isolated spindle in a concentration three times that in the cytoplasm (Mazia et al., 1972).
The theories on the mechanisms involved in chromosome separation have arisen from observations of living or fixed, intact cells. Such studies are hampered by the inability to experiment directly with the mitotic apparatus, except as it exists in the complexity of its cellular environment. Exciting possibilities have been raised by recent successful attempts to isolate and experiment with the mitotic apparatus in vitro. Intact spindles from the eggs of the surf clam show normal sensitivity to temperature and colchicine. These isolated spindles can incorporate brain tubulin during their reassembly and such hybrid spindles are also cold labile, Ca2+ -sensitive and capable of considerable increase in overall length (Rebhun et al., 1974). Moreover, early anaphase spindles isolated from kangaroo rat cells will continue chromosome motion, in the absence of exogenous spindle subunits, when ATP is added (Cande et al., 1974). These results already suggest that while spindle growth requires microtubule polymerization, anaphase movements do not.
7.3.5—
Cell Motility; Cilia and Flagella
Microtubules, arranged in a characteristic 9+2 configuration (Fig. 7.8a), are the major structural components of flagella, cilia and sperm tails (Warner, 1974). The flagellar apparatus consists of a membrane-bounded, slender, cylindrical cell extension subtended by its basal body, the intracellular organelle which appears to be its origin and kinetic centre.
Basal bodies are identical in structure and apparently homologous with the centrioles of animal and lower plants cells. During mitosis, centrioles appear to determine the poles of the spindle axis and to organize the assembly and alignment of spindle microtubules. Following mitosis, in flagellated cells, the centrioles migrate to the cell periphery and organise the assembly of the flagellar apparatus. The mechanism whereby centrioles act as microtubule initiating centres, the reasons for their stability during the life cycle of the cell, and the factors controlling basal body replication in multiflagellated cells, are all unknown.
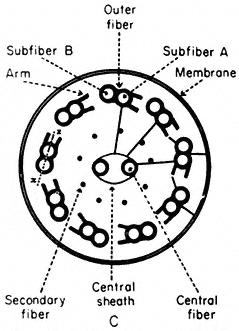
Figure 7.8a
Diagram of flagella cross section, showing
arrangement of microtubules, arms and cross-links.
Basal bodies are composed of nine sets of triplet microtubules (A, B and C) with lateral connections between the A and C tubules of adjacent triplets. At the proximal end of each basal body, a thin filament runs from the A tubule of each triplet to a central hub, forming a cartwheel pattern. At the distal end is the complex transitional region between the microtubules of the basal body and those of the cilium.
The shaft of the cilium is characterized by a central pair of axially oriented microtubules, surrounded by a ring of nine doublets (Fig. 7.8a). The A sub-fibre of a doublet possesses a complete wall of 13 protofilaments. However, the B subfibre has only 10 protofilaments in its wall, and shares the three protofilaments of the A tubule forming the common partition between them.
From the A tubule of a doublet, two short arms project towards the B tubule of the adjacent doublet. These arms have been isolated and shown to possess Mg2+ -dependent ATPase activity (Gibbons, 1965). The arms are approximately 30 nm long, 9 nm wide, and spaced at intervals of 16–22 nm along the length of the A tubule. The enzyme has been called dynein after its postulated role in converting chemical energy into mechanical force. In addition to the dynein arms, cross-bridges have been seen extending between the outer doublets and the flagellar membrane. Radial links, connecting the central pair of tubules with the outer doublets, have also been described. Further, the two microtubules of the central pair differ from one another, one member exhibits two rows of short projections, 18 nm long, and spaced at intervals of 16 nm. Thus, a complex of radially or longitudinally arranged arms, bridges or filaments is present in the flagellar matrix in association with microtubules (Fig. 7.8b).
The most widely accepted hypothesis to account for flagellar motion is the sliding microtubule model (Satir, 1974). Applied to flagella, the hypothesis states that the force responsible for motion is produced when the axonemal
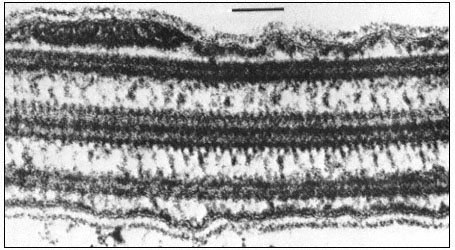
Figure 7.8b
Longitudinal section of a flagellum showing connections between the central and
outer doublet microtubules, and bridges between the peripheral tubules and the cell
membrane. Scale marker 0.1 µm. Fig. 8.8b. from Ringo (1967 J. Cell Biol. 33, 543–71,
reproduced by permission of The Rockefeller University Press.
microtubules, which do not change in length, tend to slide with respect to one another. Accordingly, the model predicts that in different stroke positions, the morphological relationships of the microtubules will change in a systematic fashion, so that the geometry of the bent flagellum will be reflected in the displacement of the microtubules. This has, indeed, been demonstrated using serial sections of the lateral gill cilia of the freshwater mussel.
Other evidence has also accumulated to support this hypothesis. Glycerinated or demembranated flagella beat normally upon the addition of ATP and Mg2+ or Ca2+ ions. When such naked axonemes are briefly treated with trypsin, the circumferential and radial links holding the axoneme together are interrupted, whereas the dynein arms are trypsin-resistant. Addition of ATP now causes a sliding of the microtubules, so that the axoneme grows very much longer and thinner as groups of doublets crawl over one another. This sliding must normally be converted to bending by the series of intermicrotubule connections, within the axoneme.
Thus, microtubules are implicated, in both plant and animal cells, in a variety of processes involving the active movement of cells or cellular components. However, the possibility that a single basic mechanism of action underlies their seemingly diverse roles is still in doubt. The dynamic equilibrium model involving assembly and disassembly of microtubules cannot be applied to the stable microtubules of flagella or the cytoplasmic microtubules of higher plant cells. Neither the assembly-disassembly hypothesis, nor the sliding filament model alone appears adequate to explain the complex functioning of
microtubules in the mitotic apparatus. Even within a process such as spindle elongation, microtubules may function in different ways. For example, the spindles of HeLa cells show an initial rapid phase of elongation, followed by a subsequent slow phase: colchicine only inhibits the slow phase (presumably by interfering with microtubule assembly) but does not affect the rapid phase which may be based on a sliding mechanism. Furthermore, dividing plant and animal cells are remarkably different in their sensitivity to drugs: the spindles of many plant cells require treatment with approximately 1,000-fold higher concentrations of a wide variety of anti-mitotic drugs to achieve mitotic arrest.
7.4—
Concluding Remarks
It is noteworthy that close collaboration between the electron microscopist and the biochemist has been demanded at almost every stage in the development of knowledge concerning microtubules. After the discovery of the microtubule as an ubiquitous organelle of the eukaryote cell, the physiological manipulation of cells in ablation experiments, together with continuing ultrastructural work, confirmed the interaction of microtubules in a variety of cellular activities. Such studies also stimulated development of the concept of the microtubule as a macrostructure in dynamic equilibrium with its constituent subunits. Biochemical investigation into the nature of the subunit initially depended heavily on the electron microscope for evidence that the moiety was relevant to the microtubule. The phase of study culminating in the development of an in vitro polymerization system also relied on the electron microscope to provide evidence of the biological relevance of both the process and final structure.
Knowledge concerning microtubules has been derived mainly from studies involving animal tissues or cells of lower plants. Elucidation of the roles of microtubules in higher plant cells has not proceeded so rapidly. Ultrastructural investigations have confirmed the ubiquity of the organelle and have shown its oriented presence to be correlated with a variety of processes. While it is clear that microtubules are involved in various stages of cell division, their precise role in the earlier events which predict the plane of cleavage is unclear. Similarly, their functioning in other aspects of plant development, including cell wall growth and differentiation, remains conjectural. There are, of course, practical difficulties in studying microtubules in the cells of higher plants. Such problems vary from the general, i.e., lack of tissue specialization and presence of a vacuole and cell wall, to the more specific paucity of microtubules in the meagre protoplasm of the differentiated higher plant cell. In addition, there would seem to be a problem in the resistance of certain plant cytoplasmic microtubules to depolymerizing agents. Since this includes, in some cases, stability to such physical agents as temperature and pressure, it would seem that it is a real property of the microtubule rather than an indirect effect of, say, cellular impermeability towards a drug. This property may be related to the stability of flagellar micro-
tubules. It remains to be seen whether the stability is due merely to a lack of an equilibrating subunit pool or to some other feature of this type of microtubule.
Much remains to be learned about plant microtubules, their subunits and associated components, not only to gain further understanding of their roles in specific processes of plant growth and development, but also for comparison of their properties with those of equivalent components from animal sources. The latter aspect, in addition to answering questions concerning the evolutionary origins of this important group of proteins, would also seem to be a prerequisite to determining any common modes of functioning. How microtubules function, what factors regulate their activity in various cellular processes, and what relationships exist among these diverse activities are some of the questions for research.
Further Reading
Bajer A.S. & Mole-Bajer J. (1972) Spindle dynamics and chromosome movements. Int. Rev. Cytol. Suppl. 3.
Gunning B.E.S. & Steer M.W. (1975) Ultrastructure and the Biology of Plant Cells, pp. 312. Edward Arnold, London.
Hepler P.K. & Palevitz B.A. (1974) Microtubules and microfilaments. A Rev. Pl. Physiol. 25, 309–62.
Inoué S. & Sato H. (1967) Cell motility by labile association of molecules. The nature of mitotic spindle fibres and their role in chromosome movement. J. gen. Physiol. (Suppl.) 50, 259–92.
Margulis L. (1973) Colchicine sensitive microtubules. Int. Rev. Cytol.34, 333–61.
Sleigh M.A. (1974) Cilia and Flagella, pp. 500. Academic Press, London and New York.
Wilson L. & Bryan J. (1974) Biochemical and pharmacological properties of microtubules. Adv. Cell Molec. Biol.3, 21–72.