10. Life History
Short-lived mammals, for example, must always gamble somewhat with their reproduction. They must reproduce as continuously as possible to counterbalance their short life expectancy. Longer-lived mammals can afford to be more selective. All other things being equal then, short-lived mammals tend to be more opportunistic and less seasonal in their reproductive effort than do longlived mammals.
F. H. Bronson, Mammalian Reproductive Biology
REPRODUCTION
the absentee system and parental care
One of the most remarkable features of Tupaia biology is the “absentee” maternal care system.Martin (1968) first described this behavior in captive T. glis belangeri and then in captive T. minor and T. tana (D'Souza and Martin 1974). The basic features of the systemMartin (1968) described are (1) the mother gives birth to her young in a nest that she never shares with them, where they stay until weaning; (2) the mother visits the young to nurse them only once every other day for less than five minutes; and (3) the mother does not show any of the care behaviors most mammal mothers have, such as retrieval of young in distress or grooming or cleaning the young or nest. Tupaia young are altricial: they are born as tiny, hairless pinkies with closed eyes and ears. For the absentee system to work successfully, which it does, an extensive suite of physiological and behavioral adaptations are needed. The nursing behavior of T. tana is described by Miles Roberts, who placed a video camera inside nest boxes with young at the National Zoo:
[The babies] have slept deeply and undisturbed for more than 40 hours, but during the last six hours they have awakened from their deep sleep every 15 to 30 minutes to groom themselves and to remove any traces of urine, droppings, and other material that might leave a telltale odor for a passing predator…. [T]he babies sense [the mother's] presence through vibrations caused by her movement on the outside of the nesting cavity. In response, they push
The mother enters the nest chamber slowly and picks up a leaf…. Quickly, she approaches the babies in the dark. …Without as much as a nuzzle or a lick of greeting, she rears up on her hind legs and extends her forelegs to expose her abdomen to the anxious babies.
The babies frantically nose her abdomen. …Within seconds, each makes contact and nurses at one of her four nipples with what can only be described as hysteria. After a few seconds, each baby switches to another nipple, nurses a few seconds, then switches again to another. Slow at first, the switching tempo quickly increases as the mammary glands empty….
After 60 seconds of nursing, in which they consume nearly one-third of their body weight in milk, the babies are bloated but continue to nurse vigorously. Now, the mother becomes restless. Suddenly, with no warning, without as much as a departing look, she simply vanishes. The babies, engorged and seemingly intoxicated with milk, collapse onto one another and laboriously reconfigure themselves into the huddled ball that will best conserve their body heat. Within seconds, they enjoy the sleep of the dead.
… Except for nursing the young for 90 seconds every other day for the next four weeks—a sum total of about 25 minutes—[the mother's] investment in the young is virtually complete the moment they are born. (Roberts 1993: 6–7)
On this regime captive young Tupaia grow rapidly, almost doubling in weight weekly, until they are weaned and leave the nest at about 25 to 33 days old (D'Souza and Martin 1974; Martin 1968; Roberts pers. com.). In captive colonies weaning is usually abrupt, on the day that the young emerge from the nest box, and from then on the mother does not pay any attention to her offspring. Pentail treeshrews have not been reported to breed in captivity, and their maternal behavior is undescribed.
Elegantly and repeatedly documented in captivity, this curious absentee system still had not been shown to exist in the wild when I started my fieldwork. D'Souza (1972) had reported a possible tree hole nest with young of T. minor, but there were two puzzling inconsistencies in the observations. First, the hole was visited once each day, rather than every other day, and quite late (0900–0915 h); on one day it was visited twice, with an evening visit at 1800 h. Second, D'Souza stated that “characteristic vocalizations of the young” were heard during these visits. Roberts (pers. com.), who recorded with video cameras inside the nests of T. minor and T. tana, never heard young vocalize under any circumstances unless removed from the nest by a human, in which case they emitted only a faint aggressive sound. Paramount among my goals in
TUPAIA TANA Despite much directed effort to find nests with young (chapter appendix), the single “nursery” nest found during the project was by serendipity. On 7 April 1989, at the beginning of the study at Poring, we captured a lactating T. tana (F109) and radio-collared her. Lactating treeshrews captured early in the morning before feeding their young had a sheet of milk covering the abdomen below the skin, whereas those captured following nursing had no milk visible at all. I started a five-day continuous sample to follow F109 on 17 April (we had followed other collared treeshrews in the meantime), and on 18 April at 0706 h, after she had been active for about an hour, I saw her come out of a tree hole that was in plain view of the trail (see fig. 6.6). I instantly suspected that this was the place she had put her young and carefully followed her for four more days. She visited the tree again on 20 April, and I again saw her jump down from it on 22 April, although she had not been there on 17, 19, or 21 April. I had observed unambiguous alternate day visits! Alim used his forester's skill to build a small platform completely screened with palm fronds, 2 m up in a tree a few meters from the nest hole. From there we could watch and videotape F109's visits. In the predawn dark one of us climbed into the blind and waited silently, listening for F109's radio signal through an earphone. Six times we saw her come, but we also watched on some “alternate” days, to verify that she did not approach. We intensively monitored this female through twenty-three days of lactation when young were in the nest and then monitored her for an additional twenty-two days after emergence of the young. We logged fifteen dawn to dusk days of radio-tracking F109 and many partial days. Our detailed observations were published earlier (Emmons and Biun 1991).
On 26 April, just after a nursing visit, we climbed to the nest to find out what it contained. We discovered two young, which we extracted briefly to weigh, measure, and mark with ear tags (fig. 10.1). The young were in a tree hollow with a large entrance 4 m high (fig. 6.6). Within the cavity they were inside a large clean nest of overlapped leaves lined with teased fibers. From the blind we watched six nursing visits to the tree hole. The mother indeed visited her nestlings only once every other day, and she spent an average of 2.74 min within the hollow (N = 5 timed visits; range 1.8–4.48 min). The nursing visits were always early in the morning, between 0615 h and 0734 h. She was videotaped on one visit,

Fig. 10.1. Nestling T. tana on 26 April 1989. Note immobility (it does not move or try to escape).
A notable feature of F109's behavior when she came to nurse her young was that she often varied her pathways, coming and going by different routes. Sometimes she jumped from tree to tree on slender understory saplings, without touching the ground anywhere near the nest tree. She usually did not run up or down the nest tree but jumped to it at the level of the entrance from a neighboring treelet (see fig. 6.6). The radio signal indicated that she often hesitated nearby before running to the nest. Before exiting, she would poke her head out of the hole and scan the surroundings. We never heard any vocalizations or deliberate sounds made by mother or young, and visits were as discreet as possible. At the hour of most visits, the light intensity was still crepuscular, which made it somewhat difficult to see the treeshrew or to get sharp video images. These cautious behaviors should hide the nest from predators that could use the mother's visible behavior or odor trail to find the young.
The pattern of alternate day visits by the mother T. tana only varied once, when on the day before the young emerged the female fed them on an “off” day, so that we assume she came on three consecutive days, the two “normal” ones and the one in between (we did not follow her
On the day that the young emerged, Alim observed the following behavior from the blind. The female arrived at 0643 h, carrying something in her mouth, and went into the nest. Two minutes later she exited slowly and stopped to wait on the branch of a sapling. The two young came out of the hole and shakily climbed around on the side of the tree. When the young reached the ground, wobbly and uncertain, the mother went away for eight minutes and returned with a food item that she gave to them and they ate. She also licked them. She led them away from the nest site by repeated calling, after a period of “training” them to follow her by calling, moving a little away, and waiting for them to approach, then moving away again and calling. The mother then spent the whole day and the first night with her young. They slept on the ground surface in a thicket under the shelter of a log. The second day, she led them to another nest site in a dense treefall, but that night she went back to her own previous nest alone and thereafter did not share nest sites with her young. The mother spent much time with her youngsters after their emergence, including the whole of the first two days (apart from racing once down to the stream and back, presumably to drink). She visited them every day in the morning and often spent many hours with them. She continued to spend time with her young to twenty-two days after emergence, when we had to discontinue tracking. Unfortunately, we did not see what she was doing when with her young, as we glimpsed them only fleetingly; but once a juvenile was seen foraging by itself when she was present. She visited the place where her young were early each morning between 0624 and 0717 h, but otherwise her behavior was unpredictable. She spent the whole day with them on 15 and 18 May and more than three hours on 31 May but only seven minutes on 30 May and eleven minutes on 19 May. Soon after they emerged the young foraged on their own. On 13 May they were seen foraging twice, once biting apart dead branches. The early morning visits suggest that she may have continued to nurse the young after emergence.
THE STRANGE CASE OF THE MISSING MALE At Poring an enigmatic social arrangement of T. tana took place when the female had young. We only observed this once, and we previously described it (Emmons and
| Date | Young | Interactions between Male and Female | |
|---|---|---|---|
| NOTE: The male was recognized by his radio signal, which was searched for often, especially after it “disappeared.” | |||
| 7 April | In nest | F109 first captured, lactating | |
| 17 April | In nest, not nursed | Together 0727–1105 h and 1338–1420 h | |
| 18 April | In nest, nursed | Not together all day | |
| 19 April | In nest, not nursed | Together 0753–0941 h | |
| 20 April | In nest, nursed | Not together all day | |
| 21 April | In nest, not nursed | Together 0642–0710 h | |
| 22 April | In nest, nursed | Together 0635–0710 h, loud, continuous chattering | |
| 25 April | In nest, not nursed | Not together all day | |
| 27 April | In nest, not nursed | Not together all day | |
| 28 April | In nest, not nursed | Together 1103–1400 h | |
| 29 April | In nest, not nursed | Together 0950–1006 h | |
| 7 May | In nest, not nursed | Male nesting at night on female's range | |
| 10 May | Young emerge | ||
| 9–13 May | Male apparently dissappears from area | ||
| 14 May | Male found once on F109's territory | ||
| 15–29 May | Male not on female's territory | ||
| 30 May | Male found nesting far across ravine outside F109 range | ||
| 5 June | Male nesting far across ravine | ||
| 27 June | Radio of male found cast on F109's range | ||
| 15–16 August | Male captured three times on F109's range | ||
OTHER TUPAIA SPECIES Field evidence for absentee maternal care and alternate-day nursing in other Tupaia species was circumstantial. (1) The nest of a lactating T. montana was inspected and found to contain no young. (2) Lactating females of T. gracilis, T. longipes, and T. tana each used several nests within a few days, so the young could not have been in them with the mother (female Tupaia have no behaviors of retrieving or carrying young [Martin 1968]). (3) While they were followed continuously, lactating females generally showed alternate-day patterns of visiting certain areas in early morning and did not show patterns of sites visited at dawn daily or more than once daily. But because nursing visits are so short, and animals in general tend to repeat their favorite travel routes on their home ranges, it was always difficult to interpret their movements unambiguously. I tried to locate nursery nests many times, with different individuals and species, but never succeeded again.
PTILOCERCUS The adult female pentail (F181) was lactating when captured on 22 August 1991. On 28 September I saw a very small young emerge from the nest tree at nightfall within 10 min of when F181 emerged, as well as another large adult and a smaller, probably subadult pentail. The following day, when at least two others emerged with the youngster, the female and another stayed near the nest tree and baby and repeatedly made alarm calls (probably because of my presence). On 4 May 1991 I saw two large pentails emerge, followed 10 min later by two apparently slightly smaller pentails (presumed subadults). On 9 May several of us saw a minuscule pentail emerge from a hole at the nest tree, with at least two others. Again an excited adult remained near the tree, calling at us. Baby pentails therefore shared the maternal nest tree from at least what seemed to be the age of first emergence.
| Species | N | Embryo No. | Reference | |
|---|---|---|---|---|
| 1 | 2 | |||
| NOTE: Data from this study are based on palpations of embryos in living animals, and from one litter in the nest. | ||||
| aSpecimens in Universiti Kebangsaan Malaysia, Kampus Sabah, Museum. | ||||
| T. minor | 4 | 4 | Davis 1962 | |
| 4 | 4 | Harrison 1955 (West Malaysia) | ||
| 1 | 1 | This study | ||
| T. gracilis | 3 | 3 | This study | |
| T. longipes | 2 | 2 | Davis 1962 | |
| 3 | 3 | UKMSa | ||
| 3 | 1 | 2 | This study | |
| T. montana | 10 | 1 | 9 | Kobayashi, Maeda, and Harada 1980 |
| 1 | 1 | UKMS | ||
| T. tana | 3 | 3 | Davis 1962 | |
| 1 | 1 | Kobayshi, Maeda, and Harada 1980 | ||
| 1 | 1 | UKMS | ||
| 1 | 1 | Lim 1965 | ||
| 4 | 4 | This study | ||
I cannot exclude the possibility that the young pentails were in a different, absentee nest before emergence and that the mother then led them to her own nest after emergence (as the T. tana mother led her young), but because of the communal nesting behavior of this species, this scenario seems much less probable than that the young shared the maternal nest from birth (see chaps. 6, 9). However, the lactating female pentail did visit one area on seven of nine nights she was followed (all prior to 28 September), often about 30 min after she became active. This was a vine-choked streamside thicket, where I could not see her. She may have gone there to drink, as lactation would increase her fluid needs. She also visited another area on all nine nights, but at different hours, and she regularly repeated several travel routes. Thus her movements during early lactation did not rule out absentee maternal care, although I believe such a system to be unlikely.
LITTER SIZE
Embryo numbers or litter sizes of the Tupaia study species in the wild seem to be fixed at two, with the occasional single young (table 10.2).
Reports of litter size for West Malaysian T. glis are likewise constant at two (Harrison 1955; Langham 1982). For various subspecies of T. glis,Lyon (1913) reported two litters of three (T. g.ferruginea) in addition to eight litters of two and one singleton. He also noted a T. chinensis with a litter of four and a T. nicobarica with a singleton. These last are of interest because T. chinensis is one of the few species with three instead of two pairs of mammae, and T. nicobarica is one of the few forms with only one pair (Lyon 1913). There do not seem to be any published records of litter size for Ptilocercus lowii, but we saw two different, single, juveniles emerge from the den, while earlier in the year two like-aged subadults lived there, evidence for litters of one or two.
BIRTH SEASONALITY
Juveniles and subadults appeared erratically during the monthly trapping at Danum Valley. Most young were not likely to encounter a trap until they were traveling some distance, at one or two months postemergence (about three months old), so young were quite large at first capture, doubtless because of the combined effects of the long interval between trapping periods, the restricted area used by young, and the narrow region of the traplines. Samples were small for all species, but the combination of dates when young were captured and when females were pregnant or lactating gives a fairly complete history of the reproductive activity of T. gracilis, T. longipes, and T. tana on the study area at Danum Valley during the thirteen months of the study (table 10.3). Evidence for the breeding patterns of T. minor and P. lowii was more spotty.
All Tupaia species at Danum Valley showed similar reproductive seasonality. There was a broad breeding season from August to November and a second one from March to May (figs. 10.2, 10.3). Between each period of increased breeding, there was an inactive period of two months. We saw pentail treeshrew juveniles in May and September, and we caught a subadult in November (probably born in Sept.–Oct.), evidence for the same general pattern. There was a slight seasonal difference in breeding between T. tana and T. longipes at Danum (figs. 10.2, 10.3). In 1991 the large breeding peak was a month earlier (Aug.–Sept.) in T. tana than in T. longipes (Sept.–Oct.), which species also virtually lacked the second breeding peak in late March.
In captivity treeshrews can breed continuously, with females becoming pregnant in a postpartum estrus and birthing every forty-five to fifty
| Female | Sept. | Oct. | Nov. | Dec. | Jan. | Feb. | Mar.I | Mar.2 | Apr. | May | June | July | Aug. | Sept. | Total |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NOTE: Y = young captured on territory; P = pregnant; L = lactating; Yn+n = young of two sequential litters. Total = probable number of litters during months monitored, (3) = number in the calendar year, when animal was monitored longer. Few females were present all year, and some were rarely captured. Female T. tana at the end of the list were new territory holders that replaced previous females. Litter numbers are estimated from the reproductive condition of captured females and the ages of young trapped on their territories. Youngsters from two litters were sometimes present simultaneously. | |||||||||||||||
| aF66 was neither recaptured nor replaced by another after December. She may have been present but trap-shy; this litter was on her territory. | |||||||||||||||
| bNo female T. longipes was trapped on the NW corner of the study area, but one was present, as young appeared. | |||||||||||||||
| Ptilocercus lowii | |||||||||||||||
| P1 181 | Y(2) | No data | Y | L | Y1 | 3 | |||||||||
| T. gracilis | |||||||||||||||
| Tg 67 | Y1 | P | LY1 | Y1? | 3 | ||||||||||
| Tg 173 | p | Y2 | P | 2 | |||||||||||
| T. longipes | |||||||||||||||
| T1 56 | P Y2 | L | Y1 | Y1 | 4(3) | ||||||||||
| T1 66 | Y2 | L | Y1 | Y1a | 3 | ||||||||||
| T1 Xb | Y1+2 | Y1 | 3 | ||||||||||||
| T. tana | |||||||||||||||
| Tt 76 | LY1 | P | Y1 | Y1 | 3 | ||||||||||
| Tt 54 | PY3 | PL | Y2+1 | 4 | |||||||||||
| Tt 58 | L | Y1 | 1 | ||||||||||||
| Tt 78 | Y2 | 1 | |||||||||||||
| Tt 176 | L | 1 | |||||||||||||
| Tt 297 | Y | 1 | |||||||||||||
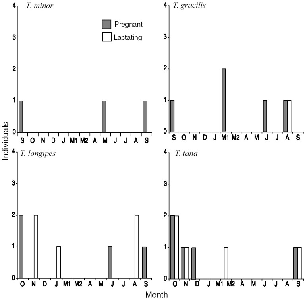
Fig. 10.2. Seasonal reproductive activity of female treeshrews trapped at Danum Valley.
In sharp contrast to the laboratory scenario, reports from field captures and collections of wild treeshrews all show a low overall pregnancy rate. In data from Sabah that Wade (1958) compiled from the April to August collections of Davis, only ten of forty-five adult females were pregnant (22%; three species): none in April or May, two in June, seven in July, and one in August. In West Malaysia Harrison (1955) found a crude pregnancy rate of only 9 percent for both T. glis (N = 53) and T. minor (N = 44). On Singapore Kawamichi and Kawamichi (1979) found no breeding in T. glis during their study from October to December. In
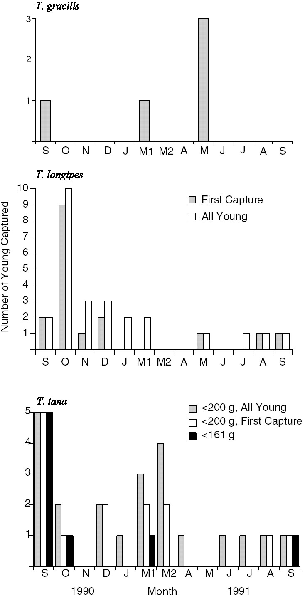
Fig. 10.3. Seasonality of the appearance of young in monthly trapping on the study area at Danum Valley. For T. tana only, young are separated into age groups by weight. All immature treeshrews that are nonparous or do not have adult dentition are classed as young.
Field studies thus show that wild Tupaia species have clear breeding seasonality, especially a lack of breeding in certain months. Moreover, although the areas from which data were drawn are at nearly the same latitude, the reproductive season differs between East and West Malaysia. My data agree with the findings of Wade (1958) for the same region of Sabah. Likewise, more than eighty squirrels in the same collections showed no reproduction in April or May, and only a small amount in June-August (Wade 1958).
reproductive output of individual females
For some resident females at Danum Valley, I can infer the timing of sequential litters from the combination of their reproductive condition when trapped or the captures of young on their telemetry-defined territories. Most captured young can be assigned to a specific female. Two young of equivalent weight on the same territory I assume to be littermates. However, there are some ambiguous cases of young caught on two neighboring territories. Juveniles may not always be aware of territorial boundaries.
The data for individual females show that during each of the broad breeding seasons, some females bred twice (table 10.3). In particular, during the great breeding peak of August-November 1990, most T. longipes and T. tana had two litters in rapid succession, and one female of each of these species seemed to have three litters in this season (F56 and F54). In the March-June season only the two T. gracilis females had two successive litters. During the calendar year of the study at Danum, the individual females, or the successive females on a given territory, produced three to four litters of young each, usually in a pattern of two successive litters in one season and one in the other. Because few females were followed throughout the entire period, better accuracy cannot be achieved from this data set. The females captured in the five months at Poring indicated a similar reproductive seasonality for the lowland population (table 10.4), with breeding in March-April and beginning again
| Female | Apr. | May | June | July | Aug. |
|---|---|---|---|---|---|
| NOTE: Abbreviations as in table 10.3; E = estrus; N = parous, nonreproductive, territorial female.a At Langanan study site (900–1,000 m) trapped only in late June and July. | |||||
| T. minor | |||||
| Tm 112 | Y | ||||
| Tm 119 | L | ||||
| T. gracilis | |||||
| Tg 105 | PY2 | N | |||
| T. longipes | |||||
| Tl 132 | L | P | |||
| Tl 133 | N | N | |||
| Tl 155a | P | ||||
| T. tana | |||||
| Tt B | L | Y | |||
| Tt 109 | LY2 | N | |||
| Tt 106 | N | N | |||
| T. montana | |||||
| Tmo 141a | N | ||||
| Tmo 148a | P | L | |||
| Tmo 150a | P | ||||
| Tmo 151a | P | ||||
| Tmo 154a | E | ||||
Because litter size is nearly fixed at two, a female of T. gracilis, T. longipes, or T. tana would have produced six to eight young in 1990–91. Combined with the data on population density of females (see chap. 8), for six young this translates to an annual production of about 1.5 young/ha for T. tana and about 0.6 young/ha each for T. longipes and T. gracilis.
The only other field data on the breeding of individual treeshrews is Langham's work on T. glis. Most of the fifteen to thirty adult females he was able to capture monthly bred only once in the year, but five older females had two or three litters (Langham 1982). Reproductive output per female may thus have been somewhat lower at his study sites than at Danum Valley in 1990–91.
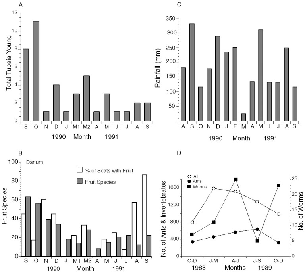
Fig. 10.4. A, the month of first capture of all young of Tupaia species at Danum Valley. B, the number of fruit species collected monthly in the transect of study area trails (shaded bars) and percent of treeshrew scats including fruit (open bars). C, monthly rainfall. D, numbers of litter invertebrates collected in standard litter samples during three-month periods in 1988–89. (Compiled from Burghouts et al. 1992 and. T. Burghouts pers. com.)
environmental correlates of reproduction
Why do treeshrews breed in some months and not others? As in all equatorial rainforest climates, in Sabah neither daylength nor temperature vary sufficiently to influence mammalian ecology, and rainfall is the chief seasonal variable. Rainfall patterns orchestrate the life histories of terrestrial organisms, directly through effects of precipitation and indirectly through proximate control of plant phenological cycles (e.g., Foster 1982).
The patterns of treeshrew reproduction, monthly fruitfall (see also chap. 5), and rainfall (fig. 10.4C) show strong similarities. However, the
Seasonal abundance of soil surface and leaf litter invertebrates next to the treeshrew study plot at Danum from October 1988 to December 1989 show invertebrates to be rarest in October-December (Burghouts et al. 1992) and most numerous from January to June (fig. 10.4). The samples with highest numbers of total invertebrates generally coincide with the March breeding peak of the litter-foraging treeshrews. Numbers of surface earthworms (fig. 10.4) seemed to be directly related to the amount of rainfall, as one would expect (highest in June, July, October, and November in 1989).
growth rates of young
The known histories of all individuals of Tupaia captured at Danum Valley can be plotted to show the phenology of body mass (figs. 10.5–10.7). This gives (1) a measure of the growth rates of young in the field; (2) adult body weight changes that could be linked to breeding seasonality or other factors; and (3) the turnover of individuals on the study area, for visual review. The weights of pregnant females are included.
Several juveniles of T. longipes and T. tana were recaptured as they grew, and five of these remained to adulthood on the study area. In both of these species young born in September or October 1990 showed a very fast weight gain of about 50 to 60 g during about the first month
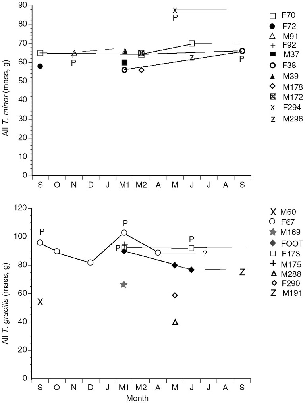
Fig. 10.5. Histories of all individuals of T. minor and T. gracilis captured on the Danum Valley study area, by captures during monthly trapping sessions. P = pregnant; adults = open symbols; young = closed symbols. Thin trailing lines are periods when the individual was known alive, from radio-tracking or sightings, beyond the last recapture date.
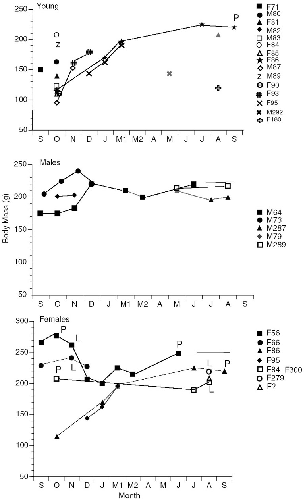
Fig. 10.6. Histories of individual T. longipes trapped at Danum Valley. P = pregnant; L = lactating. Adults = open symbols; young = closed symbols.
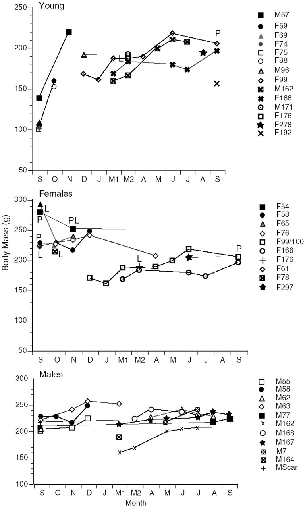
Fig. 10.7. Histories of individual T. tana on the study site at Danum Valley. Young <200 g at first capture (top), resident females, including young that became established on territories and that are also shown in the top figure (middle), and adult males (bottom). P = pregnant; L = lactating.
age at first reproduction
Two females each of large and plain treeshrews that were born on the study area at Danum Valley became resident, territorial adults there. Three of these, one T. tana and two T. longipes, were born in September– October 1990 (F99/100, F81, F86). Two showed their first reproduction in September 1991 and one in August. The fourth female (F166) was probably born in December or January 1991. When last captured in September 1991 she was still nonreproductive. At Poring a subadult female T. tana of 144 g (F122), was established by 2 May, following the April death of the resident, but she was still not reproductive in mid-August. These few cases indicate that T. tana and T. longipes first reproduced when about a year old, even when they had acquired a territory several months previously. The single exception was a young adult female T. tana (F176; fig. 10.8) of 188 g, with unworn teeth, which was captured lactating on 30 March. She may have been born the previous August-September. Her radio signal vanished within two weeks, and she was not recaptured, so her fate was unknown.
The captive-bred treeshrews studied byMartin (1968) mushroomed to adult weight (200 g) by two and a half to three months old, only two months after weaning. Sexual maturity arrived at three months and birth of the first litter at four and a half months. There is even a report of a captive female becoming pregnant at two and a half months (Sprankel 1961). Langham's (1982) excellent field data on the weight gain of wild T. glis in West Malaysia, with measurements of from thirty to sixty youngsters trapped per month, has a virtually identical pattern to that I recorded at Danum Valley. Adult weight in West Malaysia (mean = 149 g) was
SEASONAL PATTERNS OF ADULT WEIGHT
The females of T. gracilis, T. longipes, and T. tana that were followed for several months all showed maximum (nonpregnant) weights somewhere between September and November and falling weights toward December and January (figs. 10.5–10.7). Some also dipped in weight between April and June. Two subadult T. tana lost weight in December and January, and one lost weight in July.
Only in T. tana and T. longipes were males followed for extended periods (figs. 10.6, 10.7). Like the females, males showed maximum weights in the first three months of the study, but they differed from females in that they peaked later, in either December (N = 3) or November (N = 1). The lighter individuals of each species, M64 and M55, were both young and were apparently still growing to adult weight, but M73 was an old adult and M63 was mature, so their weight increases were due to improved body condition, not net growth. Males also showed a slight tendency to lose weight sometime between March and July.
If the body mass phenologies are compared with fruitfall and rainfall phenology (fig. 10.4), the patterns seem to show the closest similarity to rainfall, rather than to crude fruitfall, but treeshrews in general had the highest body masses during the peak fruiting period of September– December 1991. Too few individuals are represented to give more than a general idea of trends.
PERSISTENCE (SURVIVORSHIP)
OF INDIVIDUALS ON THE STUDY AREA
Treeshrews are for the most part highly trappable, and all individuals of the four terrestrial species studied were initially easily caught; but in any month some animals escaped capture and were not monitored (see chapter appendix). In general, I only assumed that an adult, territorial resident treeshrew was gone from the area when another adult was captured
For all three species, the population of resident adults was completely stable from September to December 1990, but there was a changeover of residents between December and March and into June (figs. 10.6–10.8). In January-February I was absent and could not record the fates of the animals.
TUPAIA GRACILIS and T . MINOR There was only one adult resident slender treeshrew on the study area at Danum at the beginning of the study and two at the end of it. No individual remained on the area throughout the study, but F67, who was displaced from the study area in March, after ten months, by a new adult female, perhaps was still alive. The population thus increased while two females shared the area formerly used by one (fig. 10.8). The adult male (Mfoot) took up residence in March and disappeared in August, after six months, and a new, barely adult male was captured in September (M191). None of the four subadults was captured a second month.
Although too few individuals were followed for much inference, at least one T. minor (F70) remained a resident adult on her territory for the eleven months of the study at Danum. This shows that the smallest treeshrews can be long-lived.
TUPAIA LONGIPES At Danum Valley persistence of resident adult T. longipes was high: three (perhaps four) of the five adults trapped in September 1990 were still present at the end of the study in August-September 1991 (60–80% survival). One old male (M73) with much tooth wear in September 1990 disappeared after December and by June was replaced by a young male with little tooth wear (M287) (fig. 10.6). One of the resident females lost half her territory in March-April to a young female born on the study area in September (see chap. 9). Young of the year thus appeared to squeeze or replace two of the older residents. This division/compression of female territories increased the total population. Two territorial females and one male (F56, F84, and M64) survived the entire study, and the other female (F66) was not replaced and may have been still alive but trap-shy (as was M64 at the end). At Poring the two resident females survived the entire study, but the male was not recaptured at the end and his status was unknown. In this species alone, I caught ancient individuals with teeth worn nearly to the gums (one of each sex), suggesting frequent high survivorship.
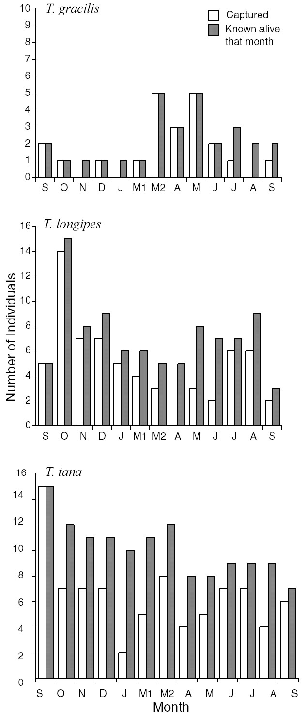
Fig. 10.8. Number of treeshrews captured (open bars) and number known alive (shaded bars) each month at Danum Valley.
TUPAIA TANA Of the nine resident large treeshrews captured in September 1990, only two males (M62, M77) are known to have survived the entire year; one of these (M77) had apparently been pushed out of part of his original territory and was no longer within the trappable sector. One female (F54) was not replaced by another on her territory, so she may have been alive but trap-shy (she was injured in a trap). Survivorship for the year was thus 22 to 33 percent. There was a large turnover of T. tana in December-March (fig. 10.7). Most animals simply disappeared and were replaced by others, but one male (M55) was evidently killed by a predator. Two females and two males were replaced by animals born on the study area, all from the August-September generation (fig. 10.7), and two other new males and a female (M167, M168, and F297) were also of this age, as judged from tooth wear.
Langham (1982) collected excellent demographic data during his three-year study of T. glis. His study population had a one-year survivorship of 33 percent for males and 40 percent for females and a twoyear survivorship of about 10 percent and 12 percent respectively. I followed too few animals for much inference, but my study populations of T. longipes seemed to have higher adult persistence than Langham's, and T. tana persistence was similar or slightly lower than his, based on the standing estimated age distribution from tooth wear at the beginning of the study (see chapter appendix).
Langham found that most adult T. glis disappeared in three periods: in December-February, June-July, and October-November. This is echoed by the strong seasonality of territorial turnover among animals at Danum. Individuals of three species (T. tana, T. gracilis, and T. longipes) were displaced by other individuals from all or parts of their home ranges. Treeshrews that disappeared from territories were therefore sometimes still alive. I conjecture that the cause of the seasonality in territorial replacement at Danum was the simultaneous growth to adulthood of a super cohort of young born in August-September. Because of the fruit masting, these may have been super-young: unusually fast growing and large, which succeeded in challenging and ousting older, established residents.
In summary, the three terrestrial species all shared some similar life history patterns: (1) breeding in two main periods that followed peak fruitfalls, August-November and March-June; (2) a shift in territory ownership (population turnover) in January-May, resulting in a higher density of resident individuals and slightly smaller territories; (3) slow final maturation of young, so that young of the year that moved into territories
DISCUSSION OF LIFE HISTORY
All fieldwork on treeshrews thus shows that the explosive reproductive potential from continual breeding, rapid growth, and early sexual maturity seen in laboratory treeshrews is not realized in the wild, where discontinuous breeding, slow growth, and late maturation are more typical. Because there is no evidence for genetic control of seasonal breeding, I agree with Langham's hypothesis (1982) that in nature treeshrew growth and reproduction are nutrient limited. Langham speculated, “The delay in attaining sexual maturity was probably a result of a decrease in food supply. As most T. glis were born in the period March to August, they would be growing to adult weight during the period of the north-east monsoon, associated with heavy rain and lowered food supply” (p. 331). And further: “Breeding was timed so that births tended to coincide with the onset of the south-west monsoon with its moderate rainfall, leading to an increase in vegetative growth and a presumed increase in insect abundance” (p. 341). In Sabah young likewise sometimes emerged when food supplies were at minima.
Langham believed that arthropod abundance was the food variable limiting treeshrew breeding. Soil arthropod abundance in his region was highest in April to August and lowest in November (Langham 1982). This does not correlate well with peak breeding of T. glis in February to June, but the sector of arthropods sampled was not the same as those eaten by Tupaia. My results point to fruitfall as the proximal factor controlling reproduction, and especially reproductive success, but there are insufficient data on arthropod abundance in Sabah to evaluate its possible role, which is likely an additive one including both fruit and invertebrates.
Several features of treeshrew behavior in Sabah support the conclusion that nutrient levels limit reproduction. The long distances of daily movements, dawn-to-dusk activity, and large home ranges of Tupaia species directly imply that these treeshrews need the entire available day and a large piece of the landscape to meet their daily food needs. The concentration of activity at fruit trees and the shortened daily ranges of treeshrews using them (table 8.4; chap. 5) indicates that the animals use fruit when they can, even to the point of overstepping territorial boundaries (Kawamichi and Kawamichi 1979; see also chap. 9). Arthropods are hunted one by one, by searching, searching, and more searching,
Field data may not discriminate the relative importance of individual food types for reproduction. Seasonal abundances and/or availability of fruitfall, arthropods, and earthworms could be synchronized, as all are keyed to rainfall. If breeding is facultatively triggered by a nutrient and/or energy threshold, then any combination of food items could suffice, such as a mix of high invertebrate prey levels and some fruit, or vice versa. The one-month phase difference in breeding of T. tana and T. longipes in September-October 1991 suggests either that a nutrient advantage for T. tana occurred earlier or that T. longipes had a larger energy deficit to overcome before breeding. Because large treeshrews have smaller daily movements and home ranges than plain treeshrews, they have energetically easier lives, which may translate into a lower nutrient threshold to overcome before breeding. Tupaia tana females were active for an average of 1.25 h longer per day than males (see chap. 7). This could reflect a greater effort by females to acquire resources for reproduction yet energy acquisition easy enough for males to be able to retire early. Such a differential was not evident in other Tupaia species, where both sexes were active for nearly 12 hours a day, suggesting a more difficult fundamental regime (see table 7.1).
Year-round physiological readiness would permit treeshrews to breed rapidly whenever food becomes abundant and to remain close to carrying
the absentee system
The absentee maternal system requires for its success a whole suite of behavioral, physical, and physiological adaptations that are highly specific to it. These include (1) highly specialized visiting behavior by the mother; (2) storage and instant release of forty-eight hours' worth of milk by her mammary glands; (3) intense, virtually instantaneous suckling behavior by the nursling; (4) storage of the milk in a giant stomach; (5) thermoregulation by newborns; (6) the ability of nestlings to maintain rapid growth and resist dehydration; and (7) energy-minimizing juvenile behavior, such as absence of exploratory behavior. The absentee nursing schedule must tightly constrain litter size. To succeed in weaning more than two young, the mother would probably have to increase the rate of feeding to at least daily. It was certain that this system had to occur in the wild, because captive treeshrews rear young brilliantly with it.
The absentee maternal care that we documented in T. tana F109 corroborated in detail the behavior seen byMartin (1968) in captivity. This includes an “extra” nursing visit on the day before emergence, which itself was on a normal feeding date. I surmise that this one extra feeding gave the young enough surplus energy to awake from their usual torpor and venture out into the world. The feeding supplement itself may have stimulated a change of activity pattern.
The most unexpected and novel of our findings on T. tana maternal
Tiny pentail young shared the den of the mother at about the time of their emergence, and evidently continued to do so for months, probably until after the next litter was born, as we saw adults, subadults, and juveniles emerge from the den together. This, coupled with other evidence that pentail groups share a nest (see chap. 9), makes it almost certain that pentail treeshrews do not have an absentee maternal care system. If this most primitive treeshrew has a “normal” nidicolous system wherein mother and young nest together from birth, then it is probable that the absentee system of Tupaia is a derived condition in the subfamily Tupaiinae. If ancestral tupaiines switched to an absentee system, what might have provoked the change?
When he described the maternal care system of Tupaia,Martin (1968) speculated at length about its evolution and possible function. He thought that only two hypotheses were likely: (1) that it functions to protect young from predation and (2) that it has no specific contemporary function but is a relict, primitive system that is carried by treeshrews as evolutionary baggage. The two hypotheses are not mutually exclusive, but Martin favored the second hypothesis and rejected the first, because he believed that treeshrew nursery nests were more likely to attract predators
| Force acts on | Selective Force | |
|---|---|---|
| Predation | Energetics | |
| Young | Seals | Seals (energy storage) |
| Tupaiids | Tupaiids | |
| Lagomorphs | ||
| Agoutis | ||
| Artiodactyla | ||
| Elephant shrews? | ||
| Mother | Seals | |
| Tupaiids? | ||
An approach to this question is to explore whether tupaiines share common features with other species that have absentee or similar maternal care systems (table 10.5). Mammals with absentee-like maternal care systems in which the young rest apart from the mother, who visits them for nursing, include taxa as diverse as elephant shrews, hares, pikas, maras, agoutis, most deer, many antelopes, and some seals. Apart from the seals, most of these are both cursorial and have precocious young (Emmons and Biun 1991), and this includes elephant shrews and many lagomorphs. The connection between these features is obvious: cursorial mammals have long legs and modified feet and rely on rapid getaway for survival from predators. Their limbs are poorly suited for escaping within narrow burrows or nests, or climbing anything, and they generally rest in sites from which cursorial escape is possible. Their young must either be precocious enough to outrun predators shortly after birth, as in the case of horses, caribou, or wildebeest, or they must be hidden distant from the mother, to which predators are attracted, as in most small deer and antelopes. Small cursorial species whose legs are unsuited to burrowing must hide their young in the open, but saltatorial species that jump from the hind legs, so that the forelegs are less modified for running and can be used for digging, such as maras, agoutis, and some rabbits, can dig a burrow for their young but do not use it themselves, as they are probably safer outside it. Elephant shrews use their forelegs for
A type of absentee maternal behavior is exclusive to the seals and sea lions, especially the eared seals (Otariidae; the following from a review in Oftedal, Boness, and Tedman 1987). These give birth on land in protected sites (usually islands), where the mother leaves the young while she goes on extended foraging trips that can last for more than ten days but usually two to three. When she returns she may stay with the young and nurse it for a day or two before leaving again. Lactation is prolonged in these species (6–12 months). In phocid seals, which place their young on pack ice, the mother stays with the young and does little or no foraging until the young is weaned after a short lactation period (4–30 days). The absentee pinniped lactation systems are driven proximately by energetics because young on land or ice are often so far from the foraging grounds that several days are needed for the mother to forage. Ultimately, they may be driven by predation avoidance, which favors the use of safe, remote nurseries for the young.
Most terrestrial species with absentee-like maternal care are diurnal, or active crepuscularly or both day and night. This may imply that visual detection by predators of mothers with their young is a more important problem for diurnal than for nocturnal species. Diurnality is quite rare among small mammals, and it may be no coincidence that elephant shrews, tupaiines, and pikas share both diurnal activity and absentee maternal care. By day predators may be more visual and able to spot maternal visits to young from a greater distance. This could select for reduced nursing visits.
Tupaia species differ from all other absentee mothers in that the adults use nests, at least one species is arboreal, and the young are altricial. Because hidden-young systems have evolved independently many times as a protection against predators, it is also likely to have this function in treeshrews, despite the different ecomorphology. A reason that predators would be less likely to find young in absentee nursery nests is that adult treeshrews have a very strong musky odor (especially their urine). When we removed them for examination, I sniffed both the T. tana nursery nest and the young. Neither had any perceptible odor, and they were clean
The absentee system of Tupaia could also confer an energetic advantage compared to “normal” nidicolous systems. Energy would be saved by the mother avoiding the commute several times a day from her foraging place to the nestlings, and more would be saved by the extreme immobility of the young nestlings, which because of their forty-eighthour fasts must hoard the energy from each feeding. The whole system is perhaps more energy efficient, which could be critical in these energylimited species.
Some hypotheses about the systematic place of treeshrews put them into the same order with elephant shrews, or rabbits and pikas (reviewed in Luckett 1980). The morphological basis for these ideas is weak (Luckett 1980), but it is curious that these divergent (from other mammalian families) and presumably old orders share with treeshrews an absentee maternal care system (Broekhuizen and Maaskamp 1976; Emmons and Biun 1991; Rathbun 1979; Smith and Ivins 1983). Is this evidence for an extremely early origin of this system in some remote common ancestor of these families? A brief review of what is known of the most primitive living mammals does not suggest that these have absentee or any less intense or more rudimentary maternal care than do members of more recent, “advanced” families (Emmons and Biun 1991): even platypus mothers stay with their young in the same nest for many weeks (Grant 1983).
APPENDIX: METHODS
Maternal Care. I knew that finding nests with young was going to be most difficult, because locating a single spot visited by an animal for less than five minutes during two twelve-hour days of activity, by means of radio-location alone, is equivalent to finding the proverbial needle in a haystack. At the outset it was not even known where most of the species nested, or what nests of any species looked like. My method for looking for nursery nests was straightforward: I would follow a lactating, radio-collared female for four days, then map her paths and look for places she went in the early morning on alternate days, or each day. If I could pinpoint a likely spot, I would track her to see if she went there on schedule on an additional day. Before daylight on the next appropriate day, I would try to hide in a place with a view of the surroundings and monitor the signal through earphones as the female approached. Despite much effort, this never worked. It was a time-intensive effort, because it took about ten days for each attempt. The second year of the study, I tried another method: because females
To observe the treeshrew nursery nest that we found at Poring, we built an elevated platform in a tree, hidden by a blind of palm leaves on all sides (Emmons and Biun 1991). One of us would take up position in the blind well before dawn and monitor the radio signal (with earphones) from the mother, whose own nest was 70 m away. When she approached, we turned off the receiver and waited motionless until after she had visited and moved well away. We videotaped her arrival on one morning. During an “off” day, when the young were not nursed, we climbed up and briefly removed them for measurement and to mark them with ear tags and describe the nest. At that time we estimate that they were eighteen days old. One was male and the other female, and their measurements were as follows: total length = 206 mm; tail = 82 mm; hind foot = 37 mm; ear = 10 mm; mass = 54, 55 g.
Life History. Life history data were derived largely from monthly capture records. At first capture each month, all treeshrews were weighed, and females were palpated for the presence and number of embryos and examined for evidence of lactation. To avoid physical handling, I did not measure them except at first capture or if they were anesthetized for fitting with a radio-collar. Ideally, monthly trapping would be scheduled so as to capture almost all of the resident animals, but in reality it was a compromise where several factors needed to be juggled. Tupaia species often became trap-happy. Some animals were caught twice daily and consequently spent almost the whole day in traps. To decrease the likelihood of injuries to these, and minimize disruption of their lives, monthly trapping was limited to two days. This was too brief to capture every treeshrew. In contrast, a few individuals, especially T. longipes, eventually became trap-shy after multiple bouts of handling, so that when their radios failed, I usually lost track of them, although some were seen from time to time so I knew they were still on their territories. Monthly records were therefore incomplete.
Capture Effects. Of great concern when doing research in which animals are submitted to trapping, handling, anesthesia, and carrying radio-collars is whether the procedures of the study cause stress or mortality that change the life histories of the animals. One way to evaluate whether the population is in a “normal” state after a year of study is to compare its age structure at the beginning of the study, when animals were first trapped, to that at its end. This can be done roughly, for T. longipes and T. tana, from notes on tooth wear. In September 1990, of six adult T. longipes caught, four were old, with very worn teeth, and two were just entering adulthood. At the end of the study at least three (perhaps four) plain treeshrews on the area were very old, and two were young of the year. Tupaia tana residents on the study area in September 1990 included five older animals