5. Diet and Foraging
Behavior
Its diet is the most fundamental ecological attribute of an animal. Virtually every feature of a species' biology, from morphology to life history, is linked to what it eats and where, when and how nutrition is acquired. The diet of treeshrews was from earliest reports correctly known to consist of fruit and insects (Cantor 1846). However, this knowledge alone does not distinguish treeshrews from other rainforest mammals, almost all of which also eat fruit and insects (Emmons 1995), nor does it define the ecology of individual species. Below I describe the nature of fruit- and insect-eating by treeshrews, along with the behavioral characteristics of foraging for each food type. I discuss how the diet is related to morphology and review the possible competitive interactions that might occur for different food types both among treeshrew species and among treeshrews and other vertebrates in their community. With this information we can start to approach the question, What is a treeshrew?
FRUGIVORY
CHARACTERISTICS OF FRUIT SPECIES EATEN
As soon as we began to follow treeshrews by radio-tracking at Poring, we discovered that they often spent much of their day at fruit trees (Emmons 1991). I was able to watch lesser treeshrews feeding on fruit, sometimes for hours, but for other species fruit tree use was inferred largely
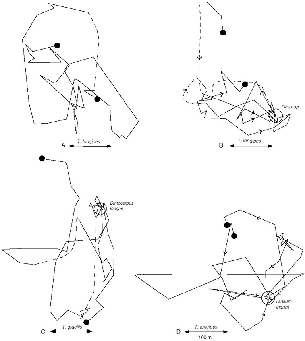
Fig. 5.1. Influence of fruit trees on treeshrew movements. A and B, movements of T. longipes female F86 on sequential days. A, without a known fruit tree (26 Mar. 1991, 44 locations, 2,137 m traveled); and B, feeding at a small fig (Ficus) tree (circle), to which she returns five times (27 Mar., 48 locations, 2,189 m traveled). C, movements of T. gracilis female F67 when feeding on fruit of a mata kuching (Dimocarpus longan), which she visits twice (26 Sept. 1990, 54 locations, 2,240 m traveled). D, movements of T. longipes male M64 when foraging at a tree of Dialium indum, which he visits four times (1 Dec. 1990, 41 locations, 2,512 m traveled). Dashed lines represent approximate routes where radio-tracking data were imprecise.
| Fruit Species | Site | Treeshrew | Size (cm) | Color |
|---|---|---|---|---|
| Alangiaceae | ||||
| Alangium ebenaceum | D | Tg, Tl, Tt | 1.5×2.0 | green |
| Annonaceae | ||||
| Polyalthia sumatrana | D | Tl.Tt | 2.8×1.2 | purple |
| Grossulariaceae | ||||
| Polyosoma integrifolia? | D | Tl, Tt | 2.5×1.0 | green |
| Leguminosae | ||||
| Dialium indum | D | Tl, Tt | 2.7×2.0 | brown |
| Meliaceae | ||||
| Lansium domesticum | D | Tl | 6.5 | yellow |
| Moraceae | ||||
| Ficus cf. subulata iv-2 | P | Tl, Tg | 2.0 | orange/yellow |
| Ficus cf. sundaica ii-7 | P | Tmi | 1.5 | red/orange |
| Ficus cf. sinuata or parietalis A660 | P | Tl | 0.8 | |
| Ficus cf. sumatrana A500 P | P | Tg | 0.6 | |
| Ficus cf. benjamina C-60 | P | Tl, Tg.Tm | 0.8 | red/orange |
| Ficus sp. C-40 | P | Tl | 0.5×0.7 | |
| Ficus sp. iii-150 | P | Tl | 1.5 | yellow |
| Ficus sp. A N1W250 | D | Tl | 2.5×1.8 | |
| Ficus sp. Tiny vine N0W025 | D | Tl, Tt, Tm | 1.0 | red |
| Ficus sp. B N2W225 | D | Tl, Tg | 1.4×1.2 | |
| Myrsinaceae | ||||
| Sp. | P | Tmi | 0.5 | red/purple |
| Rafflesiaceae | ||||
| Rafflesia keithii | P | Tt | 1.4 | brown |
| Sapindaceae | ||||
| Dimocarpus longan | D | Tt, Tl, Tg | 1.5 | brown |
| Sp. | P | Tmo | 0.6 | orange aril |
| Sapotaceae | ||||
| Payena acuminata | D | Tt | 3.4×1.8 | green |
| Cf. Madhuca sp. | P | Tt, Tl | 3.4×1.4 | green |
| Vitaceae | ||||
| Parthenocissus sp. | D | Tmi | 1.8×2.0 | yellow |
At Poring figs (Ficus spp.) so completely dominated the list of fruits eaten by treeshrews (64%) that tupaiids might have been thought to be fig specialists. However, the next year at Danum figs were only 27 percent of species known to be eaten, and it was clear that fruiting fig trees were simply much more numerous on the study area at Poring than at Danum Valley, where other fruits were eaten more often. In both areas suitable fig trees commanded intense interest from all species of treeshrews, in terms of both hours spent at them and the number of daily visits, so that figs can be said to be among the most preferred fruit (see chap. 8 for data on time spent at fruit trees). Some other fruits elicited only brief visits, perhaps because fruit fell sporadically and little was available at one time or because the animals did not much like them.
Typical fruits eaten by treeshrews were small, 0.5 to 2.0 cm in smallest dimension (table 5.1, fig. 5.2), with soft edible parts (mesocarp, aril, or syconium), and ranged from juicy to dry and to astringent, sweet (most), or quite tasteless (to me). About half were red, yellow, or orange, colors favored by monkeys and birds; two were purple, as favored by birds; and the rest were green or brown, typical of nonprimate mammal dispersed species (Gautier-Hion et al. 1985). Treeshrew fruits thus varied in probable disperser targets.
The sizes of fruit species eaten by treeshrews mirror the most common size classes in the environment (fig. 5.2) but exclude the larger sizes. The single very large fruit identified as eaten was that of the parasitic plant Rafflesia keithii. This large, sessile, indehiscent, terrestrial fruit grows from an underground stem and has a soft, oily, strong-smelling, custardlike pulp filled with thousands of tiny seeds. We observed T. tana and a squirrel (Callosciurus notatus) bite off the pulp of an opened fruit over the course of several hours (Emmons, Nais, and Biun 1991). This strange fruit, with its odd dispersal mechanism, was the only fruit species identified for which treeshrews (with squirrels) are likely to be principal dispersers.
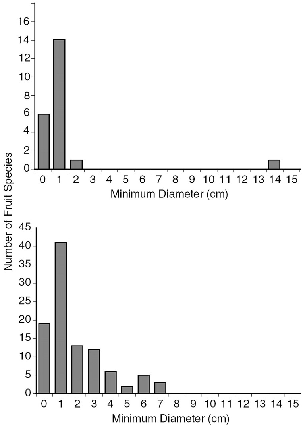
Fig. 5.2. Size of fruits eaten by Tupaia species in both study areas (top) and size of all fruits collected in transects at Danum Valley (bottom).
All Tupaia species ate fruit and were readily attracted to ripe bananas, on which they gorged themselves when they could, but the other two genera of Bornean treeshrews appeared to be less frugivorous. The smooth-tailed treeshrew, Dendrogale melanura, a montane species, cannot be captured in traps baited with fruit (Alim Biun pers. com.). Pentail treeshrews likewise showed scarcely any interest in banana bait, but they did appear to be slightly frugivorous. The sparse evidence for pentail frugivory is summarized below.
Pentails were common on the Danum Valley study area (see chap. 8), but they comprised only 5 of 420 treeshrew captures during the project. Once caught, they ate the banana in the trap, and two of only three scats of this species that were collected from traps for dietary analysis included the remains of fruits other than bait. Captives are said to readily eat fruits (Gould 1978; Lim 1967). During 199.95 h of active foraging by all pentails followed by radio-tracking, P. lowii F163 spent 40 min in a tree that supported a fruiting liana of an Annonaceae (cf. Artobotrys sp.), from which fruit fragments were falling; but this tree and liana were also popular with palm civets (Paradoxurus hermaphroditus), and I could not see into the fruiting canopy to determine what was eating the fruit. During a torrential rain, F163 spent 2.6 h in a giant fruiting fig next to her nest tree, but again her activities there could not be seen. On another night pentail F96 visited a fruiting Dialium indum and also a Polyalthia sumatrana, both highly favored fruits of Tupaia, but she did not linger in either. The degree of frugivory in wild pentails is thus unclear, but they seemed to eat fruit at least sporadically, perhaps often, although without sufficient attraction to be lured by it into traps or to reveal distinctive fruit-feeding behavior during radiotelemetry.
The three terrestrial lowland Tupaia species overlapped broadly in the fruit species they were known to eat (see table 5.1). When Tupaia of different species were simultaneously radio-collared on the same terrain, they usually visited the same fruit trees. T. minor alone was recorded at certain fruits, but conditions of observation usually did not permit viewing the ground under these, and my presence watching the tree would likely have inhibited visits by the sharp-eyed ground-foraging species. Further study is needed to establish whether differences in fruit preference exist among Tupaia, but my observations thus far indicate that such differences are not likely.
| First Marker | Last Marker | |||
|---|---|---|---|---|
| Treeshrew (N) | Mean | Rang | Mean | Rang |
| NOTE: At the start of the day's active period, treeshrews were fed one bite of banana containing marker dye, followed by their normal diet ad lib. Times are to first and last appearance of marker dye in the feces. Last appearance includes only strongly marked material; faint traces of dye persist beyond that time. N = number of independent trials. (Emmons 1991; experimental details given therein.) | ||||
| T. minor (7, 4) | 20 ± 5 | 13–29 | 45 ± 7 | 38–54 |
| T. tana (5) | 57 ± 13 | 38–73 | 163 ± 52 | 118–248 |
FRUIT FORAGING STRATEGY
FRUIT PROCESSING : TRANSIT TIME When I first captured Tupaia species, I noticed that individuals who had been only a short time in a trap were already defecating the banana bait. This led me to carry out some experiments between the two field seasons, at the National Zoological Park in Washington, D.C., to measure the passage times of fruit eaten by T. minor and T. tana (Emmons 1991). These were followed by more detailed experiments of both passage time and assimilation efficiency of insects (Roberts et al. pers. com.). These tests showed that, for fruit, Tupaia have among the most rapid transit times known in mammals, equivalent to those of fruit bats (table 5.2; Emmons 1991). The shortest passage time in seven trials on T. minor was only 13 min between eating dye-marked fruit and first defecation of a marker, and that of T. tana was 38 min.
Transit time within a taxon of mammals increases with body size, because it is evidently a function of the absolute, rather than relative, length of intestine that the food must traverse (Chivers and Hladik 1980). Predictably, then, because it is a larger animal, T. tana has a longer transit time than T. minor (table 5.2). The simple intestinal gross morphology of treeshrews is very similar to that of fruit bats (chap. 2) and is doubtless causally related to the short fruit passage times of both groups.
FRUIT PROCESSING : FEEDING BEHAVIOR Shortly before I left Poring I experienced one of the most exciting moments of discovery that year, for it afforded a completely new insight into treeshrew ecology.
20/8/89; 0928 (seen from a blind): A T. tana sits on a fallen branch on the ground, eating a green fruit [Sapotaceae: Payena or Madhuca sp.], holding it

Fig. 5.3 Fruit of Sapotaceae (cf. payena) and wads of fiber spit out by T. longipes on 20August 1989.
This fiber-spitting behavior is well known for both New World (Microchiroptera; Phyllostomidae) and Old World (Megachiroptera) frugivorous bats. I later saw this behavior of ejecting indigestible seeds and parts of fruits in the captive colony of T. tana at the National Zoo (Emmons 1991), but from watching these animals it was unclear to what extent large treeshrews masticate and suck fruit tissue before spitting out unwanted pieces. However, there is independent evidence from a study of masticatory movements in T. glis(Fish and Mendel 1982) that when eating fruit this species presses the food against the palate, which is concave and deeply ridged (Martin 1968), with the same tongue movement pattern used by flying foxes, which also have deeply ridged, concave palates. Also like fruit-eating bats, treeshrews salivate abundantly. Copious saliva may aid in the rapid extraction of soluble nutrients from masticated fruit. This whole set of behaviors is thus just like those found
Treeshrews of the genus Tupaia have a suite of morphological and behavioral traits associated with fruit processing, which together result in ingestion of only the most quickly digestible fractions of fruits, which pass rapidly through the gut. It is unlikely that any digestion or fermentation of structural plant carbohydrates can occur, as there is neither enough time during passage nor any digestive tract site for it. This implies that fruit contributes only its most readily assimilated nutrients to treeshrew diets. It is not known whether Ptilocercus or other treeshrew genera share these traits.
TEMPORAL PATTERN OF FRUIT-EATING
The physiological behavior outlined above relates to the temporal patterns of fruit-eating observed in the field.
0745: Tupaia minor F112 is foraging in a fruit tree [Myrsinaceae]. She eats a ripe, red fruit, holding it in her paws, then leaves tree at 0746…. 0816: She goes rapidly straight back to the fruit tree, arriving at 0823. 0827–0847: She rests, crouched on a branch. 0847–0858: She forages, eating fruit. 0900: She goes back and rests on the same spot. 0910: She grooms several parts of body and tail, then curls up and rests again. 0913: She gets up and forages, then leaves the tree at 0926.
The alternation of short feeding bouts with short resting bouts was characteristic of T. minor whenever I watched them feeding on any species of fruit. The interval between feeding bouts approximates the transit time of fruit through the gut of lesser treeshrews (see table 5.2), so that it seems that the animals are filling up with fruit repeatedly, as often and quickly as it can be transited through the digestive system.
At Danum Valley a huge mata kuching tree (Dimocarpus longan, a delicious, juicy fruit much exploited by people as well as by most wild frugivores) massively fruited within the ranges of radio-collared treeshrews of three species. To quantify visits to a major fruit source, on 28 and 29 September 1990, I monitored this tree continuously from afar, with a radio receiver, from before dawn until heavy rains stopped all activity in late afternoon. I also followed one of the treeshrews (T. gracilis F67) continuously from dawn to dusk during four other days of the same week. The results show that like lesser treeshrews these three other
| Treeshrew | Date | No. of Visits | Hours at tree | |
|---|---|---|---|---|
| NOTE: The T. tana and T. longipes were monitored only on 28–29 September 1990. The T. gracilis was followed continuously on the four other days. | ||||
| T. tana | ||||
| F65 | 28 September | 3 | 1.60 | |
| 29 September | 0 | 0 | ||
| T. longipes | ||||
| F56 | 28 September | 2 | 0.67 | |
| 29 September | 6 | 1.15 | ||
| T. gracilis | ||||
| F67 | 26 September | 2 | 2.60 | |
| 28 September | 3 | 1.03 | ||
| 29 September | 5 | 0.72 | ||
| 1 October | 4 | 0.48 | ||
| 2 October | 3 | 4.84 | ||
| 3 October | 2 | 0.93 | ||
| Total | 30 | |||
Although I was unable to see them eating, continuous radio-tracking showed that the pattern in which the three terrestrial species visited fruit trees was similar to that of lesser treeshrews I could watch, and I surmise that they feed similarly in bouts, perhaps with longer intervals between meals, commensurate with their longer passage times. Many other radiotracking episodes, and feeding behavior in captivity, support this view.
SEASONAL FRUIT ABUNDANCE
The amount of fruitfall registered in our monthly transects on the study plot at Danum (fig. 5.4; see also chap. 3) showed large seasonal variation. The number of fruit species recorded per month varied by a factor of nearly eight; however, the amplitude during that year was dominated
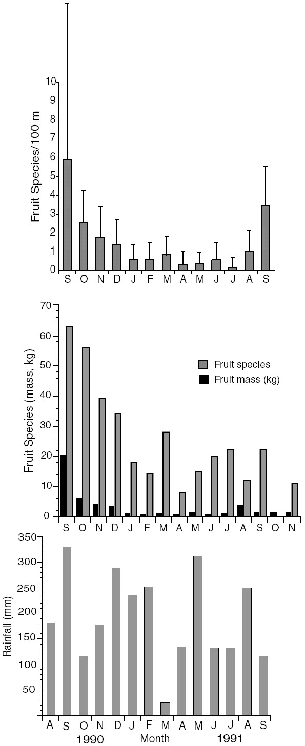
Fig. 5.4. Mean number and standard deviation of fruit species collected monthly per 100 m of trail on the Danum Valley study area (top); fruit species (light shading) and total weight of fruit (dark shading) collected monthly on a transect of 4.5 km of trails (middle); and rainfall at the research center during those months (bottom). There was no fruit collection in October 1991.
| Species | A | S | O | N | D | J | F | M | A | M | J | J | A |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NOTE: No observations were made in January and February, and full-time radio-tracking, when best observations were made, began 17 September 1990. | |||||||||||||
| Payena acuminata | X | ||||||||||||
| Dimocarpus longan | X | X | |||||||||||
| Parthenocissus sp. | X | ||||||||||||
| Lansium domesticum | X | ||||||||||||
| Polyosoma integrifolia? | X | ||||||||||||
| Polyalthia sumatrana | X | X | |||||||||||
| Dialium indum | X | ||||||||||||
| Ficus sp. tiny vine | X | ||||||||||||
| Ficus sp. | X | ||||||||||||
| Alangium ebenaceum | X | X | |||||||||||
| Ficus sp. N250W2 | X | ||||||||||||
| Ficus sp. N1W250 | X | ||||||||||||
A summary of months in which fruit species were recorded as eaten by treeshrews at Danum (table 5.4) shows that only in October of the masting peak was more than one or two species registered; I saw no treeshrews eating fruits in May or June. During particular months at Danum Valley, certain fruit species seemed to dominate the interest of treeshrews, to the point of being apparent “keystone” resources. These dominant fruit species included Payena acuminata (or similar Sapotaceae) in August; Polyalthia sumatrana in October and November; Dialium indum in December; and Alangium ebenaceum in July and August. At Poring species of Ficus as a group were the major and almost only dominant fruits in
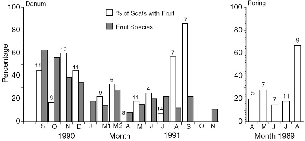
Fig. 5.5. The percentage of treeshrew scats (all species) collected on the Danum study area that contained fruit other than bait, by month (left figure). Total 104 samples, N for each month above the bar, no data for January, 0 percent fruit in April; February set collected in early March as in figure; compared with the total number of fruit species collected each month in the fruit transect. The percentage of scat samples with fruit per month at Poring, total 39 samples, including T. montana (right figure).
The general annual pattern of fruitfall (fig. 5.4) will only reflect the fruit resource available to treeshrews if the species they eat have a phenology resembling that of the forest as a whole. To evaluate this, the overall presence/absence of fruit (other than bait) in 143 scat samples collected throughout the study can be compared to the general phenology (fig. 5.5). Too few samples are available to analyze the data for single treeshrew species, but since all appeared to eat the same fruits, it seems legitimate to combine them. Considering the small monthly sample sizes, the percentage of treeshrews that had eaten fruit shows a surprisingly exact, almost perfect correspondence with the number of species collected on the transect from September 1990 to June 1991, with only October 1990 departing from the trend. Particularly notable is the complete absence of fruit in scats during the month of minimum fruitfall (April). However, the final two months of the study (August and September) show a sharp deviation from concordance, with fruit eaten often during
INSECTIVORY
FORAGING BEHAVIOR
Each treeshrew species had a distinctive way of searching for invertebrate prey. Olfaction was clearly dominant in many cases of searching and prey capture, but vision was evident in others.
PTILOCERCUS LOWII The most characteristic hunting behavior of pentails was exploring the surfaces of tree trunks, which could be bare but more often had attached lianas. The trunks on which they foraged ranged from slender understory saplings to the largest boles of emergent giants. Pentails also searched up and down branches or liana stems, and I once saw one repeatedly hunting up and down in a hanging mass of thin dead twigs in a dead, leafless vine festoon. One searched the surface of a dead, rotten, standing stump, thoroughly investigating inside an empty woodpecker hole (to which I later climbed to examine). These treeshrews were not often seen in leafy foliage, but when they were, they were in the tops of treelets in the understory or subcanopy. It was not possible to watch pentails for long, or without some disturbance from the light, because they moved rapidly and often avoided the headlight beam (although some individuals became quite habituated). Also, they could not be seen when they were in the canopy. Within these limitations, I would define P. lowii as a forager on arboreal woody surfaces— the bark of trunks, stems, and branches—with less attention to the foliage of treelets. A species-typical behavior was to hunt up and down the same tree trunk several times, before scampering quickly on to the next one.
TUPAIA MINOR Lesser treeshrews hunted arthropods by moving steadily through the vegetation, investigating every likely insect hiding place. Favored search sites included hanging masses of dead leaves, rolled dead leaves, dead twigs, holes in dead wood, crevices between stems and
TUPAIA GRACILIS Slender treeshrews also hunted by sniffing and looking. I most often saw them working along at ground level, on the ground surface, on fallen logs, roots, or rotten wood, but they typically searched the living leaves of the undergrowth plants, peering or nosing up to the undersurface of foliage above them, or climbing through the understory shrubs or vines.
T. gracilis F67 seen insect foraging in vines 1–6 m, she disturbed an insect that dropped down and she raced down the vine after it to the ground. A few seconds later I saw her eating a large insect.
A T. gracilis foraging in the lower 2 m of aboveground, dense vines, eats a large, hairy caterpillar, climbing to 1.5 m on a slanting stem to eat. She rubs it once on the branch, holding it in the mouth.
I saw slender treeshrews pick up prey from the ground surface, as well as from shrubs and a rotten stump, and I saw them sniff dead leaves. Most of the few prey items I saw were large (but large items are easier to see than small ones): one, “almost as large as she (F67), a huge caterpillar or katydid.” Slender treeshrews are surface gleaners of the ground and understory plants, with rare forays up to the lower canopy.
TUPAIA MONTANA Too short a time was spent in the habitat of this species to characterize well its hunting behavior, but both sightings and
TUPAIA LONGIPES The predominant foraging pattern of plain treeshrews was to travel quite quickly along, nose to the substrate: “A T. longipes forages rapidly along the ground, nibbling at things on the litter surface” (7 November 1990). I was not able to identify any prey item, as these treeshrews generally picked up small items and continued to travel as they chewed. The most common substrates were the ground and rotten logs, but they often climbed around, to the height of about a meter, in dense, bare snarls of liana stems in the dark recesses below blankets of climbers, or walked along the larger horizontal lianas of such places. Rarely, plain treeshrews foraged on the broad sides of large logs or trunks of live trees or higher in vines (see table 4.1). This species was a surface gleaner of its substrate. I did not see it foraging in foliage or digging in the litter.
TUPAIA TANA Unlike the other two lowland terrestrial species, large treeshrews typically foraged for items hidden under the surface litter of the decomposing organic layer.
Tupaia tana F58 came quietly sniffing along a liana on the ground, then nosed along the litter. She dug a hole in the litter, alternately digging and sticking her nose in and sniffing. She dug only into the surface layer of a place with a thick leaf mat [5 November 1990].
Large treeshrews would walk very slowly along, periodically pushing the nose into the litter and digging when they discovered a morsel of interest. Mesic sites, such as streamsides, dark tunnels under dense vinefalls, wet, rotten logs, and cavities beneath logs, upended stumps, or brushpiles were preferred foci of activity. The foraging speed of this species was strikingly slower than that of T. longipes or T. gracilis, especially after rain (see chap. 8). Unlike the other species, large treeshrews would often spend many hours in a tiny sector of their home range and return to it day after day. When I searched these areas I never found fruit sources, so I assume that the animal prey of this species was concentrated in particular microhabitats. All of these intensively used “foraging” sites (a supposition) of T. tana were on the banks of streams or rivers. Large treeshrews also picked up prey from the surface of the substrate and broke into rotten wood (Alim Biun pers. com.). They did not seem to forage on foliage or up in vines or shrubs, although they jumped up onto these to scan the surroundings.
When excavating prey, large treeshrews seemed to dig only shallowly into the top layers of decomposing litter. Digging behavior is strongly programmed into T. tana: captive individuals at the National Zoo that are kept in large cages floored with a deep layer of bark chips, though fat and satiated with food, spend much time walking slowly, probing into the substrate with the nose every few steps, exactly as they do in the wild.
GENERAL TYPES OF INVERTEBRATE PREY
The prey eaten by treeshrews was determined by microscopic examination of feces collected during monthly trapping. Warren Steiner, a Museum Specialist in Entomology at the Smithsonian Institution, undertook this tedious but exacting task, and the results below are products of his great patience and skill. Appendix IV is a complete list of identified taxa.
The many intrinsic sources of variability associated with treeshrew fecal samples make it difficult to quantify the contents in percentages of total diet. One difficulty stems from the short passage time: a fecal sample probably contains only a fraction of a day's foraging activity, so its contents may change quickly from hour to hour and depend on the time of day that the animal was trapped. Different food types have different passage times, so they have different likelihoods of being recorded in a given time frame. Some fruits might be eliminated in scats in less than an hour, insects in several hours (M. Roberts pers. com.), whereas abundant tiny items such as lepidopteran scales or worm setae from a single food item may trickle out over longer periods. Much of the fecal material is an unidentifiable matrix, which is unlikely to represent a proportional sample of the identifiable, because food items intrinsically differ in digestibility. Finally, the amount of material derived from wild meals per scat varied greatly, from one or two ant heads (the rest bait) to large quantities of entirely prey items. A treeshrew may have been in a trap from minutes to hours.
For these reasons, I evaluate the representation of kinds of invertebrates in the diets simply from the number of samples in which items occurred (percent occurrence, or frequency). The number of individuals and species of a given prey taxon that could be recognized in each sample was counted, or, for ants and termites, classed in general abundance ratings (1 = < 10; 2 = 10–20; 3 = 20–30; 4 = >30). The numbers of individuals of each prey type eaten gives an idea of the relative degree to which an animal concentrates or specializes on particular kinds of items.
The analysis of scats shows prominent divergences between treeshrew
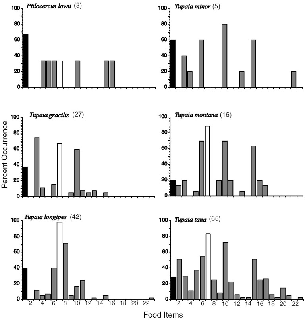
Fig. 5.6. Percent occurrence of food items in treeshrew diets x axis units: 1 = fruit; 2 = earthworms (setae present); 3 = Lepidoptera (larvae); 4 = Lepidoptera (adults); 5 = Coleoptera (larvae); 6 = Coleoptera (adults); 7 = ants; 8 = termites; 9 = wasps; 10 = Orthoptera; 11 = Hemiptera; 12 = Diptera; 13 = Homoptera; 14 = Odonata; 15 = spiders; 16 = millipedes; 17 = centipedes; 18 = uropygids; 19 = phalangids; 20 = scorpions; 21 = crabs/crayfish; 22 = molluscs; 23 = lizards.
| T. gracilis | T. montana | T. longipes | T. minor | T. tana | P. lowii | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Food | (N) | % | (N) | % | (N) | % | (N) | % | (N) | % | (N) | % |
| Fruit | (10) | 37 | (3) | 19 | (17) | 40 | (3) | 60 | (18) | 28 | (2) | 67 |
| Earthworm | (2) | 13 | (33) | 51 | ||||||||
| caterpillar | (20) | 74 | (3) | 19 | (5) | 12 | (2) | 40 | (20) | 31 | ||
| Lepidoptera adult | (3) | 11 | (2) | 5 | (1) | 20 | (6) | 9 | (1) | 33 | ||
| Coleoptera larva | (1) | 6 | (3) | 7 | (24) | 37 | (1) | 33 | ||||
| Coleoptera adult | (4) | 15 | (11) | 69 | (17) | 40 | (3) | 60 | (35) | 54 | (1) | 33 |
| Ants | (18) | 67 | (14) | 88 | (41) | 98 | (54) | 83 | (1) | 33 | ||
| Isoptera | (30) | 71 | (16) | 25 | ||||||||
| Hymenoptera | (1) | 4 | (3) | 19 | (2) | 5 | (5) | 8 | ||||
| Orthoptera | (16) | 59 | (11) | 69 | (7) | 17 | (4) | 80 | (47) | 72 | (1) | 33 |
| Hemiptera | (2) | 7 | (3) | 19 | (10) | 24 | (14) | 22 | ||||
| Diptera | (1) | 4 | (4) | 6 | ||||||||
| Homoptera | (2) | 7 | (1) | 6 | (1) | 2 | (1) | 20 | (1) | 2 | ||
| Odonata | (1) | 2 | ||||||||||
| Aranae | (1) | 4 | (10) | 63 | (2) | 5 | (3) | 60 | (33) | 51 | (1) | 33 |
| Diplopoda | (3) | 19 | (16) | 25 | (1) | 33 | ||||||
| Chilopoda | (2) | 13 | (17) | 26 | ||||||||
| Uropygida | (4) | 6 | ||||||||||
| Phalangida | (1) | 2 | ||||||||||
| Scorpions | (9) | 14 | ||||||||||
| Decapoda | (3) | 5 | ||||||||||
| Mollusca | (1) | 2 | ||||||||||
| Lizard | (1) | 2 | (1) | 2 | ||||||||
| T. minor | T. gracilis | T. longipes | T. tana | T. montana |
|---|---|---|---|---|
| NOTE: Taxa dominant found in more than 50% of scats, in boldface. | ||||
| Orthoptera(80) | Caterpillars(74) | Ants(98) | Ants(83) | Ants(88) |
| Coleoptera adults(60) | Ants(67) | Termites(71) | Orthoptera(72) | Coleoptera(69) |
| Aranae(60) | Orthoptera(59) | Coleoptera(40) | Coleoptera adults(54) | Orthoptera(69) |
| Caterpillars(40) | Coleoptera(15) | Hemiptera(24) | Earthworms(51) | Aranae(63) |
| Lepidoptera, adults(20) | Lepidoptera, adults(11) | Orthoptera(17) | Aranae(51) | Caterpillars(19) |
| Homoptera(20) | Hemiptera(19) | |||
| Hymenoptera, non-ant (19) | ||||
| Millipedes (19) | ||||
PTILOCERCUS LOWII Only three scats were collected from pentails. These included twelve prey items of seven general types (table 5.5, Appendix IV). Beetles, with four individuals, predominated in this tiny sample, which showed only ingestion of a broad range of prey types consistent with an arboreal trunk-foraging mode. This sparse data can be augmented with records from the literature:Lim (1967: 377) reported four P. lowii stomachs to contain “chiefly black ants, along with cockroaches, beetles, earwigs, cicadas, and leaf-insects. One contained remains of a young forest gecko, Gymnodactylus marmorata.”
TUPAIA MINOR Lesser treeshrews were also poorly sampled, with only five scats (feces fell out of arboreal trap sets), including twenty-eight prey of seven types. This small sample nonetheless showed a strong trend: 50 percent of all prey items were Orthoptera of two kinds, crickets and cockroaches; 21 percent were adult beetles; and 10 percent each were caterpillars and spiders. This array again seems to correspond nicely to the observed foraging behavior within arboreal debris, crevices, foliage, and stems. Ants were absent from my sample for this species, but bothDavis (1962) and Lim (1967) found ants in T. minor stomachs, as well as some of the same arthropods I report.
TUPAIA GRACILIS Slender treeshrews were remarkable for the low diversity of their eighty-six prey, from twenty-six scats, which was dominated by just three types, caterpillars (40% of individuals; Appendix IV), ants (23%) and Orthoptera (21%) (fig. 5.5). The major prey was caterpillars, and 55 percent of the samples that included these contained two or more. Although ants were the second most common prey, these were eaten in small numbers, with 89 percent of occurrences at abundance 1 and 11 percent at 2. A single orthopteran was found 87 percent of the time, two in the other 13 percent of samples. Completely absent from the samples were earthworms, termites (Isopoda), millipedes (Diplopoda), and centipedes (Chilopoda), and there was a single spider (Arachnida, Aranae). Ten general prey types were identified in the samples. The prey found in T. gracilis feces correlates well with its observed foraging behavior of surface gleaning, but the dominance of caterpillars points to underleaf scanning as the major foraging surface and mode, a tendency that was not obvious in the field, although I occasionally saw it. The few Orthoptera identified were mainly crickets and cockroaches, both of which are present at all forest levels, but by day these are generally in hidden refuges or cryptically immobile.
TUPAIA LONGIPES Plain treeshrews ate two types of prey in major amounts and three others to a much lesser extent. Ants were the most frequent prey (98% occurrence), and they were also eaten many at a time, with 80 percent of occurrences at abundances of 2 or more and 22 percent at abundance 4. Twelve percent of ant occurrences included broods (eggs, larvae, or cocoons), showing that nests were rifled. Termites were the other main prey of T. longipes, and these were likewise eaten in large numbers (at abundance 2 or more in 50% of scats). Beetles (Coleoptera), bugs (Hemiptera), and Orthoptera were the most frequent, but much more scarce, lesser prey. Eleven of the general prey types were found among 220 prey individuals in forty-two plain treeshrew scats (fig. 5.6), and they had eaten no earthworms, millipedes, or centipedes. A reptile tail tip, probably from a lizard, was in one sample.
The prey of this treeshrew would only partly have been predicted from foraging behavior that I was able to observe. The ground, log, and lianasurface nosing and nibbling of tiny items by T. longipes conforms with ant eating, or random gleaning of beetles and the eight minor prey types. In particular, the relative absence from the diet of hiding arthropods such as cockroaches and spiders, or foliage insects such as caterpillars, concurs with an observed lack of both foliage gleaning or crevice searching in this species. However, I did not see anything in the field that I would have specifically interpreted as termite hunting. The termites eaten are palebodied species, such as Nasutitermes, that are not active on the surface by day. To eat these, the treeshrews would have to open their tunnels or nests (but no brood was found in scats). Perhaps termites are concentrated in the dense treefalls and vine thickets favored by plain treeshrews.
TUPAIA MONTANA Montane treeshrews, like plain treeshrews, ate ants most frequently, and they ate many at once (abundance 2–3 67% of the time), but they ate beetles, spiders, and Orthoptera nearly as often (fig. 5.5) and four additional prey types in almost 20 percent of occurrences each. They ate no termites, but they did eat earthworms, millipedes, and centipedes. In the smaller number of montane treeshrew scats examined (16), I found that they ate twelve general prey types (among 99 individuals), or more than either slender or plain treeshrews. Compared to the latter, montane treeshrews had a more diverse and even diet, less skewed toward particular taxa, although still weighted heavily toward four. This is the only treeshrew for which spiders were a major prey (12% of all items but in 63% of all scats).
The prey array of montane treeshrews shows clearly that they seek
TUPAIA TANA The prey profile of large treeshrews reflects a distinctive feeding strategy (fig. 5.6). Each scat had both more individual arthropods and more general categories of them than did those of other species. Beetles, ants, spiders, and Orthoptera were the most often eaten categories, in common with one or another of the other treeshrews, but several prey types rarely eaten by other treeshrews were important components of large treeshrew scats, including earthworms (51% of scats), centipedes (26%), millipedes (25%), and beetle larvae (37%, mostly elaterid and scarabid). Among the ninety-one individual Orthoptera identified to family, 59 percent are cockroaches and 22 percent are crickets. This was the only species to have eaten arthropods of several other orders or classes, including nineteen scorpions, four uropygids, one phalangid, two crabs, and a crayfish. This eclectic prey range splendidly mirrors the behavior of T. tana as seen in the field. Invertebrates gleaned from the substrate surface are mixed with those from beneath the litter surface (worms) or nocturnal species hidden by day under logs, roots, or leaves (centipedes, termites). The beetle larvae may be broken from rotting wood. Many samples for this species included what seemed to be soil. This may have come from the gut contents of ingested earthworms, or else was swallowed while unearthing prey from the soil. The decapods reflect the foraging along streams that was documented by radio-tracking (see chap. 8).
SPECIFIC TYPES OF INVERTEBRATE PREY
Each treeshrew species ate a unique balance of arthropod prey types, but despite this there was a surprising uniformity among the treeshrews in the more specific taxa of invertebrates eaten (Appendix IV). The most intriguing case was that among thirty-one hemipterans that were identified to family, twenty-five (81%) were Reduviidae (plus two coreids and four cydnids). Other strongly dominant prey taxa were lycosids among spiders, scarabs and weevils among beetles, cockroaches and crickets among Orthoptera, and scolopendrids among centipedes. Considering the vast diversity of arthropods in the tropics, relatively few families were
ABUNDANCE AND SEASONALITY OF INVERTEBRATES
I did not measure the invertebrate prey base during this study, but data on the abundance and phenology of litter invertebrates at Danum Valley were collected in the year before my project during a study of litter decomposition (Burghouts et al. 1992), and some data on numbers of canopy invertebrates are available for another part of Borneo (Stork 1991). These can be used as a rough gauge of whether treeshrews forage randomly or selectively.
A comparison of what treeshrews eat (table 5.5, fig. 5.6) with what is presumably available (fig. 5.7) shows that treeshrews generally eat the most common, nonflying or slow-to-fly large arthropods. Both fast-flying (Diptera, Hymenoptera) and tiny (Collembola, Psocoptera, Thysanoptera, Isopoda, Acarina, etc.) forms are apparently eaten in far fewer than their relative numbers in the environment. Ants are by far the most numerous litter-fauna arthropods (fig. 5.7A), and they are also the most frequent prey in the diets of the three most terrestrial species. Beetles and spiders are likewise dominant both in the habitat and in scat samples. Unlike these, several prey items appear to be taken by particular species of treeshrews in proportions greater than their occurrence. Plain treeshrews seek out termites, and large treeshrews discover cockroaches and crickets with unusual frequency. Unfortunately, studies of the arthropod faunas of understory vegetation, the layer used by T. gracilis, seem to have been done only in the New World. One such study in Panama (Greenberg and Gradwohl 1980) found understory leaf arthropods in the following rank orders and numbers: 1, spiders (350); 2, beetles (304); 3, Homoptera (159); 4, Diptera (86); 5, Orthoptera (85); 6, Hemiptera (61); 7, lepidopteran adults (56); 8, lepidopteran larvae (31). Because caterpillars have not been shown to dominate the arthropod faunas of any tropical rainforest level, it is likely that the high frequency of caterpillars in T. gracilis scat samples is a result of specialized hunting behavior. The predilection of several treeshrews for reduviid bugs also seems to be the product of more
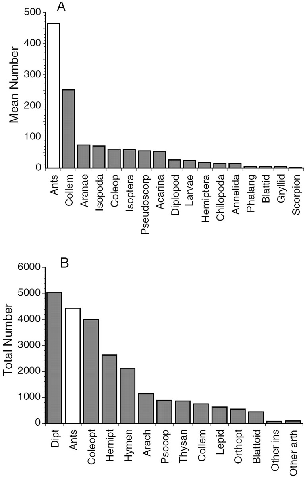
Fig. 5.7. A, average numbers of arthropods in standard samples (25 cmdia x 20 cm deep) of litter collected at Danum Valley (data from Burghoutset al. 1992). B, total numbers of arthropods collected from ten Borneantrees by canopy fogging (data from Stork 1991); most Lepidoptera arelarvae (558), and the majority of Orthoptera other than Blattoidea (listedseparately) are crickets (Gryllids, 336).
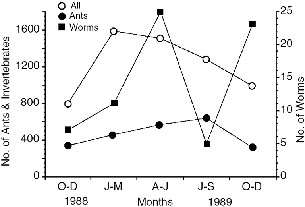
Fig. 5.8. Numbers of litter invertebrates collected in standard litter samples during three-month periods in 1988–89 (Burghouts et al. 1992; T. Burghouts pers. com. of data).
The pattern of litter arthropod abundance shows about a two- to fivefold seasonal variation in invertebrate numbers (fig. 5.8). The pattern of earthworm numbers generally follows the rainfall pattern for that year, indicating that earthworms are most numerous in the surface litter during wet periods. During periods of drought, they may burrow down out of reach. Arthropods in general had highest numbers from January to March and gradually declined to minimum numbers during October and November, but ants exhibit relatively little seasonal variation (Burghouts et al. 1992). This should make ants a more reliable food source.
DISCUSSION OF FOOD HABITS
Some information on the food habits of treeshrews is reported in the literature (Davis 1962; D'Souza 1972; Harrison 1954; Langham 1982; Lim
In frugivory I saw no evident ecological separation between the species, other than the spatial distinction imposed by the arboreality of the two smallest treeshrews. Two species (P. lowii and T. tana) seemed to be less drawn to fruiting trees than the others (see also chap. 8), but as actual amounts eaten were not measured, the evidence is circumstantial. The seasonal pattern of fruit eating seemed to follow the habitatwide overall fruiting phenology, with the exception that frugivory seemed to increase in August and September 1991, when overall fruit species availability was low. Fruit eating was facultative, in that during some months treeshrews were not detected eating any. Nonetheless, I will suggest below (chap. 10) that fruit is a most critical portion of the diet.
In contrast to their frugivory, in insectivory the six species showed strong differences in prey composition. These differences arise from distinctly divergent foraging patterns and sites (table 5.7). An unexpected discovery was that T. montana, which closely resembles T. longipes in size and superficial appearance, does not resemble it in foraging mode and diet but instead is much like T. tana, which has morphological adaptations for digging (long claws) and probing under the litter (long snout) that are lacking in the montane species. Perhaps the deep moss substrate of montane forest requires less specialized digging apparatus than the soil of the lowlands. Phylogenetically, mountain treeshrews and large treeshrews are sister taxa well removed from the lineage of plain treeshrews (Han, Sheldon, and Stuebing n.d.), so their dietary similarities reflect a shared evolutionary relationship.
The structuring of feeding roles in treeshrews closely parallels the way that birds segregate into highly specific insect-feeding guilds (e.g., Robinson, Terborgh, and Munn 1990). The similarity in insects eaten (ants, reduviids, lycosids, cockroaches) shows that the basic sensory and preycatching modalities have remained alike in different treeshrew species, while the foraging sites have diverged, to create a community of morphologically quite similar but ecologically differentiated species. In the following section and chapters we shall see that the foraging mode and
| Species | Chief Foraging Site | Dominant or Special Prey |
|---|---|---|
| P. lowii | Arboreal: tree trunks and stems, | Wide variety |
| foliage of treelets | ||
| T. minor | Arboreal: lianas and living foliage, | Orthoptera, |
| dead leaves | Aranae, beetles, | |
| caterpillars | ||
| T. gracilis | Terrestrial and understory: | Caterpillars, |
| undersurface of shrub foliage, | Orthoptera, ants | |
| ground surface, low plants | ||
| T. montana | Terrestrial: under logs, | Ants, Coleoptera, |
| digs in litter | Orthoptera, Aranae, | |
| Myriapoda | ||
| T. longipes | Terrestrial and surface of logs: | Ants, termites |
| ground surface | ||
| T. tana | Terrestrial and logs: digs under | Ants, cockroaches, |
| litter, logs, and roots | earthworms, Aranae, | |
| Myriapoda, Crustacea |
ECOLOGICAL CONSEQUENCES OF THE
DIETARY CONSTRAINTS OF TREESHREWS
The rapid passage time of fruit through the gut of Tupaia species (20–60 min depending on animal size) and batlike fruit-processing behaviors, both associated with the simple gross morphology of their digestive tracts, make it likely that only the most readily available (soluble) nutrients are extracted from fruit. The dietary role of fruit may largely be to provide quick and economically acquired calories and nutrients that are scarce in arthropods, particularly calcium. Tupaia feed on fruit in short, repeated bouts. Wild treeshrews may fill up again on fruit as soon as the previous batch has passed through the digestive tract. By resting near a tree and feeding at intervals, a treeshrew may achieve maximum intake for minimum effort. An outcome of this feeding mode is that the treeshrews seemingly must eat frequently, with repercussions on the general daily activity patterns (see chap. 7).
The use of fruit by treeshrews can without quibble be called primitive, as it is about the most rudimentary possible, the result of possession
INTERSPECIFIC INTERACTIONS AND FORAGING.
COMPETITION FOR FRUIT AMONG TREESHREWS
The treeshrew species seemed to overlap broadly, perhaps completely, in the species of fruit that they ate. Theoretically, they thus compete directly for this resource, although the arboreal and terrestrial species exploit spatially and temporally separated portions of the same fruit supply, of which the arboreal species have first choice, without competition from groundforaging species (the same individual fruits available to T. minor in a tree, if not eaten, eventually fall and become available to the terrestrial species).
I could watch interactions of arboreal T. minor in fruit trees without difficulty, but there they had no competition from other treeshrews. Several times I tried watching the ground from discreet lookouts near fig trees (with and without a blind), but both treeshrews and larger mammals tended to be so wary that I collected little useful data. I was more successful at watching interactions at artificial fruit sources (bait stations provisioned with banana for a couple of weeks) from a blind (Poring) or from a distance (Danum). Squirrels and treeshrews came to these baits.
At bait stations that were set up so that only a single animal at a time could feed there (bananas fixed to the end of a single pole), I saw encounters between T. minor, T. gracilis, T. longipes, and T. tana. Interactions between individuals of different species were all of the simplest and most straightforward kind: the larger species would simply displace the smaller, which would lurk in the background and wait to return when the larger species left. A larger species that arrived when a smaller one was feeding would displace it from the bait by simply approaching. I saw no pursuit of another species that was displaced, and a larger species feeding at the bait appeared to ignore members of other species that hovered a few meters away. The smallest species, T. minor, would often suddenly appear at the bait after the departure of another taxon, as if it had been waiting in the wings for its chance. Individuals of all species fed at bait in short bouts of a few minutes, then left and returned later, consistent with the ingestion pattern described above. The behavior at baits, and
COMPETITION FOR FRUIT
AMONG TREESHREWS AND OTHER TAXA
The fruit species chosen by treeshrews were also favorites of many other mammals and/or birds, and it is unlikely that treeshrews are of more than minor importance as seed dispersers in intact forest. A notable exception to this is for the peculiar fruits of Rafflesia spp., which have characteristics that may be best suited to treeshrew dispersal (Emmons, Nais, and Biun 1991). In secondary or highly disturbed forest, where larger mammals and birds have been extirpated by hunting or habitat loss, treeshrews could acquire a more preeminent dispersal role, especially T. longipes, which persists well in severely altered forests (chap. 4; see also Stuebing and Gasis 1989). I recorded all mammal species that I saw at fruit sources (Appendix V), and I also usually noted whether birds fed on the fruits. These were incidental notes made while radiotracking or working on other tasks in the forest, so most are daytime records. Unfortunately, there are no data on the fruits eaten by bats at Danum. The lists of other taxa are incomplete: many larger mammals were shy, did not have home ranges on the study areas, or, at Poring, were absent or rare (e.g., there were no gibbons or orangutans and scarcely any terrestrial large mammals there). I saw other taxa using the following numbers of fruit species also eaten by treeshrews: birds, 11; Prevost's squirrel (Callosciurus prevosti), 7; other squirrels, 3; artiodactyla, 3; monkeys, 3; and civets, 3. I suspect that most generalized frugivores (e.g., monkeys, civets, mouse deer) probably eat virtually all of the small, sweet fruits selected by treeshrews. Nonetheless, I think the data give the correct general relationship—that treeshrews show most overlap with birds in fruit species eaten. Despite this, two of the “keystone” species of fruits for treeshrews, Dialium indum and Alangium ebe- naceum, were strictly mammal-exploited fruits, which attracted large numbers of other mammalian taxa, from monkeys to deer (Appendix V), but apparently no birds.
Because treeshrews and squirrels are superficial look-alikes, it is of interest
As well as competing for fruits, in at least one case Prevost's squirrel facilitated feeding for treeshrews. The key species Alangium ebenaceum has a small, hard, green fruit. Callosciurus prevosti removed and ate the seeds and dropped a rain of discarded fruit fragments to the ground, where the three terrestrial tupaias fed on them. Treeshrews seemed to be attracted to Alangium trees where a squirrel was feeding, and I believe that the fruits are too hard for treeshrews to penetrate with their weak jaws, and they may only be able to eat broken fragments. An orangutan chewed and sucked off the edible aril and spat out the seed and pericarp, leaving little of value for other taxa.
In the wild treeshrews and squirrels did not seem to pay any attention to each other, but at an artificial banana bait site with room for one consumer, a horse-tailed squirrel, Sundasciurus hippurus (approx. 300 g), completely dominated both an adult T. tana and a T. longipes. The treeshrews circled in the background, once even growling, and would approach the banana only after the squirrel left. The squirrel did not chase or show any overt gestures of aggression but simply went to the banana (not toward the treeshrew) and the treeshrew left. Even after eating its fill, the squirrel deliberately kept the treeshrews away by dashing in to stand by the banana whenever it saw a treeshrew approach. At another bait site, a T. longipes chased off and preempted the banana from a Sun- dasciurus lowii of about half its weight (approx. 100 g), and this squirrel
The chief indirect rivals of treeshrews for fruit, therefore, seemed to be birds and Prevost's squirrels, followed by monkeys, artiodactyla, and civets. In a direct contest at a limited resource, such as the single fruit of a Rafflesia(Emmons, Nais, and Biun 1991), squirrels would probably outcompete treeshrews of the same weight; but this situation is likely to be too rare to have ecological significance except when resources are marginal.
INTERSPECIFIC COMPETITION FOR INVERTEBRATES
In contrast to fruit, for which the consumers can be directly seen for each fruit species, most foragers on invertebrates can be only indirectly assessed by where, when, and how they forage. Two insectivores that take invertebrates from inside rolled leaves at the tips of branches can reasonably be assumed to compete for the same prey. As Root (1967) developed in his classic paper, competition among vertebrates for invertebrates can best be studied within the paradigm of feeding guilds.
We saw above that each treeshrew species has a distinctive tactic for feeding on invertebrates. Each treeshrew that forages in the same microhabitat has a quantitatively and to some extent qualitatively divergent invertebrate diet that should effectively remove it from strong competition with any other: among the three terrestrial species that share the lowland forest floor, one gleans the undergrowth foliage for caterpillars, one gleans ants and termites from substrates other than foliage, and the third digs into the decomposing litter and woody substrate for earthworms and feeds on noxious forms and decapods not taken by the other two. During the study, the only other mammalian species that I saw that foraged somewhat like treeshrews in the same habitat were the diurnal mongooses Herpestes brachyura and H. semitorquatus, which meandered through the forest, nosing the ground, frequently digging into it for prey. These are likely to compete somewhat with T. tana for invertebrates hidden under the surface litter. Unlike large treeshrews, I saw these mongooses enthusiastically digging quite deep holes in the clay below the litter, but I never saw what they pursued.
One tiny squirrel, Sundasciurus jentinki, hunts insects in the foliage of small trees, as does T. minor, but it only occurs above about 1,000 m
Birds were the most obvious potential competitors with treeshrews for arthropods. While following treeshrews, I often watched the foraging behavior of common birds, and it was evident that babblers (Timaliidae) often foraged for prey in the same locations as treeshrews do. Like treeshrews, babblers chiefly pick sedentary prey off the foraging substrate and in particular foraged for hidden, immobile prey in sites such as the undersurfaces of curled leaves and interior parts of plants. Bulbuls, in contrast, tended to sally for prey spotted from a perch, while leafbirds and cuckoos took prey from upper, sunlit leaves, places little used by treeshrews, and so forth. The babblers often foraged in mixed flocks of species organized in distinct guilds. Table 5.8 outlines the habits of a few common species of babblers at Danum and the treeshrews with which they are most likely to compete for prey. Birds of this family are extremely common and likely to have a profound ecological impact. A few other bird taxa are also likely to have overlapping foraging roles with particular treeshrews: the pittas (Pittidae) are common terrestrial birds that favor the dank undergrowth microhabitats loved by treeshrews. They feed on invertebrates, especially ants and beetles (Lambert and Woodcock 1996), so they may compete with T. tana and T. gracilis but especially T. longipes for arthropods. Nuthatches (Sitta
| Babbler | Level | Behavior | Treeshrew |
|---|---|---|---|
| NOTE: Includes only a partial list of the babblers known from the area (see Anon. 1993, for a complete list). | |||
| Black-throated wren babbler, Napothera atrigularis | Ground, under litter | Flips over leaves with beak and looks beneath | T. tana |
| Striped wren babbler, Kenopia striata | Ground | Lifts up edges of dead leaves, pushes head and beak beneath | T. tana |
| Black-capped babbler, Pellorneum capistratum | Ground | Hops along gleaning surface of litter | T. longipes, T. gracilis T. tana |
| Short-tailed babbler, Trichasoma malaccense | Ground | T. tana, T. longipes | |
| Horsefeld's babbler, Trichasoma sepiarium | 0–3 m | Understory stems, leaf undersides, saplings | T. gracilis |
| Chestnut-winged babbler, Stachyris erythroptera | Low to midstory | Open forest saplings and leaf bottoms, hanging dead leaves | T. gracilis, T. minor, P. lowii |
| Scimitar babbler, Pomatorhinus montanus | Low to midstory | Peers into crevices and holes in trunks and branches | T. longipes, T. minor, P. lowii |
| Gray-headed babbler, Stachyris poliocephela | Midstory | Dead-leaf bunches | T. minor, P. lowii |
| Chestnut-rumped babbler, Stachyris maculata | Mid-high | Disruptive foliage gleaner | T. minor |
| Fluffy-backed tit babbler, Macronous ptilosus | Low to midstory | Dense vine tangles and festoons | T. minor |
| Scaly-crowned babbler, Malacopteron cinereum | Midstory | Undersides of terminal leaves and leaf tips | T. minor |
A FORAGING ASSOCIATION
BETWEEN trEESHREWS AND BIRDS
From the start of the study in 1989, I noted that actively foraging lesser treeshrews were often in the company of mixed-species flocks of birds.
[7 Dec. 90] 1130 h: T. minor M91 foraging at 10 m in a dense vertical vine festoon, above a yellow-bellied bulbul; 1200 h: still with birds, 15 m up; 1211 h: still with bird flock, foraging in vines 8 m; 1217 h: M91 seen still with birds 8 m, reaches out head and peers at leaf tips, drongo below, then goes to canopy 10 m (my following broken by heavy rain; M91 went with the bird flock 50–75 m in 3/4 hour). When below, the drongo and bulbul kept looking up at him; 1253 h: still a bird flock around him.
[10 Dec.] 1020 h: T. minor M91 insect hunting overhead, a bulbul and a young Sundasciurus hippurus also hunting insects near. Goes inside a large dead leaf bunch at 3–4 m. 1044 h: M91 hunting with a bird flock, moving horizontally at 10 m in a dense vine tangle, grey-cheeked bulbuls below. Bulbuls following M91, letting him get ahead, then flying to perch below, etc., etc. They have followed 20 m so far, only bulbuls following M91, other birds go in opposite direction off territory.
[21 Dec.] 0754 h: A T. minor hunting vertically in a vine tangle in the subcanopy at 10 m, a yellow-bellied bulbul closely following underneath 1.5–2 m below, hawking insects.
[13 Mar. 1991] 1059 h: Unmarked T. minor foraging at 8–10 m, mostly vertically, sniffing in dead, hung up and curled leaves. Yellow-bellied bulbul (banded light blue/dark blue) starts to follow; 1122 h: bulbul still following, T. minor at 20 m.
[21 June] 1245 h: Unidentified T. minor climbing up and down a gnarled trunk, sniffs in holes. As I watch, a yellow-bellied bulbul flies in underneath.
[20 Apr.] 0811 h: A T. minor is foraging at 1 m in a low vine blanket in a treefall gap; a yellow-bellied bulbul is waiting above, then flies in under when T. m. climbs up high enough for the bird to fit in below.
[13 July] 0914 h: T. minor F294 traveling/foraging with a male very close, in a dense vine festoon, foraging in dead leaf bunches. A male racket-tailed drongo briefly below, then a pair of drongos follow T. m. to the next tree, and the next, in the vine subcanopy 4–10 m; 0947 h: T. m. comes back to same vine tangle, the pair of drongos comes back, still following.
[15 July] 0758 h: An unmarked T. minor male foraging at 8 m moving horizontally in a vine tangle, with a pair of RT drongos beneath; a female T. m. comes by the same place and the drongos switch to following her instead.
[3 Aug.] 0830 h: T. minor M172 in vines 4 m; 0845 h: a mixed bird flock arrives, RT drongos attach to T. m. and start to follow, separate from rest of flock.
The field notes above illustrate all of the basic features of the association between birds and treeshrews. Birds of two species that are regularly found in mixed-species flocks, yellow-bellied bulbuls (Criniger phaeo- cephalus) and racket-tailed drongos (Dicrurus paradiseus), seek out a foraging T. minor and perch about 1.5 to 2 m below it, intently watching
| With YB Bulbuls | With GC Bulbuls | With RT Drongos | With Flock but without Followers | |
|---|---|---|---|---|
| NOTE: Data from 7 December 1990 to 30 September 1991 and one observation from 1989. Before December 1990 I did not recognize the association with particular species. Total observations of T. minor during this period = 140; total noted with any birds = 42 (30% of sightings). Table lists numbers of times obvious bulbuls or drongos were observed following the treeshrew. YB = Yellow-bellied bulbul (Criniger phaeocephalus); GC = Gray-cheeked bulbul (Criniger bres); RT = Racket-tailed drongo (Dicrurus paradiseus). | ||||
| With mixed flock | 4 | 1 | 4 | 7 |
| Without flock | 12 | 8 | ||
This enterprise must be a successful strategy for the birds, because when locked onto a treeshrew, they abandon their mixed flock to follow it. The pattern of observations (table 5.9) shows that both species of follower birds hunted under lesser treeshrews alone two or three times as often as when they were with mixed bird flocks and that, conversely, lesser treeshrews, when accompanied by birds, more often than not had evident followers hunting below them. On all occasions except that described above for 7 December, birds of only one species at a time followed a treeshrew. Once I saw a yellow-bellied bulbul foraging below a chestnut-winged babbler (Stachyris erythroptera) with the same behavior exhibited when foraging below a treeshrew.
During the seven months when the behavior of followers was recorded, there was a seasonal shift in the bird species that accompanied lesser treeshrews (fig. 5.9). I saw no drongos follow in April, and no bulbuls in May, July, or August. There seem to be no published reports of breeding cycles for these birds in Borneo, but in a large series of females collected in Sabah by F. Sheldon (pers. com.) during all months, the only reproductive female yellow-bellied bulbul (which had just laid an egg) was collected in July and the one racket-tailed drongo was collected in April. This hints that each species may have stopped following when it was busy nesting.
Apart from the clear foraging link of the follower birds with T. mi- nor, I was not able to identify the basis for the general associations between
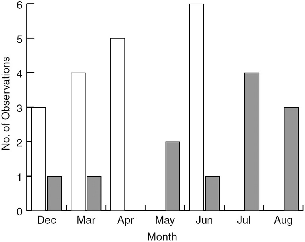
Fig. 5.9. Number of times that bulbuls (open bars) and drongos (shaded bars) were seen following T. minor at Danum Valley in months of 1991. The amount of observation time per month was not equal; the data show the relative monthly contributions of the two bird taxa.
APPENDIX: METHODS
The feces in the trap were collected each time a treeshrew was captured (large clean leaves were placed under the traps to catch them), but not all captures produced
As a cautionary note for other field biologists, I reemphasize that it would not have been possible to predict the differentiating features of treeshrew diets or foraging modes either from their morphology or from field observations. Only the tedious labor of microscopic examination of fecal samples yielded the key information about what the species actually ate.